THIS? Read Section Part E ges 803 - 817); Watch KCV 18. 4B, IWE Calculate the pH at one-half of the equivalence point. L sample of 0.165 M propanoic acid ( 3 x 10 5) is titrated with 0.300 Express your answer using two decimal places. pH = Submit Request Answer Part F Calculate the pH at 20 mL of added base.
THIS? Read Section Part E ges 803 - 817); Watch KCV 18. 4B, IWE Calculate the pH at one-half of the equivalence point. L sample of 0.165 M propanoic acid ( 3 x 10 5) is titrated with 0.300 Express your answer using two decimal places. pH = Submit Request Answer Part F Calculate the pH at 20 mL of added base.
Chapter14: Principles Of Neutralization Titrations
Section: Chapter Questions
Problem 14.38QAP
Related questions
Question
100%
Transcribed Image Text:MISSED THIS? Read Section
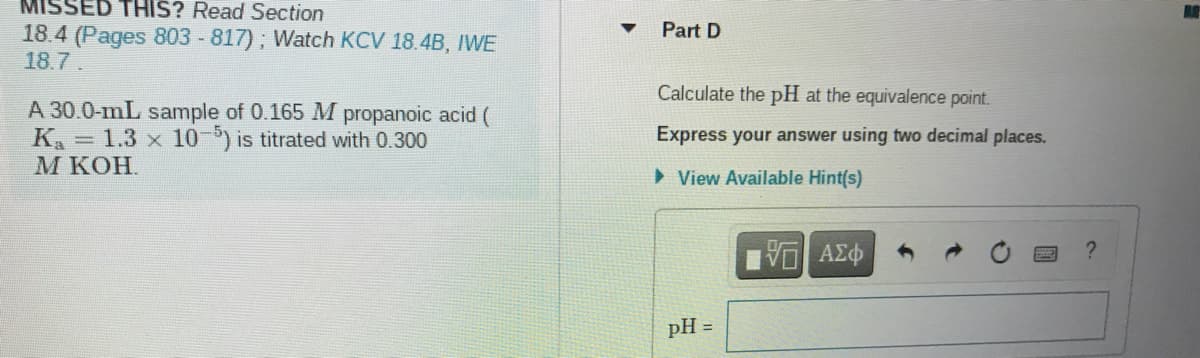
Part D
18.4 (Pages 803 -817); Watch KCV 18.4B, IWE
18.7.
Calculate the pH at the equivalence point.
A 30.0-mL sample of 0.165 M propanoic acid (
K = 1.3 x 10-) is titrated with 0.300
М кОН.
Express your answer using two decimal places.
> View Available Hint(s)
pH =
Transcribed Image Text:<Chapter 18 Homework
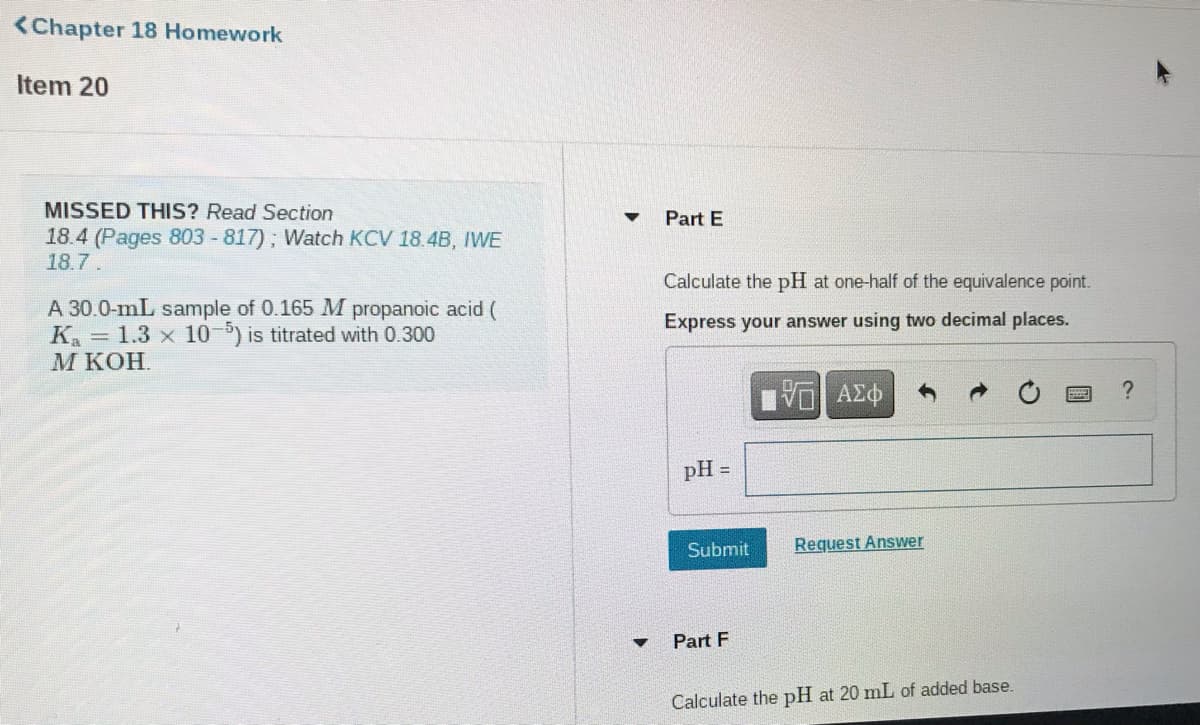
Item 20
MISSED THIS? Read Section
Part E
18.4 (Pages 803 -817); Watch KCV 18.4B, IWE
18.7.
Calculate the pH at one-half of the equivalence point.
A 30.0-mL sample of 0.165 M propanoic acid (
K. = 1.3 x 10-5) is titrated with 0.300
М КОН.
Express your answer using two decimal places.
pH
Submit
Request Answer
Part F
Calculate the pH at 20 mL of added base.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning