Three beakers are placed in a sealed container illustrated below. A B C Initially all the beakers are filled to the same volume with the following A: solution of NaCI (X = 0.01) in water B: solution of CgH1206 (X = 0.01) in water C: pure water After time has passed, which beaker would contain the largest volume of liquid. a. Beaker A and c b. Beaker A c. Beaker A and B d. All would have the same volume е. Вeaker B f. Beaker C O g. Beaker B and C
Three beakers are placed in a sealed container illustrated below. A B C Initially all the beakers are filled to the same volume with the following A: solution of NaCI (X = 0.01) in water B: solution of CgH1206 (X = 0.01) in water C: pure water After time has passed, which beaker would contain the largest volume of liquid. a. Beaker A and c b. Beaker A c. Beaker A and B d. All would have the same volume е. Вeaker B f. Beaker C O g. Beaker B and C
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter14: Equilibria In Acid-base Solutions
Section: Chapter Questions
Problem 64QAP
Related questions
Question
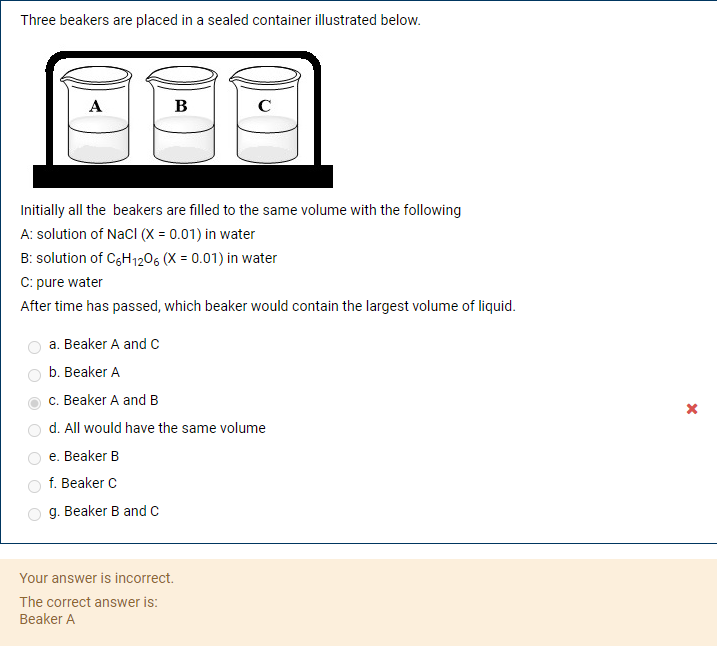
Transcribed Image Text:Three beakers are placed in a sealed container illustrated below.
А
B
C
Initially all the beakers are filled to the same volume with the following
A: solution of NaCI (X = 0.01) in water
B: solution of CgH1206 (X = 0.01) in water
C: pure water
After time has passed, which beaker would contain the largest volume of liquid.
a. Beaker A and C
b. Beaker A
c. Beaker A and B
d. All would have the same volume
е. Вeaker B
f. Beaker C
g. Beaker B and c
Your answer is incorrect.
The correct answer is:
Beaker A
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning
