Q: 2 N2O54 NO2 + O2 the following data have been obtained: [N2O5], M 0.603 0.340 0.191 0.108 time,…
A:
Q: Using Figure 14.3, calculate the instantaneous rate of disappearance of C4H9Cl at t = 0 s (the…
A: The instantaneous rate for the reaction is calculated by the formula given below.
Q: [RCOOH], M [R’OH], M (HA], M Initial Rate, M-min¨ 0.35 0.35 0.50 4.60 0.62 0.35 0.50 8.14 0.35 0.81…
A: Here we have to apply the rate law Rate law=k[RCOOH]a[R'OH]b[HA]c
Q: What is the average rate over the first 80 minutes, in M • min^-1 a. 0.81 b. 0.94 c. 1.0 d. 1.23
A:
Q: NO2(g) → NO(g) + ½ O2(g) Time, s [NO2], M 0.0 0.01000 50.0 0.007
A: Chemical kinetics is branch of chemistry in which we deal with speed at which rate of reaction…
Q: Use the data in the table below to answer the following questions. Reaction : 4.0 M Acetone (in mL)…
A: We are given data to determine the rate of the reaction or the rate law.
Q: Dinitrogen pentoxide (N2O5) decomposes as follows to nitrogen dioxide and nitrogen trioxide:…
A: Rate of a particular reaction is the concentration change of reactant or product divided by change…
Q: A 0.100 M solution decomposes by second order kinetics with a rate constant 0.400/(M⋅min).…
A: Given-> Initial concentration = 0.100 M Final concentration = 0.0200 M Rate constant (K) = 0.400…
Q: 0.075 0.050 0.025 0.000 0 10 15 Time (s) The average rate from t= 15.0 s to t= 20.0 s = 0.0030 M/s.…
A: Average rate of reaction is the change in concentration of reactant or product divided by time…
Q: For the gas phase decomposition of nitrosyl chloride at 400 K 2 NOCI 2 NO + Cl2 the average rate of…
A: Rate of reaction :- The decrease in concentration of reactants with time or increase in…
Q: A certain reactant is consumed in the course of a chemical reaction such that the reactant that…
A: Given data Concentration of reactant (in Molarity , M) is given by the following equation…
Q: Give two reasons to measure initial rates in a kinetics study.
A: When the chemical reaction takes place, the reactants are consumed and the products are formed.…
Q: A reaction is known to follow first-order kinetics and has a rate constant of (6.3700x10^-2) min1.…
A:
Q: For the gas phase decomposition of cyclobutane, (CH2)4----->2 C2H4 the rate constant in s-1 has…
A: With datas given By using Arrhenius equation the problem is solved as follows
Q: Consider the table of data collected for the reaction A Products. Determine the magnitude (value) of…
A:
Q: 5- 50 100 150 200 250 300 seconds created Is O, being created or destroyed by the chemical O…
A: The pressure v/s time curve of O2 is given as,
Q: average rate
A:
Q: Time (s) Concentration of A (M) Determine the average rate of change of B from t = 0 s to :322 s.…
A: The average rate of reaction can be expressed as follows-
Q: Determine the average rate of change of B from ?=0 s to ?=312 s. A⟶2B Time (s) Concentration of A…
A: Given information:
Q: Using the following rate law and the experimental values given in the question figures, calculate k…
A: Rate laws, also known as rate equations, are mathematical formulas that define the connection among…
Q: The data listed in the table below were obtained from the following decomposition : A products *…
A: For second order reaction- 1[A] = 1[A0] + Ktcomparing this is y = mx + cy =1[A] m = slope = Kx = tc…
Q: he decomposition of an organic molecule with the formula C4H6 at 326°C is measured as the decrease…
A: The data for the decomposition of the organic molecule will be given as follows: Time (s) [C4H6]…
Q: Upload your solution for the derivation of the following Integral form of the rate law: 1. zero…
A:
Q: Consider the reaction and the information in the table provided. Calculate the average rate of…
A:
Q: A reaction is made up in the following way: 16 mL of 4.1 M acetone + 15 mL of 1.4 M HCI + 19 mL of…
A: With datas given First we arw calculating initial concentration of I2 in the reaction mixture and…
Q: Time (s) Concentration of A (M) 0 0.800 126 0.490 252 0.180 Determine the average rate of change…
A: Given: Time (s). Concentration of A (M) 0. 0.800 126 0.490…
Q: Consider the reaction 2A ----> B. The following data has been collected during an experiment: Time…
A: Given that, a chemical reaction is 2A →B. Also, the given data for the experiment is Time (s) 0.0…
Q: For the gas phase decomposition of dimethyl ether at 500 °C CH3OCH3CH4 + H2 + CO the following…
A: Average rate of disappearance calculation:
Q: Consider the table of data collected for the reaction A → Products. Determine the magnitude (value)…
A: From graph we found the equation of slope line y = -0.1770x + 0.4215 And R2 = 0.9320
Q: Given the following data, determine the overall rate order: А + В - 2с" experiment [A], M [B], М…
A:
Q: Consider the reaction: NO2 + 03 → NO3 + 02 [NO,], M [02], M Rate, M/s 6.30 x 10-3 Experiment 1 0.21…
A: Reaction :- NO2 + O3 ---> NO3 + O2 Reaction is balanced Rate = (1/coefficient)…
Q: A reaction is known to follow first-order kinetics. What is the half-life (in seconds) if it takes…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Determine the average rate of change of B from t = 0 s to Time (s) Concentration of A (M) t = 302 s.…
A: The average rate reaction can be equivalent to the mathematical ratio of the concentration change to…
Q: For the gas phase decomposition of sulfuryl chloride at 600 K SO,C12→SO2 + Cl2 the following data…
A: Average Rate=6.89*10-6Mmin-1
Q: 4PH3 ->> P4 + 6H₂ What is the rate of disappearance of PH3 from 0 to 75 seconds? Time [PH₂] [P4]…
A:
Q: This problem is only appropriate if you have taken calculus. For a zero-order reaction described by…
A: Given, the zero-order reaction: aA → products We have to find the integrated rate equation using the…
Q: NO,(0) + O3(g) → NO3(g) + O,(g) [NO2li (M) 0.1 0.1 Initial Rate (ms1) [O3li (M) 0.33 1 1.42 2 0.66…
A:
Q: Determine the average rate of change of B from ?=0 to ?=272 s. A⟶2B Time (s) Concentration of…
A:
Q: X2Y2 + 2Z + 2Y→ Z2 + 2Y2X Trial [X2Y2 ], (M) [Z], (M) [Y], (M) Initial Rate (M/min) 1 0.25 0.11 0.33…
A: The rate of the reaction is referred to the change in the molar concentration in the distinct…
Q: Consider the table of data collected for the reaction A → Products. Determine the magnitude (value)…
A: Given: Time (s) [A] M ln[A] 0.00 0.600 -0.511 1.50 0.325 -1.124 3.00 0.178 -1.726 4.50…
Q: The reaction of ammonia decomposition on a patina surface is: 2NH3(g) àN2(g)+3H2(g) Is a zero-order…
A: Since the reaction is given 0th order reaction. Hence the rate law of reaction is Rate = K = rate…
Q: 1. From the following data find the instantaneous rate at 6 minute. 5.0 Time 3 5.1 7.6 10.2 13.3…
A: Since you have asked multiple questions questions, we will solve the first question for you. If you…
Q: [S208 2-J, M [S203 2-J, M 0.08000 8.0*10-4 0.04000 8.0*10-4 0.02000 8.0*10-4 0.04000 8.0*10-4…
A: The given experimental data is for iodine clock reaction. The balanced equation for iodine clock…
Q: Of the units below __ are appropriate or a third order reaction rate constant. M-2s-1 Ms-1…
A: We know that, for n-th order reaction , Rate = k [A]n or, k = Rate /…
Q: Consider the table of data collected for the reaction A → Products. Determine the magnitude (value)…
A: Time (s) [A] ln[A] 1/[A] 0.00 0.450 -0.7985 2.22 0.50 0.300 -1.204 3.33…
Q: Consider the following data: CO (g) + Cl2(g) --> COCI2 (g) [CO] (M) Experiment [Cl,] (M) Initial…
A: The rate law for the given reaction can be written as: rate=kCOxCl2y ......(1) Here, x is the order…
Q: From a plot or reasonable estimate, what is the instantaneous rate of change of A versus time at 10…
A: The instantaneous rate is the rate at that particular instant and is given by the slope of the line…
Q: Referring to Question 2 in the Feedback Form, calculate the rate constant (k) for the reaction of…
A: We would use the equation which is used to plot ln(abs) vs time plot.
Q: Determine the average rate of change of B from t = 0 s to Time (s) Concentration of A (M) t = 282 s.…
A:
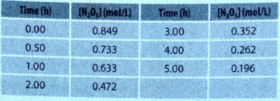
Calculate the average rate over the interval 0 to 5.0 h. Compare your result with the average rates over the intervals 1.0 to 4.0 h and 2.0 to 3.0 h, all of which have the same midpoint (2.5 h from the start).
Step by step
Solved in 4 steps

- The decomposition of aqueous sucrose to form the isomers glucose and fructose is a common organic reaction, which requires a strong catalyst: C12H22O11(aq) + H2O(l) → 2C6H12O6(aq). The following data were collected during the process: Time (min) [C12H22O11] (mol/L) 0 0.316 39 0.274 80 0.238 140 0.190 210 0.146 Use the following grids to plot three separate graphs of [C12H22O11], ln[C12H22O11], and 1/[C12H22O11] against time to determine the reaction order. (a)Write the rate law expression in its general form (water should not be included). (b)Determine the rate constant with units. (c)Determine the half-life. (d)If we performed a new trial with an initial concentration of sucrose of 0.400 mol/L, what concentration would remain after 4.0 h has passed?Time(s) [X3] (mole/L) 0 0.600 200 0.458 400 0.362 600 0.281 800 0.204 1000 0.175 1200 0.134 1400 0.104 1600 0.083 1800 0.063 2000 0.047 plot ln[X3] vs time plot 1/[X3] vs timeUse this data to determine the value of the rate constant. Ex # [A] (M) [B] (M) Rate (M/hr) 1 .240 .120 2.00 2 .120 .120 .500 3 .240 .0600 1.00 Question 4 options: 289 per hour 579 per hour 8.3 per hour 0.0144 per hour 69.4 per hour .00346 per hour 0.060 per hour 0.0173 per hour 17 per hour 0.12 per hour
- Please answer questions 4a, 4b, and 4c. The questions are provided in the attached image. Thank you.Determine the average rate of change of BB from ?=0 s�=0 s to ?=392 s.�=392 s. A⟶2BA⟶2B Time (s) Concentration of A (M) 0 0.7300.730 196196 0.4450.445 392392 0.160For the fictitious reaction: AB → A + B Some data was collected measuring the [AB] at certain time intervals. This data was then graphed as described below. A graph of 1/[AB] versus time (s) gives a straight line with the equation: y = 0.0052x + 0.1135 and a R2 = 0.9995. A graph of ln[AB] versus time (s) gives a straight line with the equation: y = -0.0068x + 1.725 and a R2 = 0.8925. Assume the data doesn't fit a zero order reaction. The rate law is: Rate = k[AB]2 Find: What is the value of the rate constant for the given reaction? Select one: a. k = 0.0157 Ms-1 b. k = 0.0052 M-1s-1 c. k = 0.0068 s-1 d. There is no way to determine the value of the rate constant from the given information.
- In aqueous solution, chlorine dioxide oxidizes iodide ion to iodine and chlorine dioxide is reduced to chlorite ion. 2ClO2 (aq) + 2 I- (aq) ----> 2ClO2- (aq) + I2 (aq) The concentration in molarity, of ClO2 was measured at various time points. The data was plotted and the plot of [1/[ClO2] versus time in seconds was most linear. The linear regression equation for the plot is y= 7.60x10-2 (t) + 2.353. What is the concentration of ClO2 at 2.00 minutes if the initial concentration of ClO4 ia 0.425 M? a.) 0.0872 M b.) 2.50 M c.) 11.5 M d.) 0.399 MA group of students compiled the data shown in data table 1 below. What is the exact calculated order for IO3-(aq) to 2 decimal places? Exp. # [IO3-]0 (M) [I-]0 (M) [H+]0 (M) Time (s) 1 0.005 0.05 2 x 10-5 22.12 2 0.010 0.05 2 x 10-5 86.84 3 0.005 0.10 2 x 10-5 5.35 4 0.005 0.05 4 x 10-5 2.65 Question 4 options: 2.44 -2.49 1.81 2.09 -1.72 -2.13 -2.36 1.49 2.24 -1.97 -1.53 1.65Can I get detailed explanation for this table , mentioning the apparatus and when’s the timer is started ? What happens next
- Determine the average rate of change of BB from ?=0 st=0 s to ?=272 s.t=272 s. A⟶2BA⟶2B Time (s) Concentration of A (M) 0 0.7300.730 136136 0.4450.445 272272 0.1600.160 rateB= __________M/sUse the information given here to answer both questions (4 and 5). For the fictitious reaction: AB → A + B Some data was collected measuring the [AB] at certain time intervals. This data was then graphed as described below. A graph of 1/[AB] versus time (s) gives a straight line with the equation: y = 0.0052x + 0.1135 and a R2 = 0.9995. A graph of ln[AB] versus time (s) gives a straight line with the equation: y = -0.0068x + 1.725 and a R2 = 0.8925. Assume the data doesn't fit a zero order reaction. What is the rate law for this reaction? Select one: a. Rate = k[AB]2 b. Rate = k[A][B] c. Rate = k[AB] d. Rate = k Using the data from the previous question. What is the value of the rate constant for the given reaction? Select one: a. k = 0.0052 M-1s-1 b. k = 0.0157 Ms-1 c. k = 0.0068 s-1 d. There is no way to determine the value of the rate constant from the given information.Time (min) Concentration of Phenyl Acetate (M) 0 0.55 0.25 0.42 0.50 0.31 0.75 0.23 1.00 0.17 1.25 0.12 1.50 0.082 what is the difference between concentration vs. time, 1/concentration vs. time, and ln concentration vs. time in terms of graphing?

