Titanium and chlorine react to form titanium(IV) chloride, like this: Ti(s)+2 Cl₂(g) →TiCl4(!) At a certain temperature, a chemist finds that a 9.7 L reaction vessel containing a mixture of titanium, chlorine, and titanium(IV) chloride at equilibrium has the following composition: compound amount Ti 4.64 g Cl₂ 1.82 g TiC14 4.64 g Calculate the value of the equilibrium constant K for this reaction. Round your answer to 2 significant digits. K = 0 Explanation 630 999 Check X 11 Ś MacBook Air Ⓒ2022 McGraw Hill LLC. All Rights Reserved. tv Terms of Use A O Privacy Center | Acc
Titanium and chlorine react to form titanium(IV) chloride, like this: Ti(s)+2 Cl₂(g) →TiCl4(!) At a certain temperature, a chemist finds that a 9.7 L reaction vessel containing a mixture of titanium, chlorine, and titanium(IV) chloride at equilibrium has the following composition: compound amount Ti 4.64 g Cl₂ 1.82 g TiC14 4.64 g Calculate the value of the equilibrium constant K for this reaction. Round your answer to 2 significant digits. K = 0 Explanation 630 999 Check X 11 Ś MacBook Air Ⓒ2022 McGraw Hill LLC. All Rights Reserved. tv Terms of Use A O Privacy Center | Acc
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter12: Chemical Equilibrium
Section: Chapter Questions
Problem 41QRT: Nitrosyl chloride, NOC1, decomposes to NO and Cl2 at high temperatures.
2 NOCl(g) ⇌ 2 NO(g) +...
Related questions
Question
Help with the following question
Round the answer to 2 sig figs
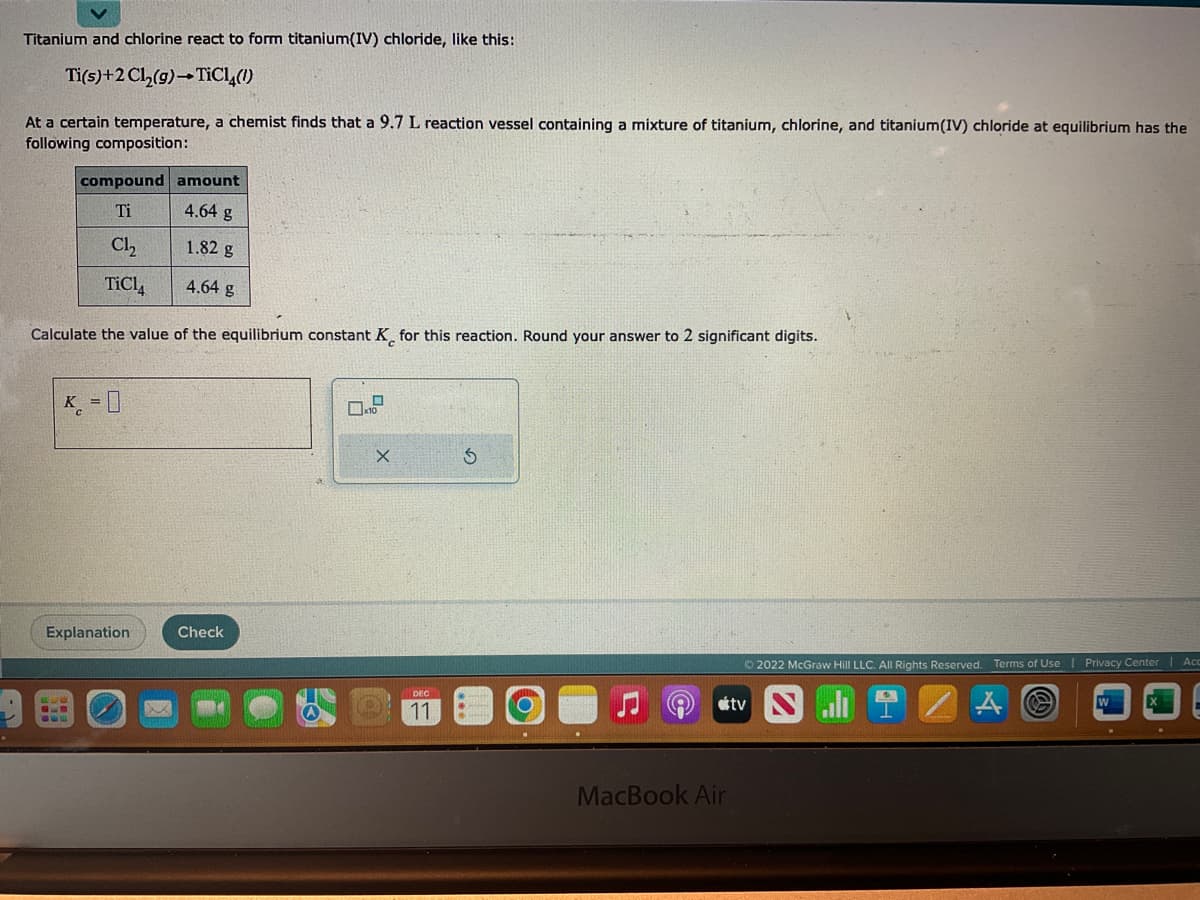
Transcribed Image Text:Titanium and chlorine react to form titanium(IV) chloride, like this:
Ti(s)+2 Cl₂(g) →TiCl4(!)
At a certain temperature, a chemist finds that a 9.7 L reaction vessel containing a mixture of titanium, chlorine, and titanium(IV) chloride at equilibrium has the
following composition:
compound amount
Ti
4.64 g
Cl₂
TiC14
Calculate the value of the equilibrium constant K for this reaction. Round your answer to 2 significant digits.
K = 0
Explanation
939
1.82 g
4.64 g
Check
X
11
Ś
MacBook Air
Ⓒ2022 McGraw Hill LLC. All Rights Reserved. Terms of Use
tv
Privacy Center | Acc
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning