To the solution at 100 °C described in #4, an additional 15.0 g of water is added. The solution is then cooled to 0 °C. You will need to refer Figure 1 as you answer the remaining questions. 260 240 220 200 180 KNO3 160 140 100 80 Cuso4-5H20 60 40 20 10 20 30 40 50 60 70 80 90 100 110 Temperature in °C Solubility in g solute / 100g H20
To the solution at 100 °C described in #4, an additional 15.0 g of water is added. The solution is then cooled to 0 °C. You will need to refer Figure 1 as you answer the remaining questions. 260 240 220 200 180 KNO3 160 140 100 80 Cuso4-5H20 60 40 20 10 20 30 40 50 60 70 80 90 100 110 Temperature in °C Solubility in g solute / 100g H20
Chapter1: Matter, Measurements, And Calculations
Section: Chapter Questions
Problem 1.126E
Related questions
Question
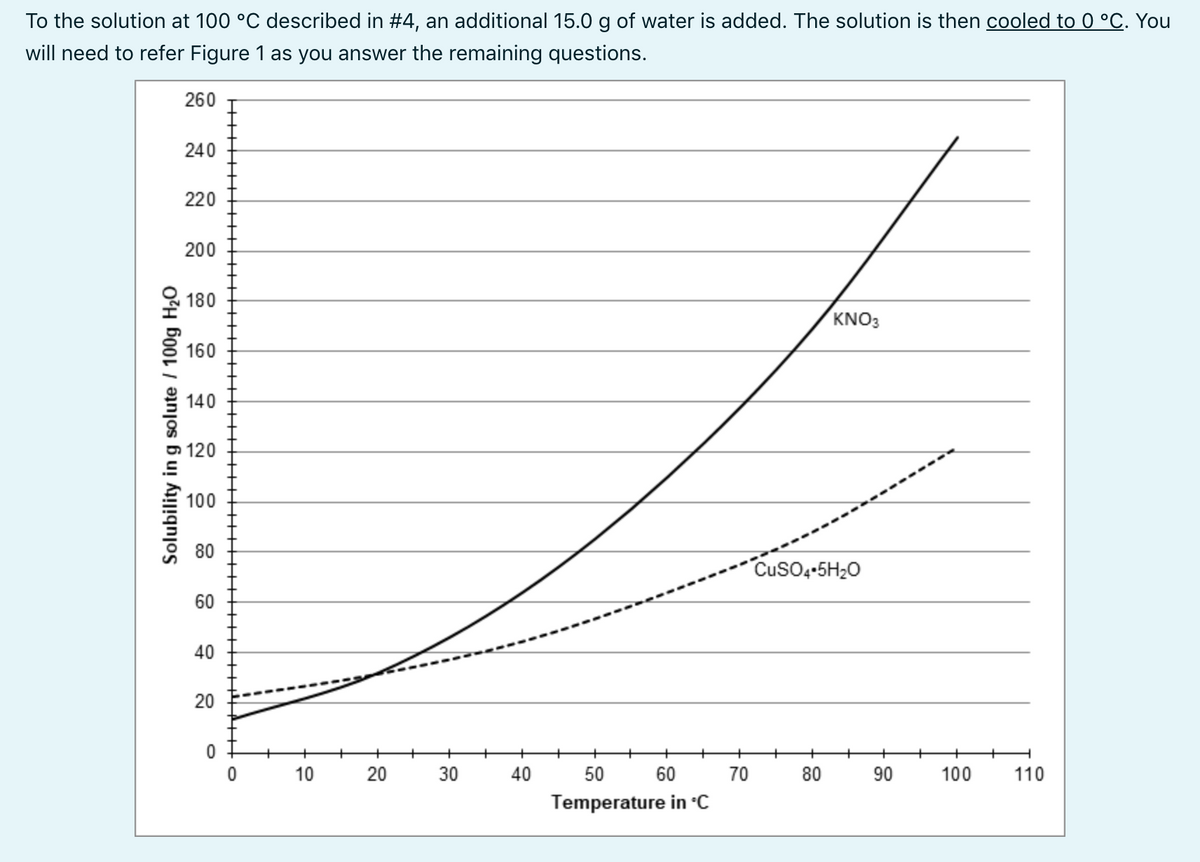
Transcribed Image Text:To the solution at 100 °C described in #4, an additional 15.0 g of water is added. The solution is then cooled to 0 °C. You
will need to refer Figure 1 as you answer the remaining questions.
260
240
220
200
180
KNO3
160
140
120
100
80
Cuso,-5H;0
60
40
20
+
+
+
+
10
20
30
40
50
60
70
80
90
100
110
Temperature in °C
Solubility in g solute / 100g H20
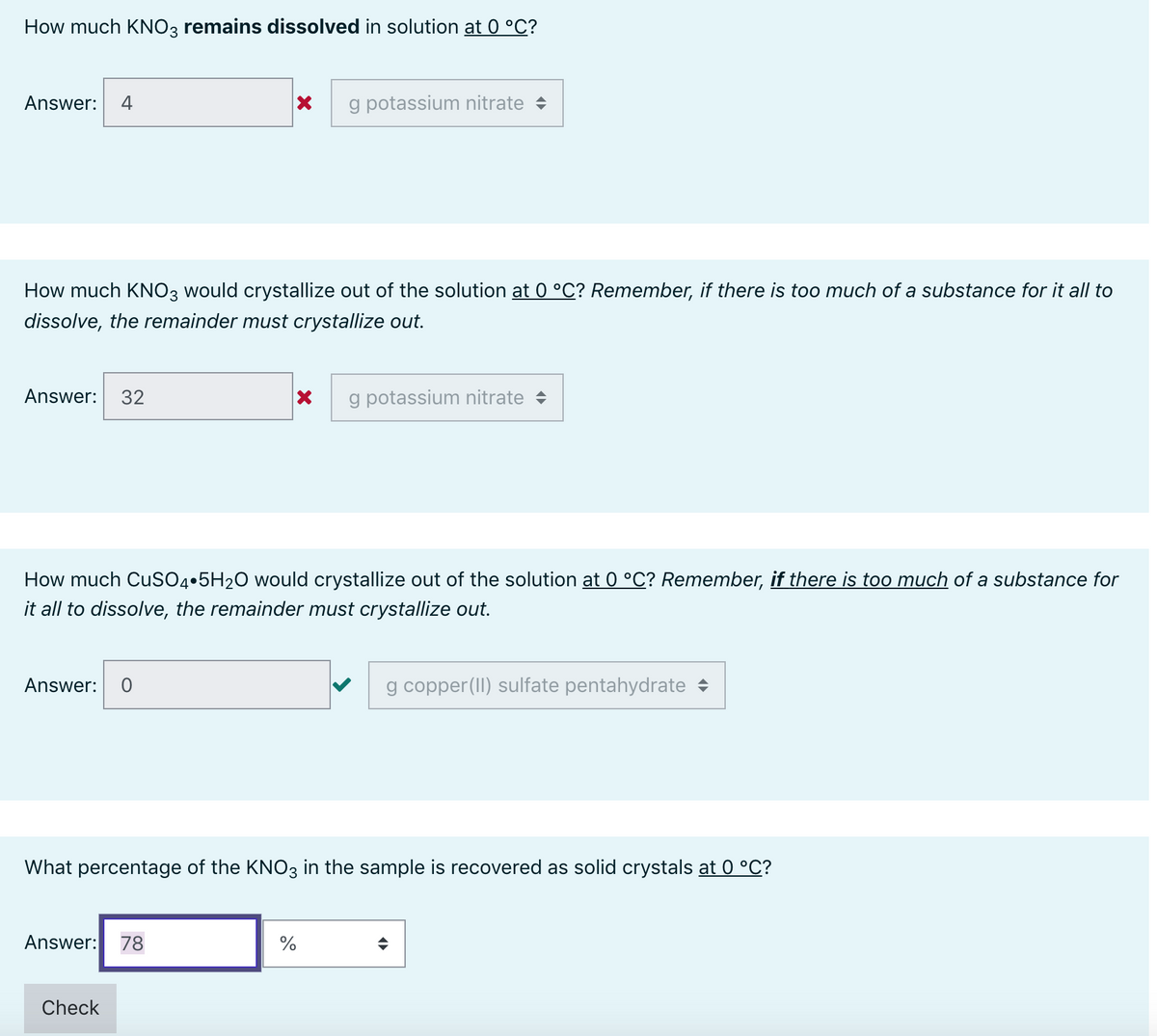
Transcribed Image Text:How much KNO3 remains dissolved in solution at 0 °C?
Answer:
4
g potassium nitrate +
How much KNO3 would crystallize out of the solution at 0 °C? Remember, if there is too much of a substance for it all to
dissolve, the remainder must crystallize out.
Answer: 32
g potassium nitrate +
How much CUSO4•5H2O would crystallize out of the solution at 0 °C? Remember, if there is too much of a substance for
it all to dissolve, the remainder must crystallize out.
Answer:
g copper(II) sulfate pentahydrate +
What percentage of the KNO3 in the sample is recovered as solid crystals at 0 °C?
Answer: 78
%
Check
Expert Solution
Step 1

Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning