Two non- reactive gases A and B are present in a container with partial pressures 2.44 X10 Pa and 2.68 x10 Pa respectively. When a third non-reactive gas C is added then total pressure becomes 1.01 ×10' Pa. Find the partial pressure of gas C? Volume and Temperature are constant Protal Pi P2 Gas C Gas A+ Gas B+ Gas C Gas A+ Gas B Select one: a. 1.01 x 10 Pa b. 5.12 x 104 Pa C. 4.98 x 104 Pa
Two non- reactive gases A and B are present in a container with partial pressures 2.44 X10 Pa and 2.68 x10 Pa respectively. When a third non-reactive gas C is added then total pressure becomes 1.01 ×10' Pa. Find the partial pressure of gas C? Volume and Temperature are constant Protal Pi P2 Gas C Gas A+ Gas B+ Gas C Gas A+ Gas B Select one: a. 1.01 x 10 Pa b. 5.12 x 104 Pa C. 4.98 x 104 Pa
Chapter2: The Kinetic Theory Of Gases
Section: Chapter Questions
Problem 92AP: Unreasonable results. (a) Find the temperature of 0.360 kg of water, modeled as an ideal gas, at a...
Related questions
Question
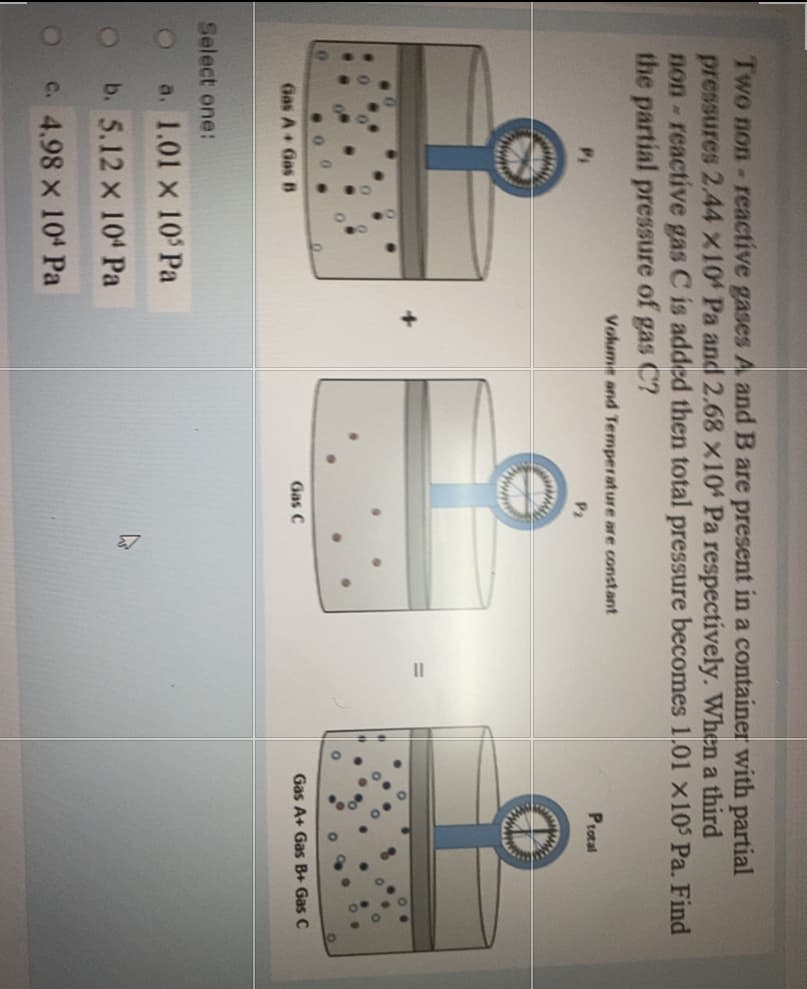
Transcribed Image Text:Two non- reactive gases A and B are present in a container with partial
pressures 2.44 x10 Pa and 2.68 X104 Pa respectively. When a third
non-reactive gas C is added then total pressure becomes 1.01 x10 Pa. Find
the partial pressure of gas C?
Volume and Temperature are constant
Protal
P2
www
Gas A+ Gas B+ Gas C
Gas C
Gas A+ Gas B
Select one:
a. 1.01 x 10 Pa
b. 5.12 x 104 Pa
OC. 4.98 X 104 Pa
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you


Physics for Scientists and Engineers, Technology …
Physics
ISBN:
9781305116399
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning


Physics for Scientists and Engineers, Technology …
Physics
ISBN:
9781305116399
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning