Two students are tasked with determining the milligrams of chloride in a simulated soil sample. Both students extract chloride into aqueous solution and perform triplicate titration with silver nitrate solution and dichlorofluorescein indicator. The students report the following results to their laboratory instructor. If the accepted value is 36.90 mg chloride/g sample, the laboratory instructor infers that student #2's work exhibits student #1. Student #1 Student #2 Trial (mg/g) (mg/g) 35.98 35.99 2 30.11 36.4 3. 32.88 36.29 Average 32.88 36.23 lower accuracy and lower precision than O higher accuracy and lower precision than O higher accuracy and higher precision than lower accuracy and higher precision than 1,
Two students are tasked with determining the milligrams of chloride in a simulated soil sample. Both students extract chloride into aqueous solution and perform triplicate titration with silver nitrate solution and dichlorofluorescein indicator. The students report the following results to their laboratory instructor. If the accepted value is 36.90 mg chloride/g sample, the laboratory instructor infers that student #2's work exhibits student #1. Student #1 Student #2 Trial (mg/g) (mg/g) 35.98 35.99 2 30.11 36.4 3. 32.88 36.29 Average 32.88 36.23 lower accuracy and lower precision than O higher accuracy and lower precision than O higher accuracy and higher precision than lower accuracy and higher precision than 1,
Chemistry: Matter and Change
1st Edition
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Chapter14: Mixtures And Solutions
Section14.2: Solution Concentration
Problem 23PP
Related questions
Question
100%
Q17 and q21
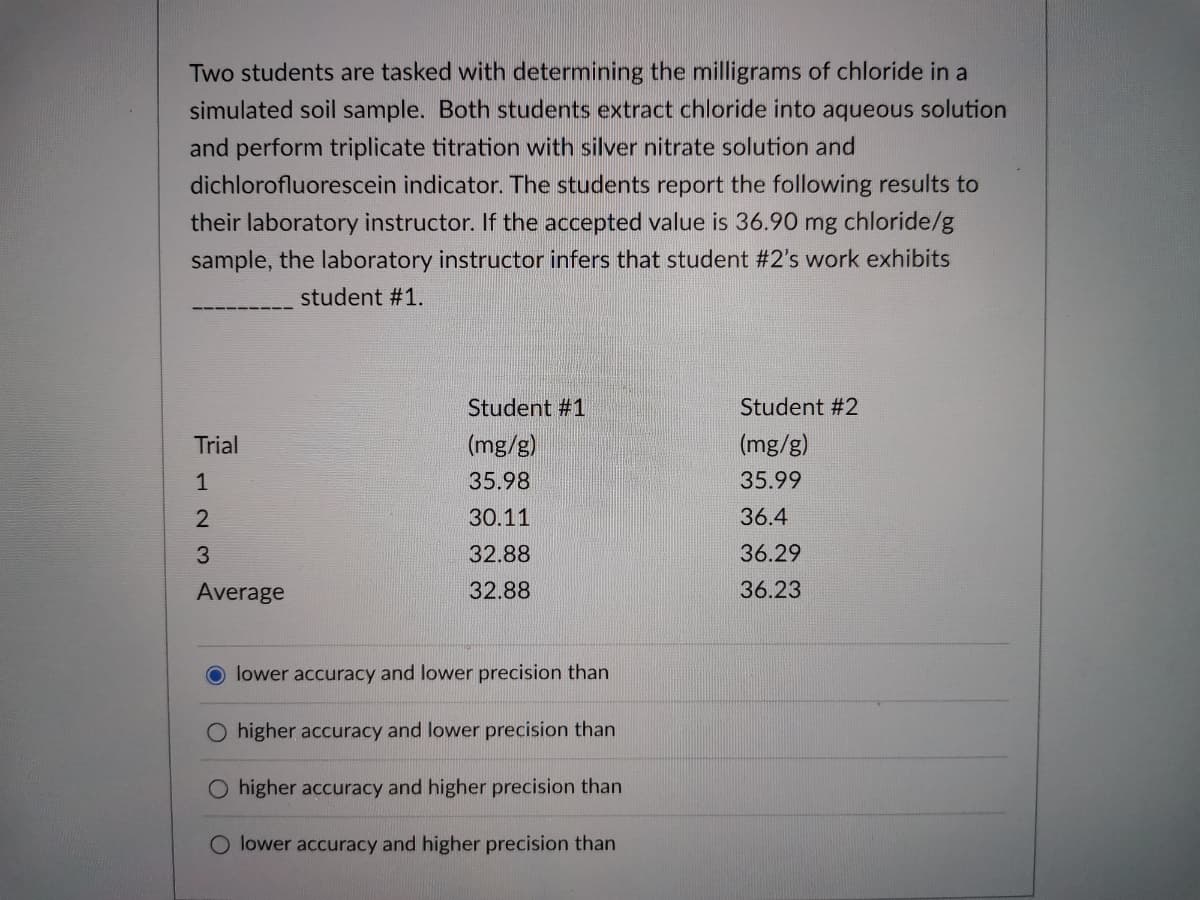
Transcribed Image Text:Two students are tasked with determining the milligrams of chloride in a
simulated soil sample. Both students extract chloride into aqueous solution
and perform triplicate titration with silver nitrate solution and
dichlorofluorescein indicator. The students report the following results to
their laboratory instructor. If the accepted value is 36.90
mg
chloride/g
sample, the laboratory instructor infers that student #2's work exhibits
student #1.
Student #1
Student #2
Trial
(mg/g)
(mg/g)
1
35.98
35.99
30.11
36.4
3.
32.88
36.29
Average
32.88
36.23
lower accuracy and lower precision than
higher accuracy and lower precision than
O higher accuracy and higher precision than
lower accuracy and higher precision than
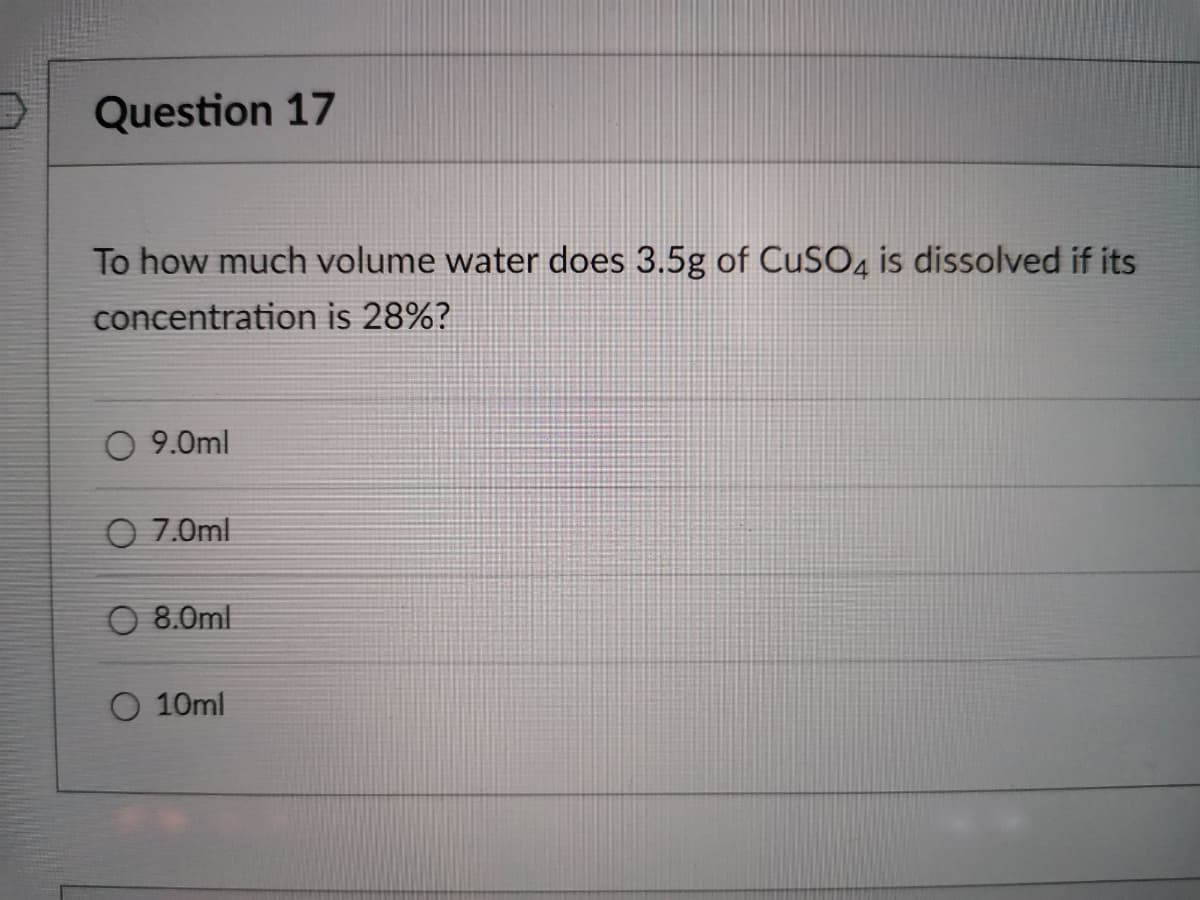
Transcribed Image Text:Question 17
To how much volume water does 3.5g of CUSO, is dissolved if its
concentration is 28%?
O 9.0ml
O 7.0ml
8.0ml
O 10ml
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning