Q: Carbon monoxide and the cyanide ion are both toxic because they bind more strongly than Oxygen to…
A: a. The given equations are: Hb + O2 ↔ HbO2 K = 2.0×1012 ......(1)Hb + CO ↔ HbCO K =…
Q: Subsequently, derive the p-bonding ligand group orbitals and draw a second qualitative molecular…
A: P- Bonding in ligand group orbital- we will discuss p-bonding in ligand by taking some eexamples-…
Q: The formations of [cis-PtCh(NH3)2] and [trans-PtCl½(NH3)2] are different although same ligands are…
A: The formation of above complex cab be explain by trans effect . the trans effect is the effect that…
Q: Does MnO4- have Ligand to Metal Charge Transfer or Metal to Ligand Charge Transfer?
A:
Q: The
A:
Q: ccording to the graph below, The ratio for One Metal is to how many LIGAND? ( Write a whole number)
A: In the given spectrometric titration graph: On fitting the graph into linear regression,…
Q: 1. "The bonding of a hydrogen atom to a metal atom is a o interaction, whereas the bonding of a…
A:
Q: Which option indicates, in order, whether the following compounds obey the effective atomic number…
A: EAN for K2[HgI4] is determined as shown below. Therefore, K2[HgI4] obeys the effective atomic number…
Q: e ligand?
A:
Q: Consider the compound (C8H8)Ru(CO)3 which is likely to be the best description of the C8H8 ligand ?
A: The stability of any organometallic compound is given by Effective Atomic Number (EAN Rule).…
Q: Define the term pyrometallurgy?
A: Pyro means “heat or temperature” and metallurgy is the branch of science which concerned with the…
Q: Give evidence that [Co(NH3)5Cl]SO4 and [Co(NH3)5SO4]Cl are ionization isomers. (ii). Calculate…
A: Isomers are those compounds which have same chemical formula but they are different from each other.…
Q: Which of the following statements is(are) correct? (1) K; is a measure of the interaction strength…
A: The correct statement is as follows, Kf s the measure of the interaction strength of a ligand…
Q: 3-They have load transfer nodes to the metal. For which of these complexes do you expect the band…
A: LMCT - it stands for ligand to metal charge transfer. During LMCT the non-bonding electrons of…
Q: Use the thermodynamic data below to compute the average bond enthalpy of the metal-ligand bonds in…
A:
Q: Define racemic mixture? And briefly explain the Racemic modification and its types?
A: A racemic mixture is a mixture of equal quantities of two enantiomers or substances that have…
Q: Use data from CRC_Std_Thermodyn_Substances and CRC_Std_Thermodyn_Aqueous-Ions to calculate ΔrH∘ΔrH∘…
A: Given reaction is : 2CaC2O4 (s) ⟶ 4CO (g) + O2 (g) + 2CaO (s) , ΔrH∘ = ? Calculate the values of…
Q: Which of the following ligands could form chelates? Indicate also the donor atom for each ligand. 1.…
A: As per our guidelines we can solve only first question. Please re-post other questions. Chelating…
Q: Construct a mode | of [Cr (C2 O4 Make sure are linked to donor atoms to indicate how the atorms…
A: The IUPAC name of this compound is Tris Oxalato chromium. It is a coordination compound. there is a…
Q: Which d orbital on M is capable of having a bonding interaction with the ligand group orbital shown?
A: Ligands are the chemical species which have the ability to donate electron pairs. Metals accept…
Q: explain anharmonicity.
A: Anharmonicity:
Q: (d) Based on crystal field stabilisation energy considerations, are FeCr204 and CuCr204, likely to…
A: The correct answer is given below
Q: Pyridine (CsHgN), abbreviated py, is the molecule N: (a) Would you expect pyridine to act as a…
A: Ligands Ligands in the coordinated complex is the ion or neutral molecule that act as the lewis base…
Q: Co ligand insertion oc | CH, to co co co n-CHOCH, oc čo oc OCH, OCH, čo CHOCH, o0 Co B D E O A O B…
A:
Q: a) Do you expect a distortion for [Ti(H2O),]³* and [CoCl]³- b) What type of distortion would be…
A: Ans
Q: In the case of ligand substitution at square planar platinum(II), which is the stronger trans effect…
A: Trans effect is a kinetic effect, that is in square planar complex how a ligand trans to the leaving…
Q: During the dissociation process, ligand is dissociated from the [RHCI(OX)(en)(PPH3)] octahedral…
A: In a dissociation mechanism, a ligand is removed from the complex.
Q: Explain The Relationship between ?G°rxn and K?
A: ΔG° is defined as the change in standard Gibbs free energy. K is the equilibrium constant and is…
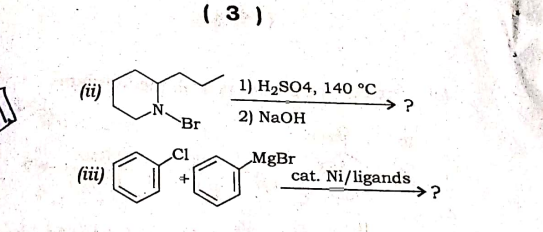
Q: (üi) 1) H2SO4, 140 °C →? Br 2) NaOH MgBr (iüi) cat. Ni/ligands →?
A:
Q: (a) Using Werner’s definition of valence, which property isthe same as oxidation number, primary…
A: The coordination complexes can be defined as the complex compounds which have coordinate linkage…
Q: What is the charge on each of the following complex ions? hexaaquairon(II), [Fe(H2O)6]?…
A: 1.) Charge on hexaaquairon(II) Is +2 The oxidation state or charge on the central metal ion, Fe =…
Q: 1- The gap between the higher and lower energy levels is called (CFSE). 2- Splitting of d orbitals…
A: As per the rules, only the first three sub-parts of a question can be answered: Co-ordination…
Q: Visualizing Conplex. Tons construct [cr(H2O),], [cr CH2O)s S]* , [CrC HzO)yC1]*, and [Cocend2 C H2…
A: The compounds in which ligands are bonded to central metal atom, usually a transition metal, are…
Q: 1-Explain the bonding nature of [Fe(CN)5 (NO)]-2 linear NO ligand. Vour anewer
A: Applying concept of 18e- rule.
Q: The complex trans-[Co(en)2Cl2]+ undergoes substitution of Cl- by H2O slower than the complex…
A: To explain: The complex trans-[Co(en)2Cl2]+ undergoes substitution of Cl- by H2O slower than the…
Q: In 4 to 5 sentences summarize which type of ligand would more easily release a trapped central metal…
A: Those ligands have higest trans effect, would more easily release a trapped central metal cation. CO…
Q: Carbon monoxide is toxic because it binds more strongly to the iron in hemoglobin (Hb) than does O,…
A: HbO2 →Hb + O2 ∆G∘=70KJHb + CO → HbCO ∆G∘ =-80KJOn adding both the reaction we getHbO2 + CO…
Q: uickly plezz!! The levigating agent role in Suppositories manufacturing ??
A: Suppositories are solid medications. It enter the body through the rectum, vagina, or urethra.…
Q: Formation constant and Gibbs free energy For the complex formation reaction where Mis any metal and…
A:
Q: Which d orbital on M (center metal) is capable of having a bonding interaction with the ligand group…
A: dx2-y2 is capable of forming bonding interaction with the ligands because ligands approach or…
Q: Name the following complex ions. formula [Fe(H₂O)]* [Cu(NH,), + [T₁(H₂O)]* name X ?
A: The given metal complexes are exclusively cationic in nature with variable positive charges.
Q: 20. If 8.4g of salicylaldehyde (sal), reacts with 1.75g of ethylenediamine (en), to produce 5.75g of…
A: Two moles of salicylaldehyde react with one mole of ethylenediamine to form salen ligand as the…
Q: In the reaction between aqueous.cobalt(Il) chloride with ammonia, the only complex to be formed is…
A: Since you have asked a multiple sub-part question, we will solve first three sub-parts for you. For…
Q: The Lewis structure :C≡O: indicates that the CO molecule has two lone pairs of electrons. When CO…
A: The binding of CO molecule to the transition metal forms coordination compounds. CO molecule has two…
Q: Which species are more likely to act as ligands? Neutral molecules that are polar or those that are…
A: To find: The species which acts as ligand
Q: واجب بيتي: اي عضوية فلزية؟ Which of the following are organometallic co Cr(CD), , PdHMe,Br WMe,,…
A: Substance which has minimum one metal carbon bond , is called organometallic compound. Thus to…
Q: From the Job's plot shown, what is ratio of the metal-to-ligand (M:LMT) ?
A: We are provided with a Job's plot and we need to determine the ratio of metal-to-ligand.
Step by step
Solved in 3 steps with 3 images

- Give an example of a double salt that can be formed via ligand exchange and give the reactions involvedMolecular nitrogen (N2) can act as a ligand in certaincoordination complexes. Predict the structure of [V(N2)6],which is isolated by condensing V with N2 at 25 K. Is thiscompound diamagnetic or paramagnetic? What is theformula of the carbonyl compound of vanadium that hasthe same number of electrons?Is the interaction between an ammonia ligand and a metal cation a Lewis acid–baseinteraction? If so, which species acts as the Lewis acid?
- Why is the pH of the medium important for the formation of chelated complexes between metal ions and ligands? Choose the best answer: Select one: a. Because the ligands give rise to acid-base reactions that influence the stability of the chelate b. Because if the pH is not very acidic, the ligands do not protonate and the complex cannot form c. Because the pH of the medium is always important for any type of reaction. d. Because in the absence of pH there is no reaction of any kindFill in the blanks : (i) When a ligand has a two-complexing or donor group in its structure, it is said to be————— (ii) ___________are compounds whose colour changes when they bind to a metal ion. (ii) If you double the wavelength, you will observe. ________the energy. (iv) In_____________ titration, we monitor changes in absorbance during the titration to find out the equivalence point. (v) Two liquids are_____________ if they form a single phase when they are mixed in any ratio. (vi) The efficiency of Chromatographic Columns can be described by____________Equation.You prepare a solution of protein and its ligand where the initial concentrations are [Protein] = 10 nM and [Ligand] = 10 nM. At equilibrium you measure the [PL] = 5 nM. If the protein - ligand association can be represented by: P + L <--> PL What is the dissociation constant (Kd) of the protein for the ligand under the solvent conditions you have chosen? a. 1 nM b. 0.2 nM-1 c. 5 nM d. 10 nM
- 19. The association constant of protein X for ligand Y is: 100 micromolar. At what concentration of ligand is half fo the protein bound?Explain why the complex [Fe(H2O)6]3+ has Do = 14,000 cm-1, while [Fe(H2O)6]2+ has Do = 9,350 cm-1.For the aqueous [Ni(NH3 )6]2+ complex Kf=5.50 x 108 at 25C . Suppose equal volumes of 0.0024 M Ni(NO3 )2solution 0.94M NH3solution are mixed. Calculate the equilibrium molarity of aqueous Ni2+ ion. Round your answer to 2 significant digits.
- Will methylamine (CH3NH2) be a monodentate or abidentate ligand? With which of its atoms will it bind to ametal ion?For the aqueous [Ag(CN)2]^- complex Kf =3.0x10^20 at 25°C.Suppose equal volumes of 0.0068M AgNO3 solution and 0.32M KCN solution are mixed. Calculate the equilibrium molarity of aqueous Ag^+ ion.Round your answer to 2 significant digits.(a) [Cr(II)(H2O)6]2+ and [Cr(III)(H2O)6]3+ display markedly different ligand exchange rates. Which species is likely to be more labile and what are the reasons for this? (b) The hexaquachromium(III) ion is violet, but addition of excess cyanide produces the yellow hexacyanidochromate(III) ion. Explain the change in colour between the two complexes.

