Understanding connections between descriptions of wea... When a 0.033M aqueous solution of a certain acid is prepared, the acid is 4.0% dissociated. Calculate the pH of the solution. Round your answer to 2 decimal places. pH = [| dlo
Understanding connections between descriptions of wea... When a 0.033M aqueous solution of a certain acid is prepared, the acid is 4.0% dissociated. Calculate the pH of the solution. Round your answer to 2 decimal places. pH = [| dlo
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter15: Acid–base Equilibria
Section: Chapter Questions
Problem 107AP
Related questions
Question
100%
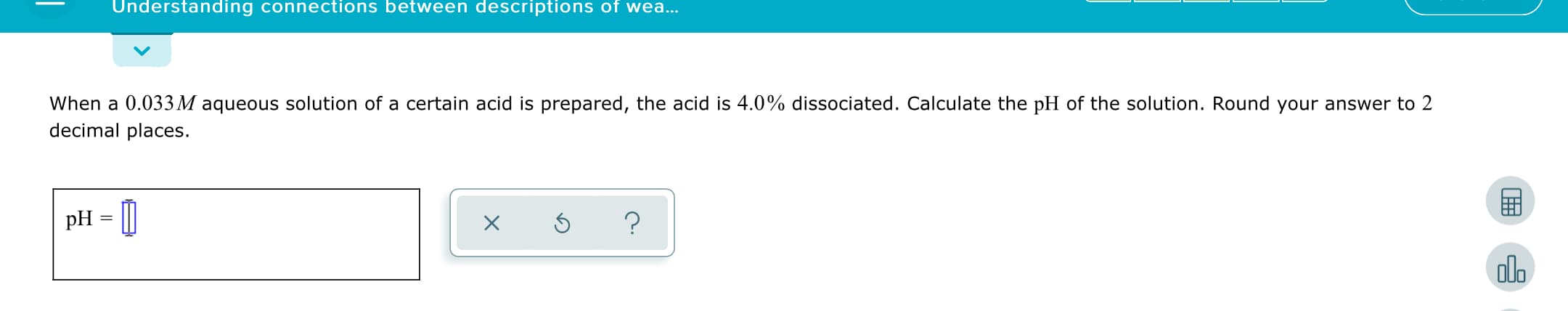
Transcribed Image Text:Understanding connections between descriptions of wea...
When a 0.033M aqueous solution of a certain acid is prepared, the acid is 4.0% dissociated. Calculate the pH of the solution. Round your answer to 2
decimal places.
pH = [|
dlo
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 2 images

Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning
