unisauaPIAL :ua ar Number of Neutrons Number of Abundance Abundance Name of isotope Mass (amu) (%) (decimal) Protons Magnesium-24 23.985 78.99% Magnesium-25 24.986 10.00% Magnesium-26 25.983 11.01% Isotope Mass Abundance
unisauaPIAL :ua ar Number of Neutrons Number of Abundance Abundance Name of isotope Mass (amu) (%) (decimal) Protons Magnesium-24 23.985 78.99% Magnesium-25 24.986 10.00% Magnesium-26 25.983 11.01% Isotope Mass Abundance
Chapter1: Excel Basics
Section: Chapter Questions
Problem 4P
Related questions
Question
Transcribed Image Text:*BIUA:
Heading 1
Russo One
30
CHD
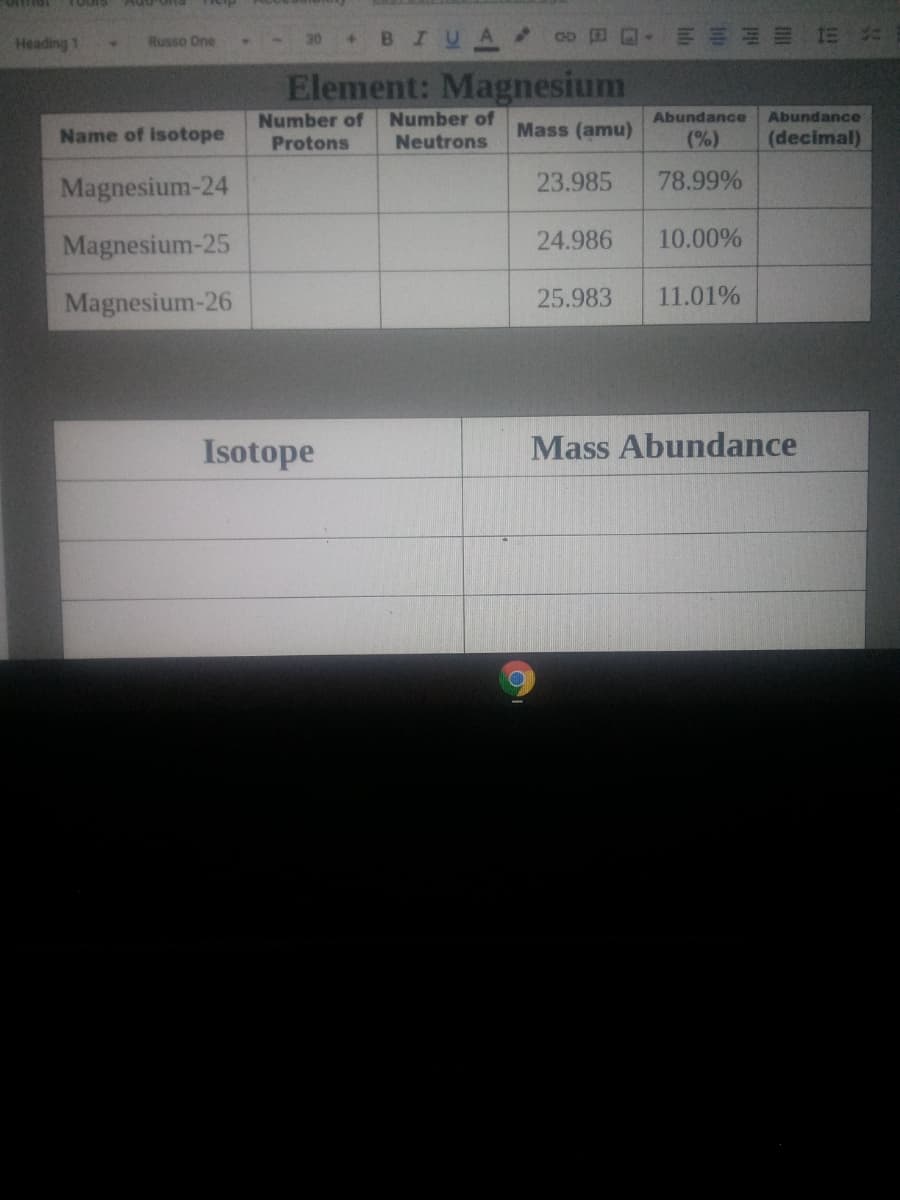
Element: Magnesium
Number of
Number of
Abundance
Abundance
Name of isotope
Mass (amu)
(%)
(decimal)
Protons
Neutrons
Magnesium-24
23.985
78.99%
Magnesium-25
24.986
10.00%
Magnesium-26
25.983
11.01%
Isotope
Mass Abundance
Transcribed Image Text:Humber of Humber of
Protons
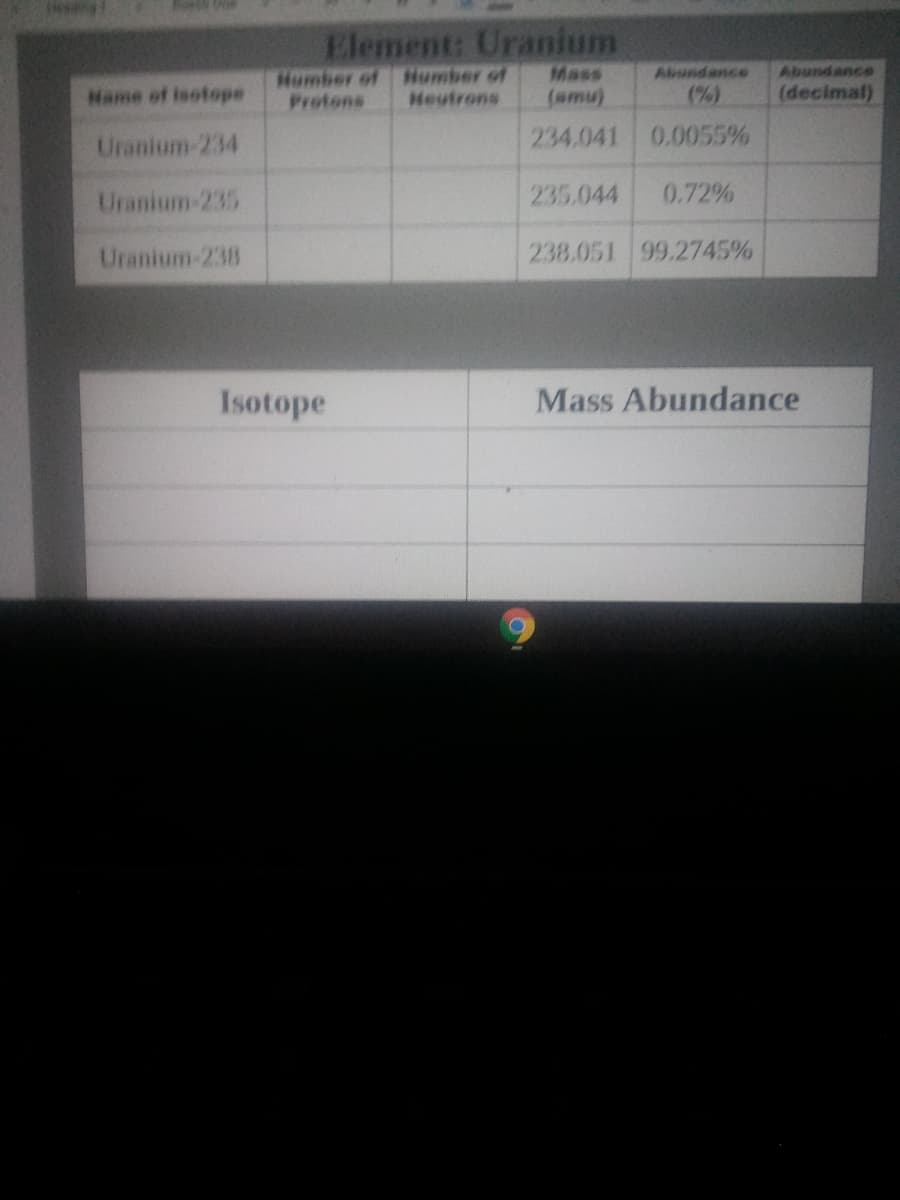
Element: Uranium
Mass
(amu)
Abundance
Abundance
Hame of isotope
Heutrons
(%)
(decimal)
Uranium-234
234.041 0.0055%
Uranium-235
235.044
0.72%
Uranium-238
238.051 99.2745%
Isotope
Mass Abundance
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning
