Use the following information to answer the next four questions. Hydroiodic acid often is used to disinfect and sanitize medical tools and products. In an experiment, 5.0 ml of 0.75 mol/L HI(aq) is mixed with 20.0 mL of distilled water. What is the hydronium ion concentration of the resulting diluted solution? (Do not show your work in the space provided; record only your final answer with the correct number of significant digits and the proper units.) Answer: What is the pH of the resulting solution? (Do not show your work in the space provided; record only your final answer with the correct number of significant digits.) Answer: If 1.0 mL of the diluted solution is added to 9.0 mL of distilled water, what is the resulting pH of the solution? (Do not show your work in the space provided; record only your final answer with the correct number of significant digits.)
Use the following information to answer the next four questions. Hydroiodic acid often is used to disinfect and sanitize medical tools and products. In an experiment, 5.0 ml of 0.75 mol/L HI(aq) is mixed with 20.0 mL of distilled water. What is the hydronium ion concentration of the resulting diluted solution? (Do not show your work in the space provided; record only your final answer with the correct number of significant digits and the proper units.) Answer: What is the pH of the resulting solution? (Do not show your work in the space provided; record only your final answer with the correct number of significant digits.) Answer: If 1.0 mL of the diluted solution is added to 9.0 mL of distilled water, what is the resulting pH of the solution? (Do not show your work in the space provided; record only your final answer with the correct number of significant digits.)
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter4: Types Of Chemical Reactions And Solution Stoichiometry
Section: Chapter Questions
Problem 7ALQ: Consider separate aqueous solutions of HCl and H2SO4 with the same molar concentrations. You wish to...
Related questions
Question
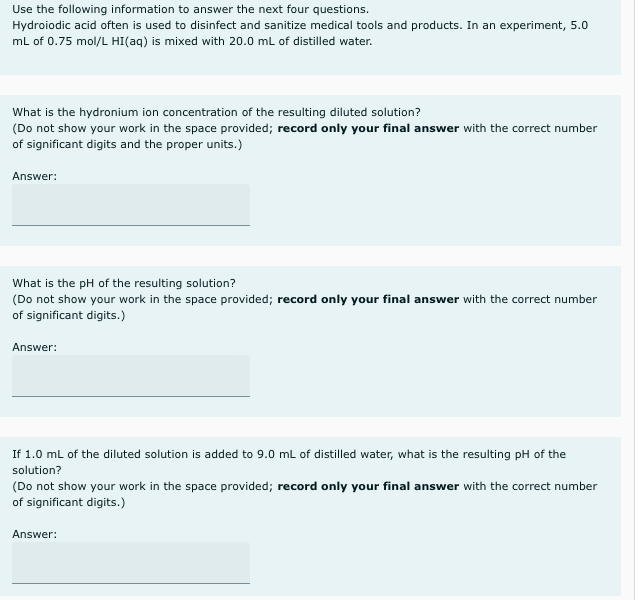
Transcribed Image Text:Use the following information to answer the next four questions.
Hydroiodic acid often is used to disinfect and sanitize medical tools and products. In an experiment, 5.0
mL of 0.75 mol/L HI(aq) is mixed with 20.0 mL of distilled water.
What is the hydronium ion concentration of the resulting diluted solution?
(Do not show your work in the space provided; record only your final answer with the correct number
of significant digits and the proper units.)
Answer:
What is the pH of the resulting solution?
(Do not show your work in the space provided; record only your final answer with the correct number
of significant digits.)
Answer:
If 1.0 ml of the diluted solution is added to 9.0 mL of distilled water, what is the resulting pH of the
solution?
(Do not show your work in the space provided; record only your final answer with the correct number
of significant digits.)
Answer:
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning