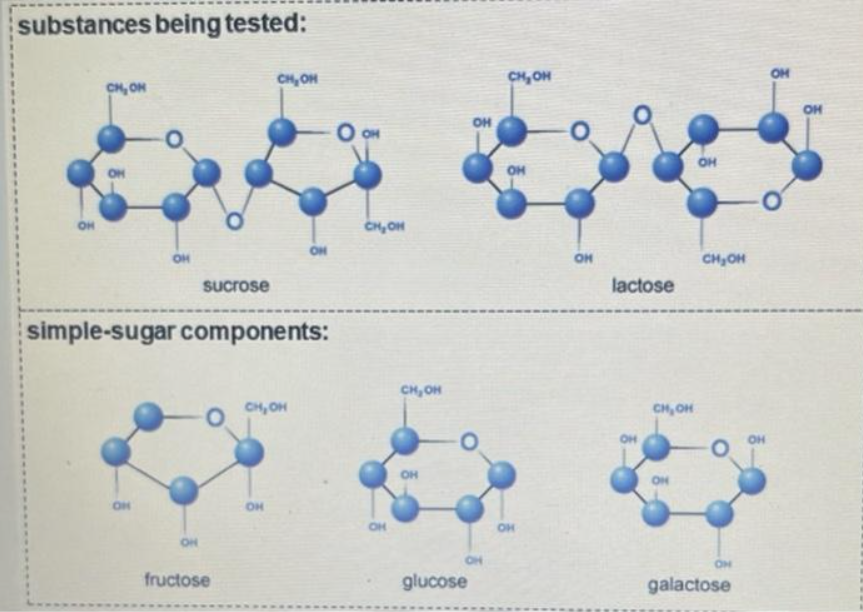
Use the molecular structures to predict which of the six solutions will test positive for glucose. Write your prediction in the answer space. Provide the reasoning behind your prediction
Q: Choose the combination of answers that most accurately completes the statement. The nitrogen bases…
A: The thread-like structure that consists of the genetic information of the organism, located within…
Q: Using the table attached, answer the following What happens when enzymes are not in their optimal pH…
A: Enzymes are 3D tertiary globular proteins that function as reusable biological catalysts. Enzymes…
Q: Consider a solution of glucose in water that is 5.25% by mass glucose. Calculate the indicated…
A: Water contains 5.25% by mass glucose. Mass of the solution=225g Therefore, Concentration of…
Q: The label on a snack food package says that one serving of the snack food contains 0 grams of trans…
A: Food is the source of energy. It contains several macronutrients such as carbohydrates, fats,…
Q: If protein synthesis was thought of as an analogy of making a pizza, the whole cookbook would be be…
A: Replication is the formation of identical copies of DNA. It occurs in nucleus of the cell.…
Q: Select the answer below that best describes the following statement: Steroids are lipids. A. True…
A: Lipids consist of a large group of molecules made up of hydrocarbons. They are insoluble or poorly…
Q: Which statement below is false about the probability of the absorption of a weak base from the…
A: Acid is a compound that donates proton and base is a compound that accepts proton. The strength of…
Q: Which elements are proteins composed of? Explain/elaborate. Describe the process of digestion and…
A: Ans no ( i ) Proteins and its components -- Food is made up of biological macronutrients Proteins ,…
Q: Discuss the results and observations in a regular laboratory experiments the denaturation of: 1.…
A: A polypeptide is fully functional if its properly folded misfolded protein got ubiquitination so, if…
Q: Briefly describe another technique you can use to study the interaction of Vitamin D3 with lipid…
A: Vitamin D3 with lipid membrane interaction.. Vitamin d3 is very important for our body as it…
Q: Directions: Write the letter of the correct answer to complete the paragraph. Use a separate sheet…
A: DNA is the genetic material in living organisms that is present within the nucleus, chloroplast and…
Q: fresh cucumber placed in a brine (salt) solution shrinks after a few days in the solution.
A:
Q: Choose the best answer for following question. Fructose and galactose are both isomers ofa.…
A: The correct option is (b) Glucose
Q: Refer to the following table below to answer the succeeding questions: Protein Isoelectric pH…
A: Two dimensional gel electrophoresis is an electrophoretic technique in which in the first dimension…
Q: Which unknown solution contains the highest concentration of sucrose? Three lab groups carried out…
A: Osmosis is defined as the movement of water molecules across the semipermeable membrane from the…
Q: a) mentioned the name of simple laboratory method to roughly estimate the concentration of an…
A: Proteins have wide variety of essential function in our body like providing energy, structural…
Q: Of the four types of biomolecules, which is the most important? Support your argument.
A: Biomolecules are naturally occurring molecules in living organisms. These are building blocks of…
Q: glycoproteins call
A: blood group antigens
Q: classical method to check the quality of Nucleic Acid Product
A: It is most commonly used today to perform a quick assessment of the purity of nucleic acid samples.…
Q: Graph the standard curve for the starch based on this data
A: In this question, we are given concentration of starch in g/L along with the absorbance. We have to…
Q: Nucleotides have roles in addition to being components of DNA and RNA. Give an example.
A: Nucleotide consists of three components: a five-carbon sugar, a nitrogenous base, as well as…
Q: TRUE OR FALSE Palmitic oil is (soluble) to hot ethanol and chloroform.
A: Hi. Thank you for the question. The second question looks incomplete. Please provide the details…
Q: Which of the following molecules would have been necessary to achieve what is occuring in
A: Answer: DNA is the deoxynucleotide acid which acts as the genetic material in many individuals , it…
Q: True or False: The output of the Mass Spectrometer gives us the exact mass of a molecule.
A: The output of the mass spectrometer is the separation of molecules based on their m/z (mass to…
Q: Two cells of an organism usually differ from each other because of their proteins
A: The cell types in a multicellular creature become not the same as each other on the grounds that…
Q: Why are buffers important in a biological system such as your body? Give reasons in your own words…
A: A biological system is a complicated network that connects a variety of physiologically significant…
Q: What is amino acids in Biochemistry. Answer in 3 paragraphs with references.
A: Biochemistry is a field of study of chemical processes among various biomolecules in living…
Q: Base on the data below, what type of lipid is being present in the sample?
A: A lipid is any of various organic compounds that are insoluble in water.. Some lipids are -- include…
Q: write a summary of how the amino acid substitution would affect the protein structure.
A: Proteins are the ultimate products of the genes. DNA is transcribed into m RNA and this is…
Q: Structural formulas for glucose and fructose. What difference do you notice?
A: Difference between glucose and fructose
Q: Give reason why potato cubes when placed in water become firm and increase in size.
A: Osmosis is a process through which the movement of water takes place through a semi-permeable…
Q: Suppose you have 150 mL of a 2.4 M glucose solution. How many moles of glucose do you have
A: Molarity is a term to define the concentration of a solution.
Q: Some students tested two samples of a mixture of starch and water with two different indicators. The…
A: Starch is a polysaccharide made up of repeated units of glucose monomer. It is tasteless and is…
Q: Answer SIMPLE or CONJUGATED ENZYME. a. An enzyme that contains a carbohydrate portion b. An enzyme…
A: The enzymes which are made from proteins only are known as simple enzymes, for example - pepsin,…
Q: `N*
A: The plasma membrane is a lipid bilayer structure that encloses the content of the cell of the…
Q: Select the BEST answer. Increasing the concentration of Fructose-2,6 bisphosphate would
A: Fructose-2,6 bisphosphate: It is synthesized of through a bifunctional enzyme containing both PFK-2…
Q: Four of the five answers are lipids. Select the exception.
A: Lipids can be defined as organic substance that are moderately insoluble in water but soluble in…
Q: Read the paragraph below and answer the question: What makes the information on blood type…
A: Blood typing is the Differentiation of blood groups based on the presence of type of antigens on…
Q: Use the following to answer the questions below: In each of the following multiple-choice questions,…
A: Lipids are insoluble molecules made up of carbon, hydrogen, and oxygen just as carbohydrates but in…
Q: Match the term and its description. Each term can only be used once.
A: Hormones are chemical messengers that are released directly into the bloodstream, where they are…
Q: The set of proteins produced by a cell is called its _____________.
A: Molecular biology deals with the omic sciences that include genomics, proteomics, and metabolomics.…
Q: Write a conclusion about Qualitative Analysis of Amino Acids and Proteins You can use this as…
A: Amino acids are the building blocks of the protein. Several quantitative and qualitative tests are…
Q: Define and simplify each of the words meaning in your own words. amino acid transporter:…
A: Introduction: Amino acids are molecules that contain an amine group, a carboxylic acid group, and a…
Q: Please draw a sketch that shows the location of each type of molecule (glucose, starch, iodine) at…
A: All are carbohydrate, we can specify the presence by different experiments.
Q: Create a concept map employing the different chemical tests for the differentiation of the types of…
A: Carbohydrates are polyhydroxy alcohols which are derivatives of aldehydes and ketones which are…
Q: Give one example of a molecule that is made from cholesterol
A: Lipids are macromolecules that are made up of carbon, hydrogen, and oxygen just like carbohydrates…
Q: Study the following data and its description. Write a conclusion of what you observe for each of…
A: Immunoprecipitation or IP is a technology that precipitates a protein from the solution with the…
Q: When would a medical professional such as a doctor or nurse want to know the concentration of a…
A: By knowing the concentration of a protein in solution the nurse and other health care professional…
Q: Explain the importance of isoelectric point in proteins in the given data below
A: pH is the scale which measures the acidity and basic nature of an aqueous solution. pH in chemistry…
Use the molecular structures to predict which of the six solutions will test positive for glucose. Write your prediction in the answer space. Provide the reasoning behind your prediction
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Given ß- Cyclodextrin Briefly explain its expected reaction (based on their structural formula) to the different qualitative tests for carbohydrates which are: (a) Molisch’s test (b)Fehling’s test (c) Bial’s testBased on this video https://www.youtube.com/watch?v=rKng5-ij6kQ Provide a schematic diagram for the Benedict ’s test methodologies in determining the presence of carbohydrates. Also, give the basic principle for the test. (this is not a graded question)Given Sorbitol, Briefly explain its expected reaction (based on their structural formula) to the different qualitative tests for carbohydrates which are: (a) Molisch’s test (b)Fehling’s test (c) Bial’s test
- Describe the evidence of Picric acid test to show a positive result for the presence of a specific sugar in a material or substance.Table of caffeine standards concentration Sample Conc, ppm Std1 16 Std2 32 Std3 48 Std4 64 Std5 80 If the volume used to make 100 mL of std 1 is 2 uL what is the concentration in M used to make a standard calibration curve? The standards are going to be used to build calibration curve to analyze caffeine in an energy drink. If 500 mL of the energy drink has target of 400 mg caffeine, how will you prepare the sample if you need 10 mL for the analysis ? . Caffeine MM=194.19 g/mol.Based on this video https://www.youtube.com/watch?v=rKng5-ij6kQ Provide a schematic diagram for the Fehling’s test methodologies in determining the presence of carbohydrates. Also, give the basic principle for the test. (not a graded question)
- Based on this video https://www.youtube.com/watch?v=rKng5-ij6kQ Provide a schematic diagram for the Fehling’s test methodologies in determining the presence of carbohydrates. Also, give the basic principle for the test. (not a graded question, video is very brief)The following are the sugars that formed brick red precipitate during Barfoed's test, except:* A. GlucoseB. Fructose C. Lactose D. Galactose E. None of the given optionsDescribe the evidence of Bial's test to show a positive result for the presence of a specific sugar in a material or substance.
- Can you please briefly describe the reaction mechanism that permits the detection of reducing sugars especially with the fehling's test ( please provide all the mechanisms involved in this test ) and also on how to interpret the results from this test Want solution ASAP in detailhttps://www.youtube.com/watch?v=rKng5-ij6kQ Provide a schematic diagram for the Seliwanoff’s test methodologies in determining the presence of carbohydrates. Also, give the basic principle for the test. (not a graded question, video is very brief)Based on this video https://www.youtube.com/watch?v=rKng5-ij6kQ Provide a schematic diagram for the Benedict's test methodologies in determining the presence of carbohydrates. Also, give the basic principle for the test.

