Use the References to access important values if needed for this question. (A) A sample of oxygen gas at a pressure of 0.742 atm and a temperature of 295 °C, occupies a volume of 672 mL. If the gas is heated at constant pressure until its volume is 922 mL, the temperature of the gas sample will be °C. (B) A sample of oxygen gas at a pressure of 942 mm Hg and a temperature of 57 °C, occupies a volume of 9.01 liters. If the gas is heated at constant pressure to a temperature of 77 °C, the volume of the gas sample will be L. Submit Answer 5 question attempts remaining
Use the References to access important values if needed for this question. (A) A sample of oxygen gas at a pressure of 0.742 atm and a temperature of 295 °C, occupies a volume of 672 mL. If the gas is heated at constant pressure until its volume is 922 mL, the temperature of the gas sample will be °C. (B) A sample of oxygen gas at a pressure of 942 mm Hg and a temperature of 57 °C, occupies a volume of 9.01 liters. If the gas is heated at constant pressure to a temperature of 77 °C, the volume of the gas sample will be L. Submit Answer 5 question attempts remaining
Chapter6: Random Errors In Chemical Analysis
Section: Chapter Questions
Problem 6.22QAP
Related questions
Question
Transcribed Image Text:People
Tab
Window
Help
14% O
Sun 9:41 PM
gage Learning Online Asse x
Mind Tap - Cengage Learning
ro/index.html?deploymentld%355750828934189288909969212&elSBN=9781305657571&nbld%3D2199898&snapshotld... ☆
NDTAP
Q Search this cou
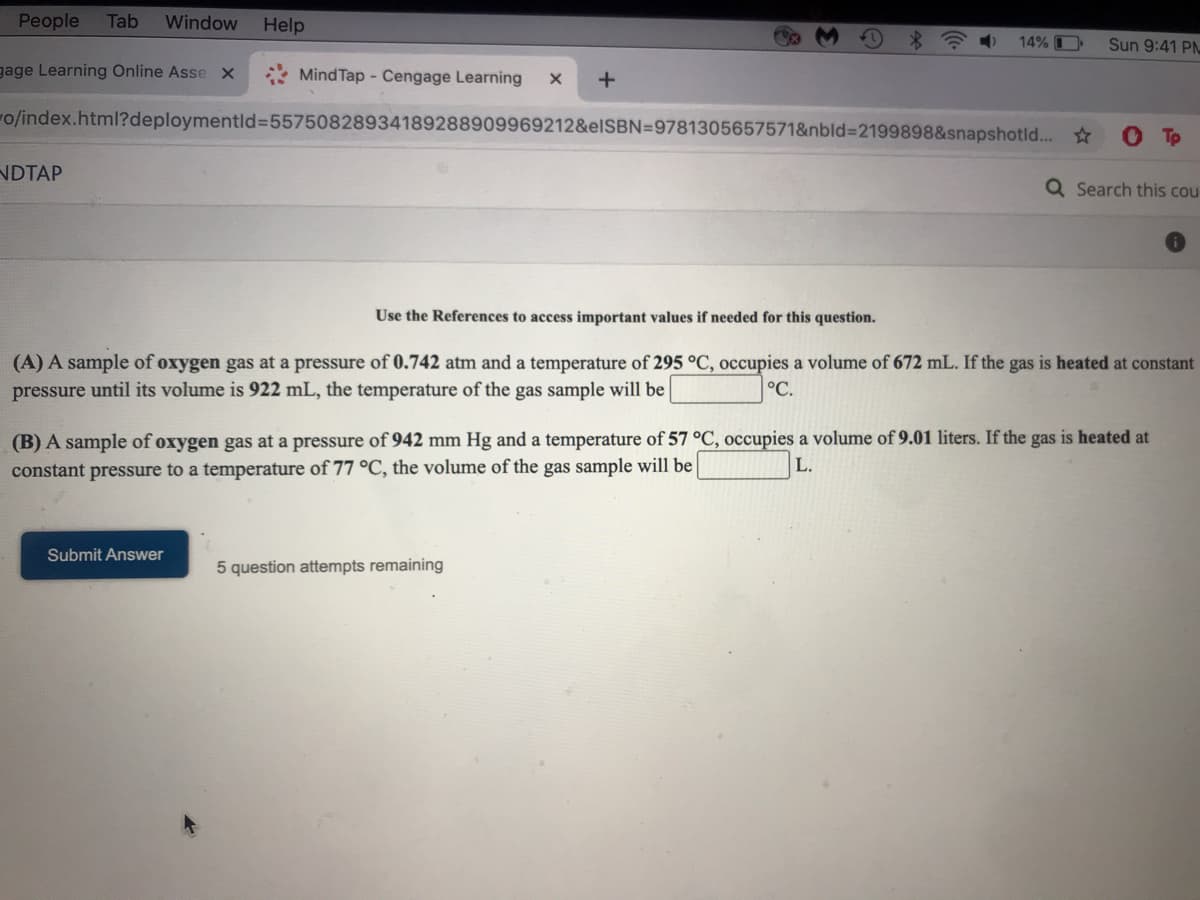
Use the References to access important values if needed for this question.
(A) A sample of oxygen gas at a pressure of 0.742 atm and a temperature of 295 °C, occupies a volume of 672 mL. If the gas is heated at constant
pressure until its volume is 922 mL, the temperature of the gas sample will be
°C.
(B) A sample of oxygen gas at a pressure of 942 mm Hg and a temperature of 57 °C, occupies a volume of 9.01 liters. If the gas is heated at
constant pressure to a temperature of 77 °C, the volume of the gas sample will be
L.
Submit Answer
5 question attempts remaining
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



