Use this graph to answer the following questions. Round each of your answers to the nearest m/s. Note: your answers must be within 25 m/s of the exact answers to be graded correct. m What is the most likely speed of a Xe atom in this sample? 0-²-2- S m What higher speed is only half as likely as the most likely speed? S What higher speed is only 10% as likely as the most likely speed? S
Use this graph to answer the following questions. Round each of your answers to the nearest m/s. Note: your answers must be within 25 m/s of the exact answers to be graded correct. m What is the most likely speed of a Xe atom in this sample? 0-²-2- S m What higher speed is only half as likely as the most likely speed? S What higher speed is only 10% as likely as the most likely speed? S
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter5: Gases
Section: Chapter Questions
Problem 5.67PAE
Related questions
Question
100%
Here is a graph of the probability of an atom moving with a particular speed for example of xenon gas -58. Degrees Celsius. Use this graph to answer the following questions . Round each of your answers to the nearest m/s.
Transcribed Image Text:Use this graph to answer the following questions. Round each of your answers to the nearest m/s.
Note: your answers must be within 25 m/s of the exact answers to be graded correct.
m
What is the most likely speed of a Xe atom in
this sample?
m
What higher speed is only half as likely as the
most likely speed?
m
What higher speed is only 10% as likely as
the most likely speed?
X
?
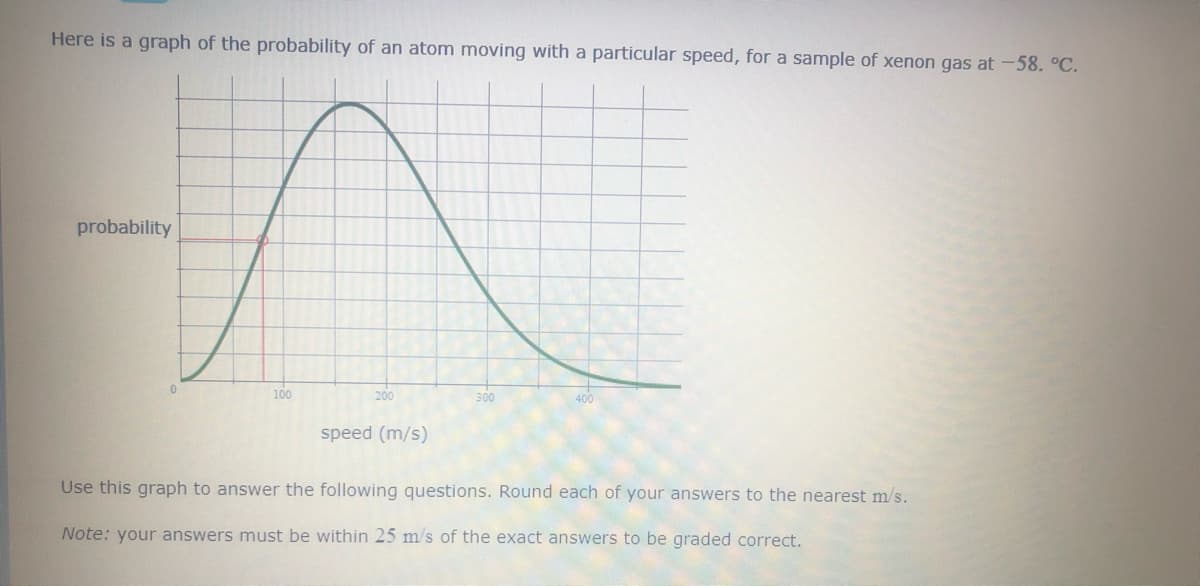
Transcribed Image Text:Here is a graph of the probability of an atom moving with a particular speed, for a sample of xenon gas at -58. °C.
probability
100
200
300
400
speed (m/s)
Use this graph to answer the following questions. Round each of your answers to the nearest m/s.
Note: your answers must be within 25 m/s of the exact answers to be graded correct.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning