Using s rd reduction potentials from the ALEKS Data tab, calculate the standard reaction free energy AG" for the following redox reaction. Round your answer to 2 significant digits. 3Fe* (aq) + Cro (ag)+ 4H,0 (1) → 3Fe* (aq) + Cr(OH), (s) + 50H (aq) dla Data Cr2+(aq)+2e¯¬Cr(s) -0.913 Cr3+ (aq)+e+Cr+(aq) Cr3+(aq)+3e¯+Cr(s) -0.407 -0.744 Cro4? (aq)+4H20(1)+3e¯¬Cr(OH)3(s)+50H"(aq) Cr20,2 (aq)+14H*(aq)+6e¯¬2Cr³+(aq)+7H2O(1) -0.13 1.36 Cu*(aq)+e→Cu(s) 0.521 Cu2+(aq)+e¯¬Cu*(aq) Cu2+(aq)+2e¯¬Cu(s) 0.153 0.342 F2(g)+2e-2F (aq) 2.866 Fe2+(aq)+2e¬Fe(s) -0.447 Fe3+ (aq)+e-Fe?+(aq) 0.771 Fe3+(aq)+3e¬Fe(s) -0.037 2H*(aq)+2e¯¬H2(g) 0.000 2H20(1)+2e-H2(9)+2OH"(aq) -0.828 Explanat H202(aq)+2H*(aq)+2e¯→2H2O(I) 1.776 Ha2+(ag)+2e-Ha) 0.851 enter
Using s rd reduction potentials from the ALEKS Data tab, calculate the standard reaction free energy AG" for the following redox reaction. Round your answer to 2 significant digits. 3Fe* (aq) + Cro (ag)+ 4H,0 (1) → 3Fe* (aq) + Cr(OH), (s) + 50H (aq) dla Data Cr2+(aq)+2e¯¬Cr(s) -0.913 Cr3+ (aq)+e+Cr+(aq) Cr3+(aq)+3e¯+Cr(s) -0.407 -0.744 Cro4? (aq)+4H20(1)+3e¯¬Cr(OH)3(s)+50H"(aq) Cr20,2 (aq)+14H*(aq)+6e¯¬2Cr³+(aq)+7H2O(1) -0.13 1.36 Cu*(aq)+e→Cu(s) 0.521 Cu2+(aq)+e¯¬Cu*(aq) Cu2+(aq)+2e¯¬Cu(s) 0.153 0.342 F2(g)+2e-2F (aq) 2.866 Fe2+(aq)+2e¬Fe(s) -0.447 Fe3+ (aq)+e-Fe?+(aq) 0.771 Fe3+(aq)+3e¬Fe(s) -0.037 2H*(aq)+2e¯¬H2(g) 0.000 2H20(1)+2e-H2(9)+2OH"(aq) -0.828 Explanat H202(aq)+2H*(aq)+2e¯→2H2O(I) 1.776 Ha2+(ag)+2e-Ha) 0.851 enter
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter18: Electrochemistry
Section: Chapter Questions
Problem 18.102QE: At 298 K, the solubility product constant for Pb(IO3)2 is 2.6 1013, and the standard reduction...
Related questions
Question
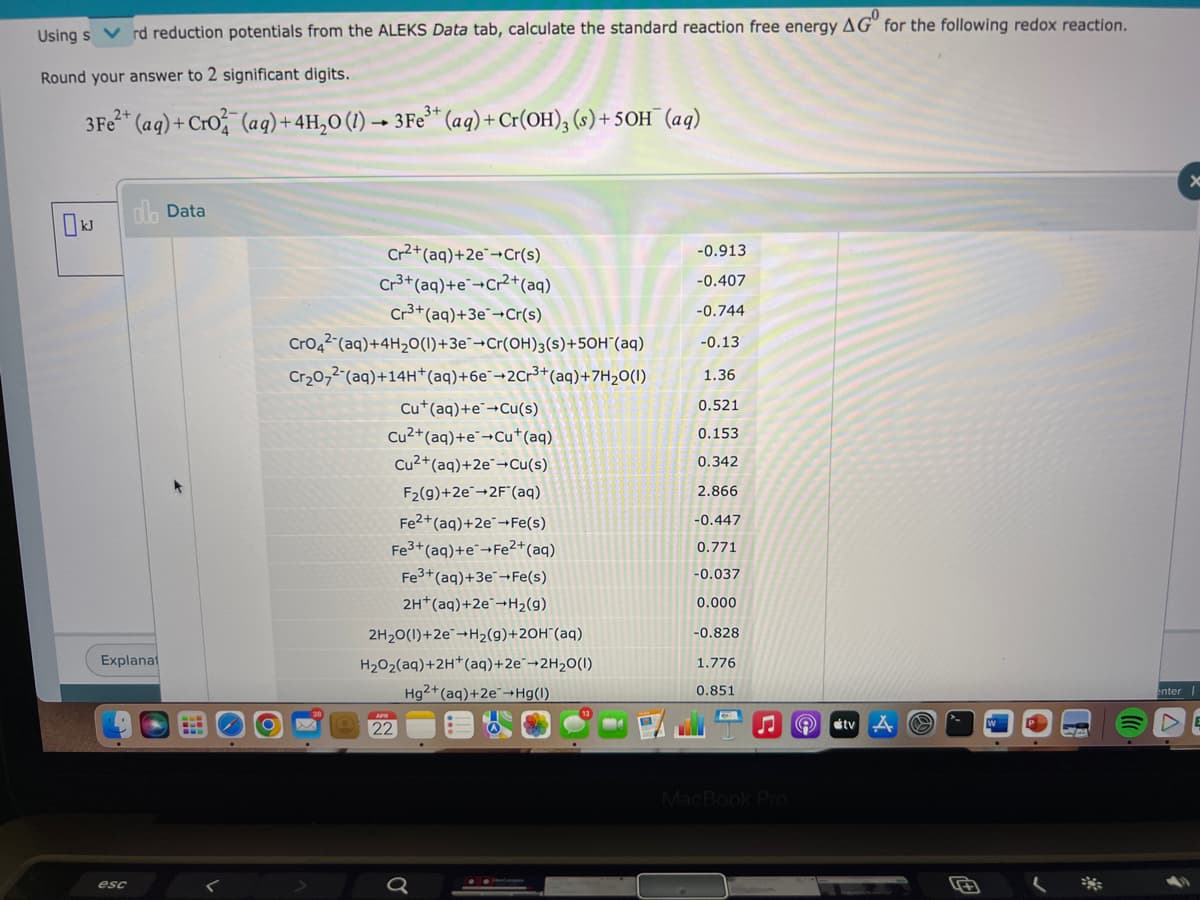
Transcribed Image Text:Using s
rd reduction potentials from the ALEKS Data tab, calculate the standard reaction free energy AG" for the following redox reaction.
Round your answer to 2 significant digits.
3Fe* (ag) + Cro (aq) +4H,0 (1) –→ 3Fe* (aq) +Cr(OH), (s) + 5OH¯ (aq)
dlh Data
Cr2+(aq)+2e¯¬Cr(s)
Cr3+(aq)+e¯¬Cr²+(aq)
Cr3+(aq)+3e¯¬Cr(s)
-0.913
-0.407
-0.744
Cro42 (aq)+4H20(1)+3e¯¬Cr(OH)3(s)+50H"(aq)
-0.13
Cr20,2-(aq)+14H*(aq)+6e¯¬2Cr³+(aq)+7H2O(1)
1.36
Cu*(aq)+e¯→Cu(s)
0.521
Cu2+(aq)+e¯¬Cu*(aq)
Cu2+(aq)+2e¯¬Cu(s)
0.153
0.342
F2(9)+2e-2F"(aq)
2.866
Fe2+(aq)+2e¯¬Fe(s)
Fe3+ (aq)+e¬Fe?+(aq)
Fe3+(aq)+3e¯¬Fe(s)
-0.447
0.771
-0.037
2H*(aq)+2e¯¬H2(g)
0.000
2H20(1)+2e"→H2(g)+2OH"(aq)
-0.828
Explanat
H202(aq)+2H*(aq)+2e¨→2H2O(1)
1.776
Hg2+(aq)+2e¯¬Hg(I)
0.851
enter
22
tv
MacBook Pro
esc
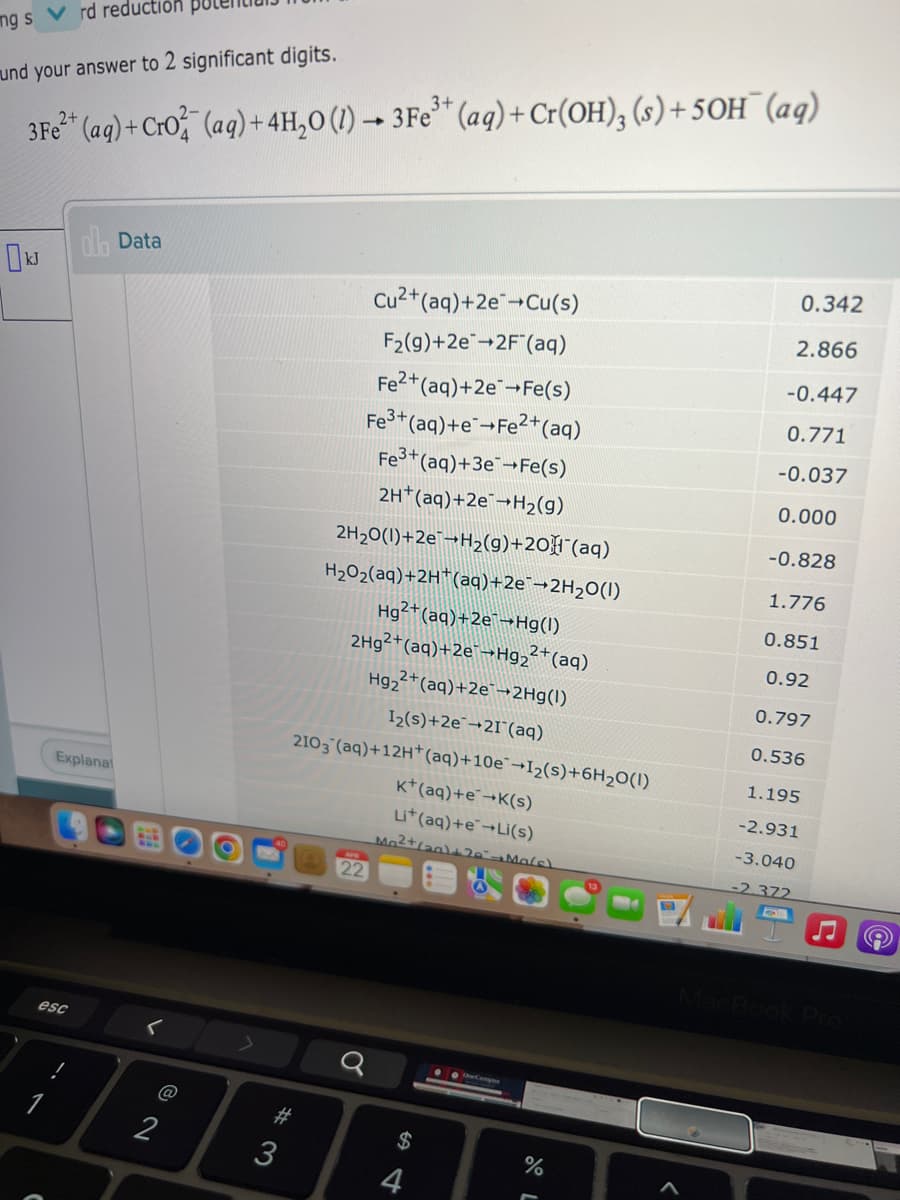
Transcribed Image Text:ng s V rd reductión
und your answer to 2 significant digits.
3+
3Fe (ag) + CrO (ag) + 4H,0 (1) → 3Fe** (aq) + Cr(OH), (s) + 5OH¯ (aq)
0.342
IJ olb Data
Cu2+(aq)+2e+Cu(s)
2.866
F2(g)+2e-2F"(aq)
-0.447
Fe2+(aq)+2eFe(s)
0.771
Fe+(aq)+e¯+Fe2+(aq)
-0.037
Fe*(aq)+3eFe(s)
0.000
2H*(aq)+2E¯¬H2(g)
-0.828
2H20(1)+2E¯¬H2(g)+20F"(aq)
H2O2(aq)+2H*(aq)+2e¯¬2H2O(I)
1.776
Hg2+(aq)+2e¯¬Hg(I)
0.851
2H9²+(aq)+2e¯¬Hg2²†(aq)
0.92
Hg, 2+(aq)+2e¯-2Hg(l)
0.797
I2(s)+2e¯¬2I°(aq)
0.536
2103 (aq)+12H*(aq)+10e¯→I2(s)+6H2O(I)
1.195
Explanat
K*(aq)+e¬K(s)
-2.931
Li*(aq)+E¯→Lİ(s)
-3.040
-2.372
22
esc
8888
@
#
2
2$
3
4
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning