Using the data you have collected for Part II of the experiment, complete the following table. For the freezing point depression calculation, be sure to use the average value for the freezing point of pure water and the value for the molality of the solution that you entered earlier. Trial # Tf (ave), pure H₂O (°C) Unrounded ● Temperature values should be entered in to nearest 0.1 °C. Tf, NaCl (aq) (°C) Freezing Point Depression of NaCl (aq) (AT) (°C) Unrounded Rounded Freezing Point Constant of Water, Kf (°C/m) Unrounded Rounded
Using the data you have collected for Part II of the experiment, complete the following table. For the freezing point depression calculation, be sure to use the average value for the freezing point of pure water and the value for the molality of the solution that you entered earlier. Trial # Tf (ave), pure H₂O (°C) Unrounded ● Temperature values should be entered in to nearest 0.1 °C. Tf, NaCl (aq) (°C) Freezing Point Depression of NaCl (aq) (AT) (°C) Unrounded Rounded Freezing Point Constant of Water, Kf (°C/m) Unrounded Rounded
Chapter4: Least-squares And Calibration Methods
Section: Chapter Questions
Problem 3P
Related questions
Question
please help me find the average and standard deviation
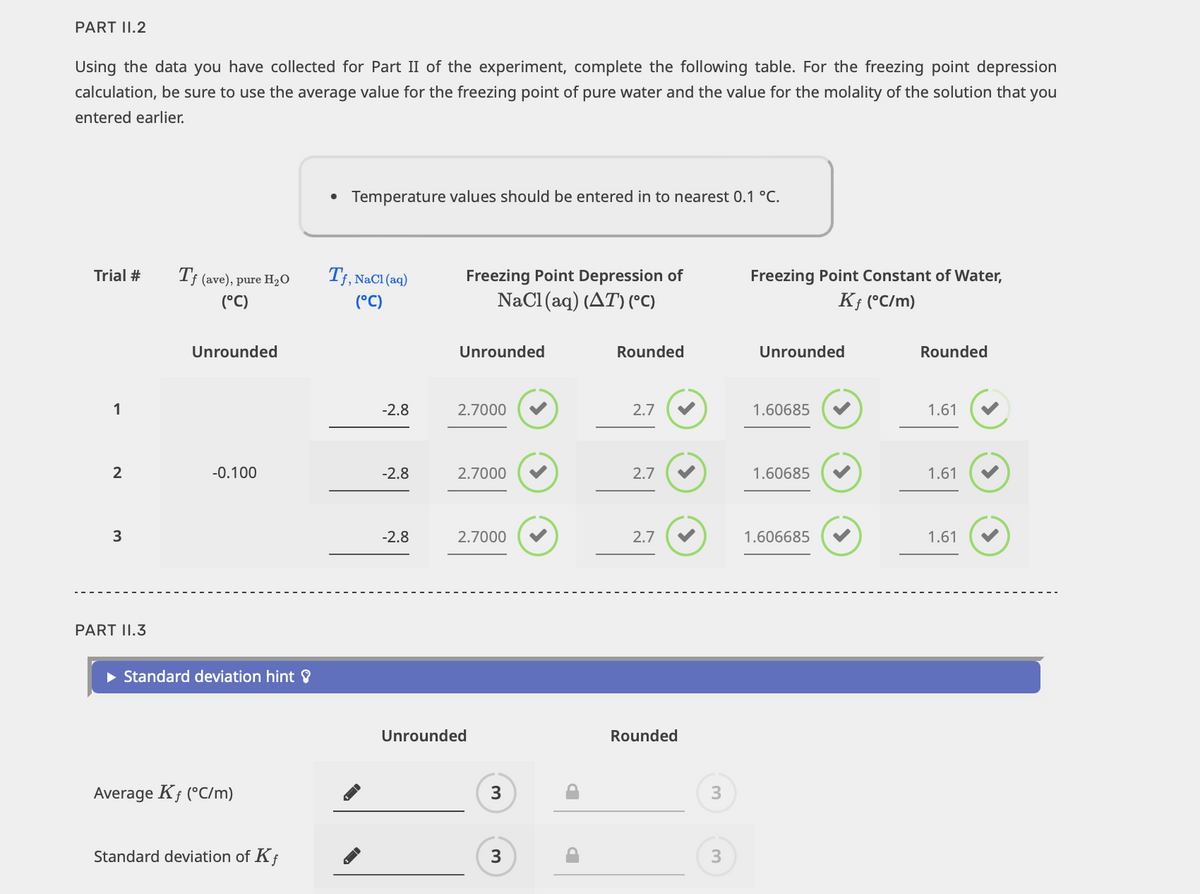
Transcribed Image Text:PART 11.2
Using the data you have collected for Part II of the experiment, complete the following table. For the freezing point depression
calculation, be sure to use the average value for the freezing point of pure water and the value for the molality of the solution that you
entered earlier.
Trial #
1
2
3
PART 11.3
Tf (ave), pure H₂O
(°C)
Unrounded
-0.100
► Standard deviation hint ?
Average Kf (°C/m)
Standard deviation of Kf
• Temperature values should be entered in to nearest 0.1 °C.
Tf, NaCl (aq)
(°C)
-2.8
-2.8
-2.8
Freezing Point Depression of
NaCl(aq) (AT) (°C)
Unrounded
2.7000
2.7000
2.7000
Unrounded
3
3
Rounded
2.7
2.7
2.7
Rounded
3
3
Freezing Point Constant of Water,
Kf (°C/m)
Unrounded
1.60685
1.60685
1.606685
Rounded
1.61
1.61
1.61
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 5 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT



EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning