Using the model above, illustrate the urea cycle.
Chapter21: Heparin Infusion Calculations
Section: Chapter Questions
Problem 9SST
Related questions
Question
100%
Using the model above, illustrate the urea cycle.
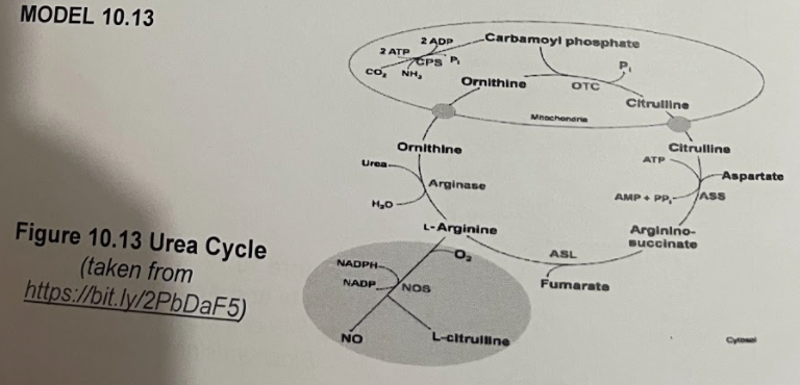
Transcribed Image Text:MODEL 10.13
Figure 10.13 Urea Cycle
(taken from
https://bit.ly/2PbDaF5)
2 ATP
co,
Urea-
H₂O
NADPH-
NADP
NO
2 ADP
CPS
NH₂
Ornithine
Carbamoyl phosphate
OTC
NOS
Ornithine
Arginase
L-Arginine
O₂
L-citrulline
Mnachoneria
ASL
Fumarate
Citrulline
ATP
AMP+ PP-
Citrulline
ASS
Arginino-
succinate
Aspartate
Cytowal
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you


Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax


Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Biochemistry
Biochemistry
ISBN:
9781305961135
Author:
Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:
Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College