Q: The compound MgBr2 is an ionic compound. What are the ions of which it is composed? Cation formula...
A: Since we know that ionic compounds are composed of two ions , in which one is cation having positive...
Q: 13.58 An automobile antifreeze mixture is made by mixing equal volumes of ethylene glycol (d = 1.114...
A: To solve this problem let us consider the volume of the mixture i.e solution = 1000 mL So the volum...
Q: The half-life for Americium-241 is ______432 years__________. ? A smoke detector requires about 0.3 ...
A: As per guidelines we are allowed to answer one question but first question answer is very small , so...
Q: Copper (2) fluoride contains 37.42% F by mass. use this percentage to calculate the mass of fluorine...
A: Mass percentage = (mass of a component in a mixture/total mass of mixture) × 100%
Q: A parkinsonic 61-year-old man complained to his physician that recently he felt increasingly confuse...
A: Given: A parkinsonic 61-year-old man complained to his physician that recently he felt increasingly ...
Q: Propose a structure for an aromatic hydrocarbon, C10H14, that can form only one C10H13Cl product on ...
A: The single substitution product will be : The compound from which the above-given compound prepared...
Q: Would the average rate of decomposition be negative since concentration is decreased ?
A: The rate of the reaction is defined as the change in the concentration of the reactants or products ...
Q: A sample of the element Phosphorus is shown to have a mass of 33 and å charge of -3. HoW many Heutro...
A: Neutral Phosphorus having mass 30.9 P having mass 33. Atomic number of P is 15, also atomic number o...
Q: If the pH is 2, what is the %HA at of a molecule has a pKa = 4? Report the answer to one dpcimal pla...
A: Given : pH of solution = 2 And pKa of acid = 4. The partial dissociation reaction of HA can be writ...
Q: A sample of C₃H₄Br₂ has a normal boiling temperature of 131.5 °C. The enthalpy of vaporization for t...
A: The heat required to change the temperature form T1= 97.5 °C ...
Q: At a certain temperature, 0.640 mol SO3 is placed in a 2.50 L container. 2SO3 (g)↽−−⇀2SO2 (g)+O2 (...
A: Sulfur trioxide decomposes into sulfur dioxide and oxygen. The equation for the balanced chemical re...
Q: Calculate the pH of a 2.00 M Na2CO3 given that the pKa’s of the first and second dissociation of H2C...
A: (b) Given For H2CO3 , pKa1 = 6.37 , pKa2 = 10.32 Molarity of Na2CO3 solution = 2.00 M Molarity of...
Q: Suppose you have a mixture of copper sulfate (CuSO4) and azulene (C10H8, you may wish to know the st...
A: Given information: A mixture of copper sulfate (CuSO4) and azulene
Q: If 5.3 g of gallium reacts with 5.3 g of oxygen to produce gallium oxide, what will we be left with ...
A: Limiting reactant :- A reactant whose number of moles are less than the number of moles required a...
Q: Identify all possible product(s) in the following reactions. a) 1-bromocyclohexane with sodium hydro...
A: NaOH follows SN2 mechanism with secondary alkyl halide. Tertiary alkyl halide undergoes SN1 reactio...
Q: What is product of the following reaction? 1. xs NABH4 OH 2. H3O* OH ОН он HO. ОН OH `OH OH HO,
A: The reaction given is,
Q: What is the osmotic pressure when 0.3 g of nonelectrolyte compound A (MW 75) is diluted to 100 ml of...
A: The given data contains, number of moles =0.3 g75 g/mol=0.004 moles Temperature =298 K Volume =100 m...
Q: A patient requires an injection of 0.012 g of a pain killer available as a 1500 mg/dL solution. How ...
A: Given : Mass of drug required = 0.012 g = 12 mg (since 1 g = 1...
Q: Determine the number of lone pairs on the nitrogen atom in the following structures.
A:
Q: At 18°C a 0.001-M aqueous solution of potassium hydrogen carbonate,KHCO3 , conducts a current in a c...
A: Potassium bicarbonate dissolve in water. It dissociate into water to produce potassium ion ( K+) an...
Q: Draw the following molecule and answer the questions below. H3CCHCHC(O)CH2CCH What is the valence el...
A: Since you have posted question with multiple sub-parts, we are entitled to answer the first 3 only. ...
Q: Write complete nuclear equations for the following processes: 1.The carbon-14 isotope undergoes bet...
A: 1) Given : Beta decay of carbon 14 i.e 146C.
Q: Explain what do you mean by Radioactive dating?
A: Radioactive decay (also known as nuclear decay, radioactivity, radioactive disintegration or nuclear...
Q: Consider the following molecules: Na2O, HCl, NH3, N2, CH3CH2OH. Which would have the highest viscosi...
A: Viscosity is the property of the the fluids, It is resistance o the flaw of the fluids. When the flu...
Q: Please help
A: Osmotic pressure is a colligative property. The osmotic pressure of a solution can be calculated by ...
Q: An aliquot of a 8.50 M stock aqueous solution of sodium chloride is used to create 800. mL of a 0.10...
A: Given ; Concentration of dilute solution = 0.100 M And volume of dilute solution = 800 mL = 0.800 L ...
Q: Chemistry Question
A: To explain whether the given test is helpful or not in identifying the substance.
Q: QUESTION 16 The rate constant for a second-order reaction is 0.54 M1s O 6.9s What is the half-hfe of...
A: The given data contains, rate constant for second order reaction is 0.54 M-1s-1. concentration is 0....
Q: Please show all work
A: number of moles = mass ÷ molar mass molar mass of C6H14 = 86.18 g/mol at STP 1 mol = 22.4 L
Q: Determine the geometry about each interior atom in each molecule and draw the molecule. (Skeletal st...
A: (a) CH3OH The Lewis structure of the molecule gives four electron pairs on the carbon atom with no l...
Q: Consider the following molecules: Na2O, HCl, NH3, N2, CH3CH2OH. Which would have the highest viscosi...
A: Viscosity is defined as any fluid's resistance to flow. Higher viscosity means that the resistance o...
Q: Why does the ideal gas law predict a pressure for the Ar sample with a volume of 0.500 L at 160 K th...
A: Given : The ideal gas law predict a pressure for the Ar sample with a volume of 0.500 L at 160 K tha...
Q: What is the IUPAC name of the following alkanes?
A: IUPAC nomenclature Identify the longest carbon chain. Identify the functional groups present. Give ...
Q: Which of the following could enter into H-bonding interactions with water? (Select all that apply.) ...
A:
Q: The value of Kp for the reaction 2 A(g) + B(g) + 3 C(g) 2D(g) + E(g) is 19090 at a particular temper...
A: The reaction taking place is given as, 1) 2 A (g) + B (g) + 3 C (g) -------> 2 D (g) + E (g) Giv...
Q: Describe problems encountered with chemical pesticides.
A: The problems encountered with chemical pesticides has to be given,
Q: if the unknown liquid does not completely vaporize during the heating process, will the formula weig...
A: In the question, we need to determine upon heating if the unknown liquid does not completely vapori...
Q: How much heat (kJ) is absorbed by 948.0 g of water in order for the temperature to increase from 25....
A: Specific heat: The amount of heat energy required to raise the temperature of one gram of a substanc...
Q: 3. A solution contains 5.0 g ethylene glycol, C2H«(OH)2, and 95 g water, H2O, by mass. The density o...
A: Mass of ethylene glycol = 5 g Molar mass of ethylene glycol = 62.07 g/mol Mass of water = 95 g Densi...
Q: How are the fatty acids from adipose tissue transported?
A: Fatty acids are released from adipose tissue to become water soluble they associate with albumin, a ...
Q: If you have a sample which has an absorbance of 1.17, a molar absorptivity of 1 Mcm1, and a cuvette ...
A: The given data contains, Absorbance=1.17path length=1.15 cmMolar absorptivity=1 M-1.cm-1
Q: Write the symbol for an atom/ion with the following information 2+ charge, atomic number 38, and mas...
A: To write the symbol for an atom/ion that has a charge of +2, atomic number 38 and mass number 87
Q: Show that the number of electrons in the shell equals 2n2 and that the number in each subshell is 2(...
A: The number required to denote the energy, shell, subshell are termed as quantum number.
Q: Write the balanced chemical equation between BH 3 and water. Label out the oxidizing agent and reduc...
A: Given BH3 ,H2O Oxidizing agent is the one which provides oxygen for oxidation process.(oxidation i...
Q: Greetings and thank you for help 4. A titration was done where 50ml of 0.1M hydrochloric acid neutra...
A: Given : Concentration of hydrochloric acid i.e HCl = 0.1 M Volume of HCl solution = 50 mL And volume...
Q: Explain please
A: Given data=> mass of polypeptide = 2.63 g Volume of solution = 239 mL Osmotic pressure = 0...
Q: Give mathematical explanation for the effect of temperature on viscosity.
A: The property of the liquids which determines their resistance to flow is called viscosity. Temperatu...
Q: I have no idea what to do.
A:
Q: Radius of an atomic nucles is of the order 2.2fm. What is the minimum energy that an electron should...
A:
Q: How many hours are in exactly 18 days? The answer has how many stick figures?
A: Since we know that 1 day = 24 hours So by using this relation we will calculate the number of hour...
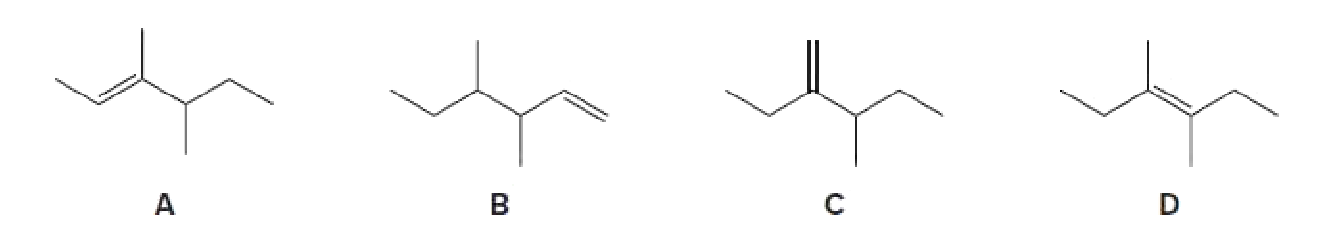
Rank the following
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images




