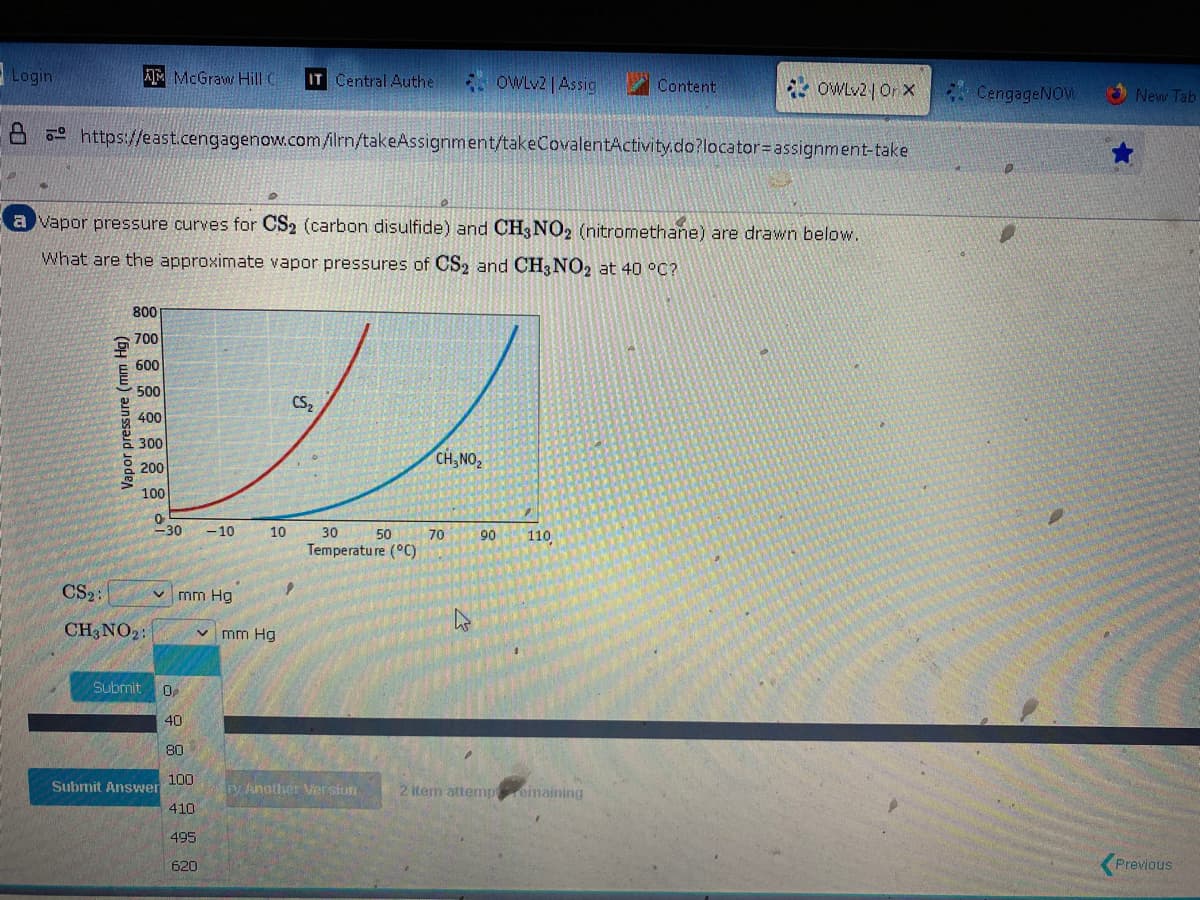
Vapor pressure curves for CS₂ (carbon disulfide) and CH3 NO₂ (nitromethane) are drawn below. What are the approximate vapor pressures of CS2 and CH3 NO2 at 40 °C? Vapor pressure (mm Hg) 800 700 600 500 400 300 200 100 0 <-30 -10 CS₂: CH3 NO₂: mm Hg 10 ✓ mm Hg CS₂ 30 50 Temperature (°C) CH, NO₂ 70 90 110
Vapor pressure curves for CS₂ (carbon disulfide) and CH3 NO₂ (nitromethane) are drawn below. What are the approximate vapor pressures of CS2 and CH3 NO2 at 40 °C? Vapor pressure (mm Hg) 800 700 600 500 400 300 200 100 0 <-30 -10 CS₂: CH3 NO₂: mm Hg 10 ✓ mm Hg CS₂ 30 50 Temperature (°C) CH, NO₂ 70 90 110
Macroscale and Microscale Organic Experiments
7th Edition
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Kenneth L. Williamson, Katherine M. Masters
Chapter7: Extraction
Section: Chapter Questions
Problem 1Q
Related questions
Question
Transcribed Image Text:Login
AM McGraw Hill C IT Central Authe
Vapor pressure (mm Hg)
https://east.cengagenow.com/ilrn/takeAssignment/takeCovalentActivity.do?locator assignment-take
800
700
600
500
400
300
200
100
0
<-30
a vapor pressure curves for CS2 (carbon disulfide) and CH3 NO2 (nitromethane) are drawn below.
What are the approximate vapor pressures of CS2 and CH3 NO2 at 40 °C?
CS₂:
CH3 NO₂:
✓mm Hg
Submit 0₂
40
80
100
410
495
Submit Answer
-10
V mm Hg
620
10
CS₂
30
50
Temperature (°C)
ry Another Version
CH, NO₂
OWLv2 | Assig
70
90
Content
110
OWLv2 | Orx
2 item attempemaining
CengageNOW
New Tab
Previous
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole


Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole
