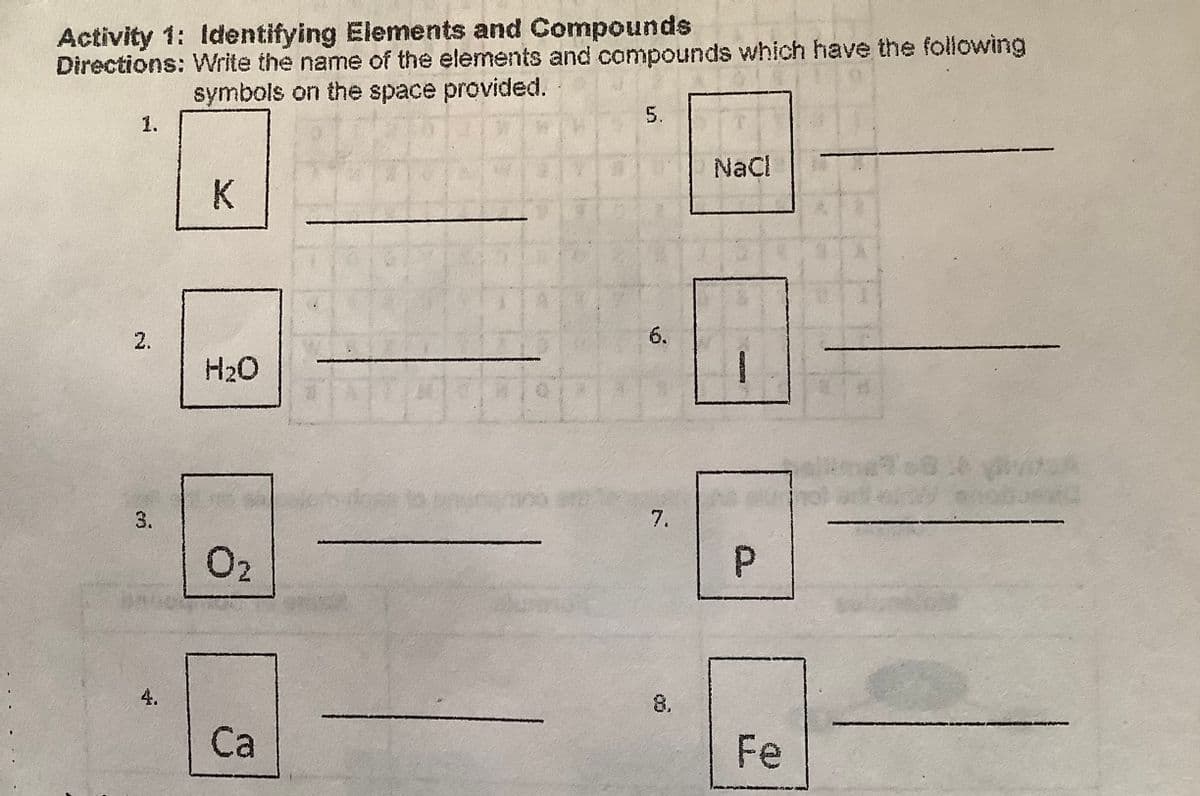
vity 1: Identifying Elements and Compounds tions: Write the name of the elements and compounds which have the following symbols on the space provided. 5. 1. NaCl K
Q: 405 163 405163 from D Exit Ticket dame: Haya Date: ADMIT ADMIT dentify the total number of atoms,…
A:
Q: Mg(OH), is a compound. How many different elements are found in this compound? 3.
A: The substance which is formed by one type of atoms are known as element.
Q: AB CD CB Mg2+Cl₁- Li¹+02- MgCl2 Li₂O AD LI¹+Cl¹- LICI ANSWER: LICI + MgO Mg2+O²- MgO A (1st cation)…
A:
Q: 13. СО, # of molecules: # of elements: Name of element: # of atoms Total # of atoms: The #2 is a…
A: The name of the element and number of atoms in the given molecules has to be given.
Q: PbCO3(s)→PbO(s)+CO2(g)PbCO3(s)→PbO(s)+CO2(g) Express your answers as integers separated by commas
A: Note: Assuming that the question is to balance the given reaction.
Q: Copper(II) sulfate forms a bright blue hydrate with the formula CuSO4⋅?H2O(s) . If this hydrate is…
A: Molar mass of Cu is 63.55 g/mol Molar mass of S is 32.07 g/mol Molar mass of O is 16 g/mol Molar…
Q: 4. Which is not a physical change? a. Melting of candle c. breaking a glass b. Digestion of…
A: We will answer the first question since the exact one wasn't specified. Please submit a new question…
Q: 1.1) What is a balance used for ? 1.2) What are the four major type of balances used ? 1.3) Why when…
A: 1.1) In chemistry, a weighing balance is used to determine the weight or mass of the chemical…
Q: Element X forms three different compounds with element Y. Information about these compounds is…
A: We have to predict the formula for the other two compounds.
Q: 8. You work for a company called Farquahr and Daughters. Your expense account allows you to spend…
A: Given, Allowance for spend = 80.00 $/day Total days of trip = 1 week = 7 days Currency exchange rate…
Q: MCQ 27: Alcohol (C,HOH) with lowest molecular mass at room temperature is A. solid B. liquid C. gas…
A: An alcohol can be defined as an aliphatic system in which one or more hydroxyl group(s) are directly…
Q: 04. During chemical reactions, atoms combine or combination of atoms break apart and the atoms…
A: For a chemical reaction to occur, the rearrangement of atoms has to take place. In this process,…
Q: For methylcyclohexane, shown below, the total number of 1°, 2° and 3° hydrogens, respectively is O…
A: The correct answer is given below
Q: A hydrate initially weighs 5.672 grams and is heated. After heating, it weighs 4.483 grams.…
A: Given ,Initial hydrate weighs = 5.672 gAfter heating weighs = 4.483 gSo,mass in water form = Initial…
Q: In the following images, the white spheres represent hydrogen atoms and the red spheres represent…
A:
Q: Part A Find the number of ibuprofen molecules in a tablet containing 155.0 mg of ibuprofen (C13 H18…
A: Given , Mass of ibuprofen = 155.0 mg = 0.155 g And , Standard value, Molar mass of ibuprofen =…
Q: A 194g sample of caffeine contains 6.02 *10^23 molecules of caffeine. If a typical 10-hour energy…
A: 1000 mg = 1 g Given that: Amount of caffeine = 422 mg Number of molecules of caffeine per 194 g =…
Q: 1.12 The photo below shows a picture of an agate stone. Jack,who picked up the stone on the Lake…
A: Pure chemical substances that are made up of two or more chemical elements are known as chemical…
Q: Use the web to determine the mass of a steel beverage can from the 1970s and the mass of a modern…
A: Mass is a property, which represents the amount of substance present in a sample and it is generally…
Q: Part A The American Dental Association recommends that an adult female should consume 3.0 mg of…
A:
Q: H ball & stick H G C + labels 27 H H ball & stick labels The name of the first compound is The name…
A: Ball- stick model is the 3D representation of a molecule. In IUPAC nomenclature generally we find…
Q: Which of the following accurately describes the number of aluminum and oxygen atom A 3 atoms of…
A: Al2O3 is named as Alumina. It is a compound of aluminium.
Q: A chemist is trying to identify an unknown metal, and finds that 25.0 cm³ of the substance has a…
A: Given: Volume of the metal, V=25 cm3 Mass of the metal, m=224.43 g Temperature, T=20°C
Q: lead ball is added to 158 grams of water in a coffee cup calorimeter. If the ball loses 47.35 °C of…
A:
Q: Balance the following reaction by placing the coefficients into the blanks. F2 + AICI3 AIF3 --> Cl2
A: A blanced chemical equation means it contains equals number each element same on product side and…
Q: Namethe chemical compound, K-0. Uselowercase letters only. Your answer Namethe chemical compound,…
A:
Q: An unknown substance has a mass of 13.9 gg . When the substance absorbs 1.251×102 JJ of heat, the…
A: Given : Mass of the substance, m =13.9 g Heat absorbed, q=1.251×102 J Temperature change,…
Q: What are the missing coefficients for the equation below? Al,(SO4)a(aq) + KOH(aq) → Al(OH)a(aq) +…
A: 1) The right answer is option D) 1,6,2,3 2) The right option is B) New atoms are formed as…
Q: A Chemical compound is formed by combination of different substances and therefore it is a mixture.…
A: The law of constant composition states that the ratio by mass of the elements in a chemical compound…
Q: Which of the following represents anammox? A. N2 → NH3 B. NO2- → NO C. NH3 → N2 D.…
A: Which of the following represents anammox?
Q: Typical household bleach sold in the store contains 0.89 M NaClO. How many grams of NaClO are in…
A:
Q: 1. Peter, James, nd John perfomed an experiment on acids, bases, and solvents inside the laboratory.…
A: Hello. Since your question has multiple parts, we will solve the first question for you. If you want…
Q: Label each of the following as an atomic element, a molecular element, or a compound.
A: Atomic element : It is a pure substance consisting of same atoms ( having same number of protons in…
Q: Name/Period: Koja Keley Type of Compound (Ionic or Molecular) Conversion Worksheet the following…
A: Given incomplete table is : Complete the table ?
Q: Empirical formula lab : compound Magnesium Mg metal with Chlorine Cl the chemical reaction involves…
A: The chemical change occurs when the chemical identity of a compound is destroyed to form a new…
Q: (1) Give to example of 1D Nano materials.
A:
Q: A mixture contains Aluminum Chloride, Sand and Sodium Chloride. Sodium Chloride makes up 22.48% of…
A: Given : Not all the aluminium chloride is sublimed.
Q: mass of matter (in grams)
A:
Q: Aspartame is an artificial sweetener that is 160 times sweeter than sucrose (table sugar) when…
A: Molecular formula of( aspartme) C14H18N2O5= No of molecules in aspartme = 1.1 x 1020 molecules…
Q: BE’s are: C=O 799, C–H 413, O–H 467, O–O 498, H–H 432 kJ/mol CO2(g) + 4H2(g) → CH4(g) + 2H2O(g)…
A: By using this we can calculate the ∆H°rxn for both the reaction .
Q: Question 7 An empirical formula always indicates the simplest whole-number ratio of different atoms…
A: we need to identify whether the given definition of empirical formula given in the question is…
Q: 2.115 Classify each of the following as a compound, a homoge- neous mixture, or a heterogeneous…
A: To solve this problem first we have to know about compound, mixture, homogeneous mixture and…
Q: Relerences) Choose examples of an element, a compound, a heterogeneous mixture, and a homogeneous…
A: Elements forms from same kind of atoms. Whereas , compound forms froms different kind of atoms.…
Q: the mass of o
A:
Q: A chemist has two alloys, one of which is 5% gold and 15% lead and the other which is 40% gold and…
A: The given information about the alloys are shown below.
Q: ome bismuth tablets, a medication used to treat upset stomachs, contain 262 mg of bismuth…
A: Stoichiometry involves the calculation of concentration of solutions in the given conditions of…
Q: PaterencentT 10 If powdered elemental calerum and powdered elemental iodine are poured into a metal…
A: Answer: B. Compound. Thermal treatment of Calcium powder with elemental iodine in a metal beaker,…
Q: 5. 3Na,SO, # of molecules: # of elements: Name of element: # of atoms Total # of atoms: The #3 is a…
A:
Q: LAW OF DEFINITE PROPORTION Direction: Answer the following problems. Show your solution. 3] A…
A: With the help of percent by mass we can calculate the ratio of each element present in the compound.
Q: How many different chemical compounds are in the gas phase? 2. 3. 4.
A: From the given picture, it is clearly shown that there are three different compound present in the…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- 1. Which of the following substance(s) displays properties of an ionic compound? Select all that apply 2. Which of the following substance(s) displays properties of a covalent compound? Select all that apply.Statement A: When a metal loses electron, it becomes an anion.Statement B: When a nonmetal gains electron, it becomes a cation.a. Both statements are incorrectb. Both statements are correctc. Statement A is incorrect, and Statement B is correctd. Statement A is correct, and Statement B is incorrectPls help with practice problem Provide proper, accepted chemical name
- Question 35. Name the following compound:( the first letter of all elements names should be lower case) SnO2Why is the ion of lead found in both Group 1 and Group 2? Please have an in-depth explantion thank you.Morton’s makes table salt, also known as sodium chloride. What is the chemical name, what is the generic name, and what is the brand name?
- Name FeCl3 with variable chargeGive the IUPAC (systematic) name for the compound GaNO3 Select one: a. gallium nitrate b. gallium (I) nitrate c. gallium mononitrate d. gallium (III) nitrate. Please show work, SO I can understand!Tu Re-order each list in the table below, if necessary, so that the atoms or lons in it are listed in order of decreasing size. Please don't provide the handwriting solution
- Consider NaBr and LiF. The LiF has a stronger ionic bond because A. The number of protons and electrons is greater for the Na and Br. B. There are less screening electrons in the Na and Br molecule C. The charge on Li and F is greater than the charge of the Na and Br D. the radius of Li and F is much smaller than the radius of Na and Br. Should 100% sure of final answer for any ratings.Which one of the following is not a match? Group of answer choices A. CO32- and carbonite ion B. CaCl2 and calcium chloride C. FeS and iron(II) sulfide D. NH4NO3 and ammonium nitrateWhich molecular formulas are also empirical formulas?SELECT ALL THAT APPLY. THERE IS MORE THAN ONE. C2H4 C5H5N5O C8H10N4O2 K2SO4 C6H6 Na2C2O4 C3H8 C6H12O6

