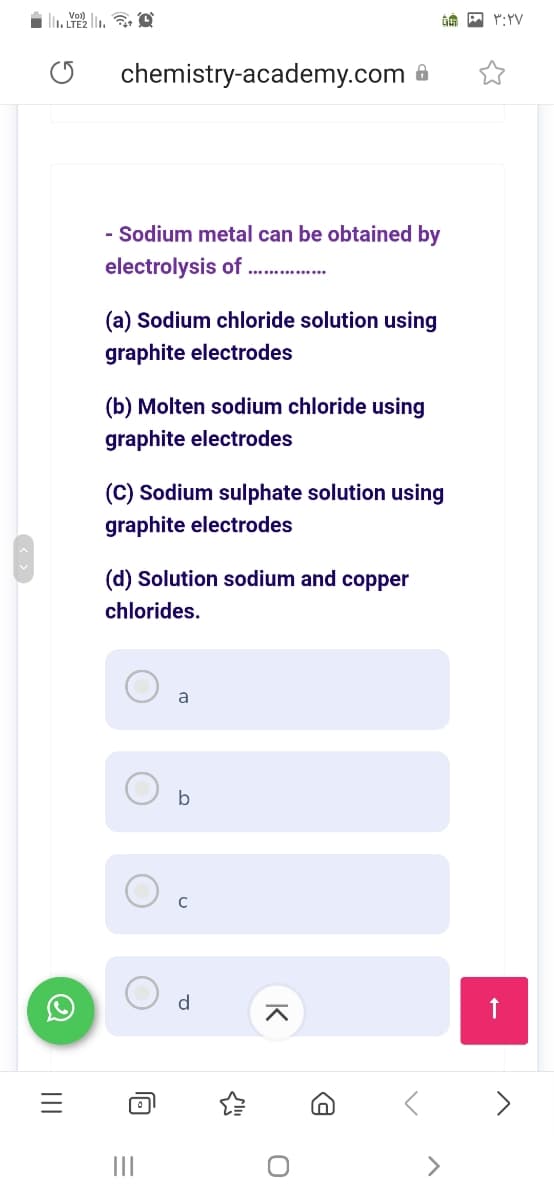
Vol) chemistry-academy.com 6 - Sodium metal can be obtained by electrolysis of . . (a) Sodium chloride solution using graphite electrodes (b) Molten sodium chloride using graphite electrodes (C) Sodium sulphate solution using graphite electrodes (d) Solution sodium and copper chlorides.
Vol) chemistry-academy.com 6 - Sodium metal can be obtained by electrolysis of . . (a) Sodium chloride solution using graphite electrodes (b) Molten sodium chloride using graphite electrodes (C) Sodium sulphate solution using graphite electrodes (d) Solution sodium and copper chlorides.
Chapter18: Introduction To Electrochemistry
Section: Chapter Questions
Problem 18.6QAP
Related questions
Question
Open the photo
Transcribed Image Text:chemistry-academy.com 8
- Sodium metal can be obtained by
electrolysis of . .
(a) Sodium chloride solution using
graphite electrodes
(b) Molten sodium chloride using
graphite electrodes
(C) Sodium sulphate solution using
graphite electrodes
(d) Solution sodium and copper
chlorides.
a
b
d
>
K
II
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax