Vrite the spontaneous reaction of the following reactions as a cell diagram. (If the reaction is not pontaneous, rearrange the reaction to make it spontaneous then write the cell diagram.) ollow the general format of a cell diagram: Anode II Cathode a. Nics) + Sn²* (aq) Ni2* (aq) + Sn(s) b. Curs) + Fe2* (aq) Cu2"(ag) + Fe(s) elow are standard reduction potential: Cu2+ (aq) + 2e- Cu (s) E° = +0.34 Sn2+ (aq) + 2e- Sn (s) E° = -0.14 Ni2+ 2e- Ni (s) E° = -0.26 + Fe2* (aq)+ 2e- Fe (s) Eo = -0.45
Vrite the spontaneous reaction of the following reactions as a cell diagram. (If the reaction is not pontaneous, rearrange the reaction to make it spontaneous then write the cell diagram.) ollow the general format of a cell diagram: Anode II Cathode a. Nics) + Sn²* (aq) Ni2* (aq) + Sn(s) b. Curs) + Fe2* (aq) Cu2"(ag) + Fe(s) elow are standard reduction potential: Cu2+ (aq) + 2e- Cu (s) E° = +0.34 Sn2+ (aq) + 2e- Sn (s) E° = -0.14 Ni2+ 2e- Ni (s) E° = -0.26 + Fe2* (aq)+ 2e- Fe (s) Eo = -0.45
Chapter18: Electrochemistry
Section: Chapter Questions
Problem 83E: Consider a concentration cell that has both electrodes made of some metal M. Solution A in one...
Related questions
Question
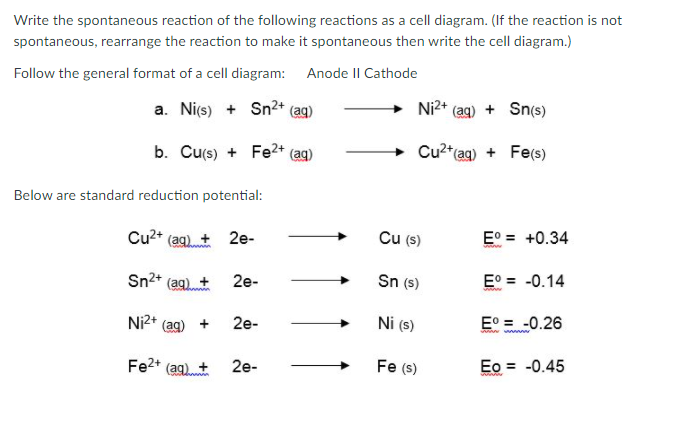
Transcribed Image Text:Write the spontaneous reaction of the following reactions as a cell diagram. (If the reaction is not
spontaneous, rearrange the reaction to make it spontaneous then write the cell diagram.)
Follow the general format of a cell diagram: Anode II Cathode
a. Nics) + Sn2+ (aq)
Ni2+ (ag) + Sn(s)
b. Cu(s) + Fe2+
Cu2"(ag) + Fe(s)
(ag)
Below are standard reduction potential:
Cu2+ (aq)+ 2e-
Cu (s)
E° = +0.34
Sn2+ (ag) +
Sn (s)
E° = -0.14
2e-
Ni2+ (ag) +
Ni (s)
E° = -0.26
2e-
Fe2* (aq) +
Fe (s)
Eo = -0.45
2e-
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning