Q: Draw a stepwise mechanism for the conversion of dihalide A and cyclohexane-1,4-diol to polyether B…
A: Since AlCl3 is a Lewis acid and hence it will extract Cl from the compound A forming a carbocation…
Q: A. Oxidation of 2° alcohol to a ketone Reagents: Jones Reagent (CrO3 in aquous H₂SO4; acetone)
A:
Q: Classify each reaction as oxidation, reduction, or neither. a.
A: “Since there are multiple questions and it is not mentioned that which one has to be solved so I am…
Q: Draw the structure of the major product of Benzene + HNO3 in H2SO4
A: Benzene undergo electrophilic substitution reaction and in the reaction, a hydrogen atom of benzene…
Q: Draw a stepwise mechanism for the conversion of heptane-2,6-dione to 3-methylcyclohex-2-enone with…
A: First, the general equation is as follows,
Q: Draw the product formed when each compound is treated with NaNO2 and HCl.
A: Primary and secondary amines on reaction with NaNO2 in the presence of HCl yield different products.…
Q: Draw a stepwise mechanism for the conversion of hex-5-en-1-ol to the cyclic ether A.
A: H2SO4- H2SO4 is sulphuric acid This is strong acid. An acid completely dissociate in aqueous…
Q: Draw a stepwise mechanism for attached reaction
A: The given reaction is represented as follows:
Q: What alcohol starting material is needed to prepare each carbonyl compound as a product of an…
A: Alcohols are oxidized to formaldehyde, ketone and carboxylic acids.
Q: What cyclic product is formed when each 1,5-dicarbonyl compound is treated with aqueous OH?
A: a). The cyclic product is given as
Q: 1. Which alkene is the major product of this dehydration? HO H2SO4 heat А В C D
A:
Q: MAKE: CEN CEN FROM: or USING ANY REAGENT WITH 42 CARBONS (SHOW STEPS)
A: GIVEN:-
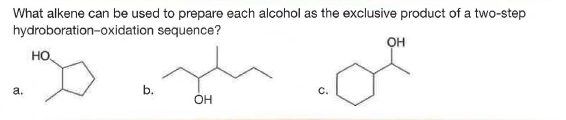
Q: What alkene can be used to prepare each alcohol as the exclusive product of a two-step…
A:
Q: Convert each compound to its enol or keto tautomer. HO- a. b. CH CH3
A: The given compounds has to be converted into keto or enol form.
Q: Draw all possible constitutional isomers formed by dehydrohalogenation of each alkyl halide.
A: a.
Q: Draw a stepwise mechanism for the dienone–phenol rearrangement, a reaction that forms…
A: A stepwise mechanism for the dienone–phenol rearrangement can be shown as follows,
Q: Draw the organic products formed when 2-bromopentan-3-one (CH3CH2COCHBrCH3) is treated with each…
A: The products formed when 2-bromopentan-3-one is treated with Li2CO3, LiBr, DMF is given as,
Q: Draw the organic products formed when each alkyne is treated with two equivalents of HBr.
A: The reaction of alkyne with HBr is markovnikov reaction In this reaction negative part of the…
Q: Draw the most stable form of the major mixed Claisen product formed in the reaction. Select Draw…
A: Claisen condensation is the reaction in which enolates are added to an ester molecule. Enolate is…
Q: Devise a synthesis of each compound from benzene. You may usealcohols with one or two carbons and…
A:
Q: What
A: First OH- attack on carbonyl carbon and then π bond transfer towards more electronegative atom…
Q: Draw a stepwise mechanism for the conversion of dihalide A and cyclohexane-1,4-diol to polyether B…
A:
Q: Label the α and β carbons in each alkyl halide. Draw all possible elimination products formed when…
A: The carbon to which the functional group is attached is called the alpha carbon. The next carbon to…
Q: Name each epoxide: CH3 a. (two ways) (two ways) CH b.
A: An epoxide is a cyclic ether consisting of one oxygen and two carbon atoms. During the IUPAC naming…
Q: Convert benzene into each compound. You may also use any inorganic reagents and organic alcohols…
A: The formation of grignard reagent is shown below.
Q: Draw the products formed when the alcohol (shown above) is dehvdrated with H,SO
A: The reaction will be E1 elimination as HSO4 - acts as base .
Q: What alkenes are formed from each alkyl halide by an E1 reaction? Use the Zaitsev rule to predict…
A: By using the Zaitsev's rule the major product can be predicted as it states that the more…
Q: Explain Addition of Alcohols—Acetal Formation ?
A: Acetals are derivatives of aldehydes or ketons They have the common formula R2C(OR')2 . The…
Q: Do ethers have a good leaving group ?
A: Ethers are the organic compounds which have the general formula R-O-R'. A group which leaves as a…
Q: Draw the major organic product formed by reaction of 2-hexyne with the following reagent: H₂O in…
A: The starting material is 2-hexyne which is represented as: In this reaction, a molecule of water is…
Q: What alkene can be used to prepare each alcohol as the exclusive product of a two-step…
A: Hydroboration oxidation is an oxidation reaction of alcohols which convert alkenes to alcohol by…
Q: Label the α and β carbons in attached alkyl halide. Draw all possible elimination products formed…
A: Biomolecular elimination (E2) reaction: E2 reaction involves a one-step mechanism in which…
Q: What product is obtained from the reaction of each of the following alcohols with a. H2CrO4? b.…
A: (a)Product obtained from the reaction of alcohol and H2CrO4: (1) The 3-pentanol is undergoes…
Q: Structure A Reagent A Reagent B
A:
Q: Label the α and β carbons in attached alkyl halide. Draw all possible elimination products formed…
A: Dehydrohalogenation Reaction: The secondary halide undergo either elimination or SN2 depending upon…
Q: Draw a stepwise mechanism for the conversion of lactone A to ester Busing HCl in ethanol. B is…
A:
Q: Which compound is hydrated more rapidly?
A: The compound which will have more stable intermediate i.e carbocation formed during the addition of…
Q: What alkene or alkyne yields each set of products after oxidative cleavage with ozone?
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: What alkene is the major product formed from attached alkyl halide in an E1reaction?
A: Elimination reaction (1) : Elimination reaction or E1 reaction a unimolecular elimination reaction…
Q: Draw all products formed by treatment of each dibromide (A and B) with one equivalent of NaNH2
A: Introduction: NaNH2 is a strong base. It is often used for deprotonation in weak acids or also for…
Q: Convert benzene into attached compound. You may also use any inorganic reagents and organic alcohols…
A: The formation of grignard reagent is shown below.
Q: Draw the structure of the most stable enol tautomer(s)
A: In organic chemistry, Ketones generally show keto-enol isomerism, which is also known as…
Q: Axial alcohols are oxidized faster than equatorial alcohols by PCC and other Cr6+ oxidants. Which OH…
A: Three-dimensional structure of the molecules show how the possible arrangements actually are: a) The…
Q: What cyclic product is formed when each 1,5-dicarbonyl compound is treated with aqueous −OH?
A:
Q: Convert benzene into each compound. You may also use any inorganic reagents and organic alcohols…
A: (a) The first step in the reaction is the halogenation of alkene in the presence of aluminum…
Q: Q3/ Label each statement as True (T) or False (F). a) PCC oxidizes 1° alcohols to ketones. b)…
A: PCC oxidizes primary alcohol to aldehyde and secondary alcohols to ketones. Therefore, the given…
Q: Label the α and β carbons in attached alkyl halide. Draw all possible elimination products formed…
A: The dehydrohalogenation takes place in accordance with the Saytzeff's rule, according to which the…
Q: Draw a stepwise mechanism for the conversion of dihalide A and cyclohexane-1,4-diol to polyether B…
A: First the Lewis acid, AlCl3 takes chlorine from dihalide compound A results to form AlCl4- and…
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

- Label the α and β carbons in attached alkyl halide. Draw all possible elimination products formed when each alkyl halide is treated with K+−OC(CH3)3.5. WHAT IS GRIGNARD REACTION? WHAT CONSTITUTES A GRIGNARD REAGENT? 6. WHAT IS KETO-ENOL TAUTOMERIZATION?Draw a stepwise mechanism for the conversion of hex-5-en-1-ol to the cyclic ether A.
- β-D-Glucose, a hemiacetal, can be converted to a mixture of acetals on treatment with CH3OH in the presence of acid. Draw a stepwise mechanism for this reaction. Explain why two acetals are formed from a single starting material.Do ethers have a good leaving group ?Label each molecule as oxidation, reduction, or neither.
- Axial alcohols are oxidized faster than equatorial alcohols by PCC and other Cr6+ oxidants. Which OH group in each compound is oxidized faster?Ethers are oxidized with O2 to form hydroperoxides that decompose violently when heated. Draw a stepwise mechanism for this reaction.Draw all products formed by treatment of each dibromide (A and B) with one equivalent of NaNH2
- Label the alkene in each attached drug as E or Z. Enclomiphene is one component of the fertility drug Clomid. Tamoxifen is an anticancer drug. Clavulanic acid is sold in combination with the antibiotic amoxicillin under the trade name AugmentinWhat other alkene is also formed along with Y in Sample Problem 9.3? What alkenes would form from X if no carbocation rearrangement occurred?

