Q: ОН OH H,SO4 H20 1. ВНз 2. NaOH, H2O2 OH OH DMP CH,Cl,
A: The above reactions can be solved easily by mechanistic pathway.
Q: The common pain relievers – aspirin (C9H8O4), acetaminophen (C8H9NO2), and naproxen sodium…
A: To solve this question, first, we need to find out the molar masses of the given chemical compounds.…
Q: What is the coefficient that should be placed in front of lead (IV) iodide (Pbl4)? * Nal + Pb(SO4)2…
A: see the balance equation in picture solution and write the answer of your question.
Q: .) Refer to the following equation: 3Cu(s) + 8HNO3(aq) → 3Cu(NO;);(s) + 2NO(g) + 4H,0(1)
A:
Q: Balancing Equations Worksheet1) ____ NaCl + ____ KOH ____ NaOH + ____ KCl2) ____ MgO + ____ Li 2…
A:
Q: Balance the following combustion reaction: (Write whole-number coefficients in each box.)…
A: The Chemical reaction follows the law of conservation of mass. According to the law of conservation…
Q: Br Method 1: 1) t-BUOK 2) H30 3) PCC 4) MeMgBr 5) H20 6) PCC Method 2: 1) NaOH 2) PCC 3) MeMgBr 4)…
A:
Q: Sort the following compounds according to the increasing ionic characteristic? Bel2, MgCl2, NH3
A: As the size of the cation increases ionic character increases
Q: H. Al,0, O Al+_0, Туре: I. N, +H2 → NH3 Туре: J. Al+ HCI → _AICI, + H, Туре: K. V,05 + _ HCI → O…
A:
Q: Calculate [H+][H+] for [OH−]=4.0×10−4 M[OH−]=4.0×10−4 M. Express your answer using two significant…
A: For an aqueous solution, we know that the production of the concentration of the hydrogen ions and…
Q: Write a balanced equation for the following by inserting the correct coefficients. If the…
A: • The given unbalanced chemical equation is, CaSiO3(s) + HF(g) → SiF4(g) +…
Q: 1. NaOH 2. Hg(OAc)2, H₂O 3. NaBH4 1. NaOH 2. BH3-THF 3. H2O2, NaOH 1. NaOtBu 2. Hg(OAc)2, H2₂O 3.…
A:
Q: In the answer box, type the number of OH e following molecule.
A: -> Asymmetric centers are centers at which stereoisomerism is possible . ->A chiral carbon is…
Q: When writing formulas for compounds, What do the parenthesis mean? Co(C2H3O2)2 I'm supposed to…
A: The parenthesis is used to represent a group of atoms present together. The example mentioned is…
Q: Below is the nutritional label for a typical commercial apple juice. Assuming a daily allowance of…
A: a) It is written that the container has 120% vitamin C of the daily requirement. And, the container…
Q: So for the question below, I have the answer worked out from class but I don't understand certain…
A: Since you have asked multiple questions, we will solve the first part of the question for you. If…
Q: Low of constant composition
A: In this question, we will see the defination of Law of constant composition and it's example. You…
Q: Organic chemistry:What does a percentage yield of 55.16 and percentage recovery of 181.30g mean .I…
A: Percentage yields and percentage recovery are two different things that are used to indicate the…
Q: An organic compound is made up of 67.7% carbon, 11.1% hydrogen and 22.2% oxygen. Circle the…
A:
Q: ) LIAIHA (excess) CN 2.) H30*, H20 pyridine a) 1.) b.) 1.) c.) 1.) H2N. 2) 2) NH2 2) 3.) 3.) 3.) d)…
A: Given reaction,
Q: OH Na2Cr207 H20, CH3CO2H Drawing
A: Na2Cr2O7 is a strong oxidising agent which oxidises 2° alcohol to ketones.
Q: C3H2NC3H2N, 208.25 g/molg/mol Express your answer as a chemical formula.
A: Given Molar mass = 208.25 g/mol Empirical formula = C3H2N To determine the chemical formula
Q: Place coefficients where needed to balance the following equation: (enter 1 if no coefficient is…
A: According to the Law of conservation of mass " all atoms of different elements must be equal on both…
Q: Which of the following represents the coefficients for the molecules (left to right) for the…
A: A balanced chemical equation is that which consists of an equal number of atoms on either side of…
Q: In one sentence explain what is wrong with these equations, and rewrite them so that the…
A: Given, 1.) 2Mg(OH)2+4HCl-> 2MgCl2+4H2O 2.) C3H6+4.5O2-> 3CO2+3H2O What is wrong in these…
Q: What is the excess reagent and its amount in grams in the active ingredient Potassium Iodid
A: Chemical reactions are those reactions which undergo any chemical change. Chemical reaction is…
Q: Balance the following and list the coefficients in order from left to right. ___ Cr + ___ H 2SO 4 →…
A:
Q: 42.Primary Standard reagent used in standardization of 0.1N sodium hydroxide a. Sulfamic acid c.…
A: A primary standard is a solution that itself do not need any calibration and is used for calibration…
Q: 7. Using the formulas provided for you in the lab introduction, fill the missing areas in the…
A: 7.) The relations used to solve are : 1.) pH + pOH = 14 2 ) pH = - log [H+] Also, [H+] = 10-pH…
Q: In the expression, 3CaBr2. O 3 is a subscript and 2 is a coefficient O 2 and 3 are subscripts O 2 is…
A: Ionic compounds are formed by the transfer of electrons from cations to anions. In the ionic…
Q: Calculate the number of atoms of hydrogen (H) in 394 cm³ of the colorless gas butane at 25 °C and…
A:
Q: Balancing Equations Worksheet 1) Na3PO4 +L KOH → NaOH + K3PO4 2) MGF2 + LizCO3 > M9CO3 + LiF 3) P4 +…
A: Since you have posted question with multiple subparts, we have authored the first ten for you.…
Q: 2KMNO4(aq) + 3H2SO4(aq) + 5(COOH)2(aq) → K2S04(ag) + 2MNSO4(aq) + 10CO2(g)+ 8H2O1) (1) 2KMnO4(aq) +…
A: Specify the oxidation state of the metal ions in the products---
Q: g(s) + Zn2"(aq) Mg2"(aq) + Zn(s)
A: In the Spontaneous reaction cell potential is positive while cell potential is negative. According…
Q: (Use the lowest possible coefficients. Be sure to specify states such as (ag) or (s). If a box is…
A:
Q: Write a balanced equation for the following by inserting the correct coefficients (if a coefficient…
A: A balanced chemical equation is equation in which the number of each element in both side are equal.
Q: Balance
A: Since you have asked multiparts, we will solve the first three subparts for you. If you want any…
Q: Br Method 1: 1) t-BUOK 2) H3O° 3) PCC 4) MeMgBr 5) H2O 6) PCC Method 2: 1) NaOH 2) PCC 3) MeMgBr 4)…
A: The reaction of alkyl bromide with NaOH gave the respective alcohol, which on further oxidation gave…
Q: Using the following equation: Fe203(s) + 3H2(g) What is the molar ratio of iron (III) oxide (Fe,03)…
A:
Q: A solution containing 15 mmole of CaSO, is diluted to 250 mL. The number of milligrams of CasO4 2H;0…
A:
Q: Ni Ni2* (0.750 M) || Cu²* (0.0500 M) | Cu EP (V)
A:
Q: If we need the information on how to dispose a certain chemical substance, we need: GHS Label NFPA…
A: If we need the information on how to dispose a certain chemical substance, we need GHS Label and…
Q: Start your trial now! First week only $4.99! Expand Transcribed Image Text Question 3 Study the…
A: The metal which is used for making pennies before 1982 is given below with explanation.
Q: How many carbon (C) atoms are present in 0.39 g of pencil lead or graphite? Group of answer choices
A:
Q: Questions 13 through 16 pertain the following medication order. PHARMACY STOCK Calcium chloride 13.6…
A: Since you have asked multiple questions ,we will solve the first question for you .If you want any…
Q: HO, HO, 1) HO но H+ LIAIH, 1) ВН 1) LIAIH(O'Bu)a -78°C -78°C 2) H20 2) LIAIH, 3) H20 2) Hо A C E
A: The question is based on the concept of organic reactions. We have to convert the reactant into…
Q: Table 2. Qualitative Analysis of Group V Cations Result/ Observation Group V Confirmatory Substance…
A: In the qualitative inorganic analysis, the cations and anions present in an inorganic salt can be…
Q: when drawing melting point composition diagram for a mixture of something such as cinnamic acid and…
A: The melting point composition diagram for mixture cinnamic acid and urea depicts the comparison…
Q: Balance the equations by inserting coefficients as needed. H_{2}S + LiOH -> Li_{2}S + H_{2}O…
A: It is required to balance the following chemical equations, which can be done as,Part AThe balancing…
Q: Determine the empirical formulas for the compounds with the following percentage compositions. 34.…
A:
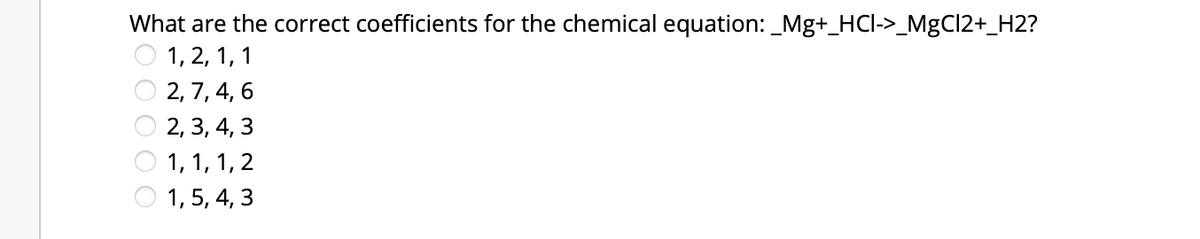
To get the coefficients for chemical reaction we have to balance the equation.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- A mixture containing only Al2O3 (FM 101.96) and Fe2O3 (FM 159.69) weighs 2.019 g. When heated under a stream of H2, the Al2O3 is unchanged, but the Fe2O3is converted to metallic Fe plus H2O(g). If the residue weighs 1.774 g, what is the weight percent of Al2O3 in the original mixture?How much iron oxide (Fe2O3) is needed to make a 500 mg fuel rich mixture of aluminum (Al) and iron oxide that has an equivalence ration of 1.3? What is the actual F/O ratio for this mixture? Please provide only typed answer solution no handwritten solution needed allowedIs 31-33 true or false
- Sample Physical Appearance 0.1 M Na2CO3 0.1 M CaCl2 Na2CO3 + CaCl2 mixture Part II filtrate + CaCl2How to manipulate formula mgh=1/2mv2 to find velocity and mass2 CoCl2(aq) + 8 NH3(aq) + H2O2(aq) + 2 NH4Cl(aq) 2 [Co(NH3)5Cl]Cl2(s) + 2 H2O(l) In a 50-mL Erlenmeyer flask, cobalt(II) chloride hexahydrate (1.075 g), an aqueous solution of ammonia (1.2 mL, 15 M), ammonium chloride (0.505 g), and hydrogen peroxide (1.0 mL, 30.0% (w/w), density = 1.10 g/mL) were combined. After completion of the reaction, [Co(NH3)5Cl]Cl2 (0.450 g) was isolated. What was the percent yield of the product?
- C2H2- 227.4 H2- 0 C2H6- (-84)Product-related impurities can be identified using SV-AUC.True/FalseWhich of the following statements is false?A. Temporary hardness is the hardness caused by calcium and magnesium carbonate and bicarbonates.B. Temporary hardness can be removed by boiling water.C. Permanent hardness is the hardness due to chloride, nitrate, sulphate, silicate and phosphates of calcium and magnesium.D. Among the hardness units, there is equation 1.25 AS = 1 IS = 1.79 FS.
- Aleks data for AgBrO3 is 5.38 x 10^-5Show your work: Balance the following chemical equation: C7H16O5 + O2 ---> CO2 + H2OHow would each of the following errors affect the determination of the molar mass of the unknown (Increase/Decrease/No effect)? a. Thermometer reads 2.0o higher than the true temperature. b. Some of the t-butanol was unknowingly spilled after it had been weighed but before the solute was added. 3. A student accidentally added acetylsalicylic acid (MW = 180.157 g/mol) rather than salicylic acid (MW = 138.121 g/mol), if no other mistakes were made, how would this error affect the determined molal freezing point depression constant for t-butyl alcohol (Kf)? 4. Based on the Tf and Kf you determined for t-butyl alcohol, predict the freezing point of a t-butyl alcohol solution containing 0.530 m NaCl. Assume 1.9 is the van’t Hoff factor for NaCl in t-butyl alcohol. Show your work.

