Q: B A D +y -IN -IM
A:
Q: Determine the following values for a saturated aqueous NaOH solution at 20 °C to the indicated numbe...
A: As per the solubility, msolute=100 gmsolvent=100 gmsolution=200 g
Q: Under what conditions is the volume of one mole of a gas equal to 22.4 L? What do you call this volu...
A: At what condition, volume of one mole of a gas = 22.4 L What do you call this volume ?
Q: Find the density of methane at 22oC and 0.961 atm.
A:
Q: The following reaction is investigated (assume an ideal gas mixture): 2N20(g) + N2H4(g) = 3N2(g) + 2...
A: Dinitrogen oxide reacts with hydrazine to form nitrogen and water. The equation for the balanced che...
Q: 2 A P The rate coefficient for this elementary reaction isk = 8.63 x 10 M' min'. The initial concent...
A:
Q: Complete the following table. Directions: Remember that spelling counts. Do NOT capitalize names. Ca...
A:
Q: What is the number of molecules in 1.55 mol O2?
A: Number of moles of O2 = 1.55 mol Number of molecules of O2 = ?
Q: 4. (a) Draw all possible stereoisomers of HOCH2CHOHCHOHCH2OH using zig zag format. Calculate how man...
A:
Q: ST3D.3- At constant temperature and pressure, what is the minimum mass [in g] of glucose needed for ...
A: Answer: Value of gibbs free energy change is always equal to non-expansion work and over here electr...
Q: ΔH
A:
Q: Give the condensed structure and IUPAC Name of the two
A: The given form of hydrocarbons are in condensed structures. In the given IUPAC name of hydrocarbon ...
Q: Suppose that you started with 5 ml of 200 mg/L Cu2+ solution and added it to 1.0g of soil. After mi...
A: (1) Here we are provided with 5 mL of 200 mg/L of Cu2+ solution which is added to 1.0 g of soil. Aft...
Q: Which of the following equilibrium constants corresponds to a situation where AG = 0? OK>1 ОК»»»1 OK...
A:
Q: 526 g of NaCl (m.m. = 58.44 g mol–1) is dissolved in pure water. How many moles (to the nearest whol...
A:
Q: I. MELTING IN LOVE 1) In a frying pan, place a small piece of chocolate, butter, and ice. Turn the s...
A: Our test was to discover what would melt first; ice , butter or chocolate. We discovered that ice m...
Q: Under certain conditions the rate of this reaction is zero order in dinitrogen monoxide with a rate ...
A: Given, 2N2O (g) ➝ 2N2(g) + O2(g) Reaction is zero order Rate constant (k) = 0.0065 M.s-1 Volume of ...
Q: (d). When the bidentate neutral ethylenediamine (en, -NH2CH2CH2NH2-) reacts with Co(lII) chloride, C...
A:
Q: Delermine the average rate of change of B from t 0s to 1= 202 s. Time (s) Concentration of A (M) 0.6...
A: Given, A 2 B Time (s) Concentra...
Q: A solution of the antibiotic, penicillin contains 300 units/mL. If it is eliminated from plasma thro...
A: First order linear process indicates that the metabolism of antibiotic follow first order rate Find...
Q: at is the significance of knowing the properties of elements (periodic table) in the food industry a...
A: There are certain elements like carbon , oxygen, hydrogen etc which are used in food industry as by ...
Q: Match the description with the corresponding example: а. HF b. NH3 1. A charged, amphoteric molecule...
A:
Q: Match the description with the corresponding example: a. HF b. NH3 1. A charged, amphoteric molecule...
A: Answer Match the description with the corresponding examples Acc...
Q: Develop a general equation for the standard heat of reaction as a function of temperature for one of...
A:
Q: Approximate the enthalpy of vaporization (in kJ mol to two decimal places) for the given data provid...
A: Given: The best fit line equation for the ln(P) v/s 1/T plot is, => y = -9327.9 x + 31.491
Q: In an ITC measurement, how much heat (J) will be released upon injection of 10 µL of a 200 µM ligand...
A: Here we have to determine to determine the amount of heat energy released in protein - ligand bindin...
Q: Ammonia Concentration = 0.17 M Ammonium Chloride Volume = 42 mL | Ammonium Chloride Concentration = ...
A: Volume of Ammonia = 68 mL Volume of Ammonium Chloride = 42 mL Total volume of buffer = Volume of Amm...
Q: The solubility of Lorazepam (a tranquilizer found in Emotival , Lorax, Wypax, and other prescription...
A: “Since you have asked multiple question, we will solve the first question for you. If youwant any sp...
Q: a. N2(g) + 3H2(g) → 2NH3(g)
A: Solution -
Q: Oxygen's solubility in water is 0.00412 /100 ml at 20 °C and 760 mm Hg Calculate the solubility of o...
A: Oxygen solubility in water = 0.00412 g/100mL Temperature = 20°C Pressure = 760mmHg Pressure, P2 = 11...
Q: The equilibrium constant for the reaction N. (g) O (g) - 2NO (g) is 1.4 x 10^3 at 2600 K. A mixture ...
A:
Q: A sample of an alkali metal that has a B.C.C unit cell is f0und t0 have a mass of 1.000 g and a volu...
A: Alkali metals react with water to produce hydrogen according to the equation M + H2O ------> MOH ...
Q: The potential of the cell Cd(s) | Cdl2, Agl(s) | Ag(s) is 0.2860 V at 25°C. Calculate the mean ionic...
A: The equations at LHE and RHE can be represented as: LHE: Cd(s)→Cd2+(aq)+2e- ...
Q: CH3 + HzO , HC-C C - OH H it
A:
Q: 10. Finish the reaction below (all potential products, and label those products.not with exact names...
A:
Q: TRUE FALSE 1. The hydroxyl group found in an organic compound is responsible for its basicity. 2. Ar...
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first 3 rows of mult...
Q: A solution containing an equimolar amount of NaCl and HCl is prepared. Assuming 1 mol of NaCl and 1 ...
A:
Q: The vapor pressure of toluene at 90 deg celcius is 0.532 atm. The molar enthalpy of vaporization of ...
A:
Q: Raising the temperature of a reaction causes which of the following? O The rate of reaction will inc...
A:
Q: What are the 4 monoclinic symmetry? give a drawing of it.
A:
Q: A general reaction written as A + 2B C + 2D is studied and yields the following data: [A]o [B]o Init...
A: To find order of reaction, write rate law with A and B. Then find out order of reaction using given ...
Q: 2. Find the oxidation number of the underline element: HNO3
A: Oxidation number of underlined element (i.e. N) in HNO3 = ?
Q: A volume of 0.25 L is acquired when you combine 0.2 g of Mg metal with HCl. Assuming the solution co...
A: Given, Volume of HCl solution = 0.25 L Mass of Mg metal = 0.2 g Temperature changes from 24.1 degree...
Q: For every given acid or base, write the equation of their reactions with water when they donate or a...
A: Given, 1. Hydrocyanic acid 2. Carbonate ion 3. Ammonium ion 4. Sulfuric acid 5. Acetic acid The rea...
Q: Give the Expanded Structure and IUPAC Name
A: The bond line notation of a molecule inbetween atoms is known as expanded notation of a molecule.
Q: What volume (in ml) of concentrated HCl (with a concentration of 10 M) do you need to prepare 10 ml ...
A:
Q: 4. ONOH
A: Here we have to write the Lewis structure and molecular geometry of central atom of the given chemic...
Q: :0-H RーC- R
A:
Q: Balance the following chemical reaction: C2H5OH + 02 --> CO2 + H20
A: A balanced chemical reaction can be defined as the reaction in which equal number of atoms of each e...
Q: For measuring concentration of a sucrose solution, a polarimeter with a 15 cm tubing sample was used...
A: Interpretation- To determine the concentration of the sucrose solution in percentage when a polarime...
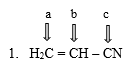
What are the hybridization states of each of the carbon atoms in the given molecule?
Step by step
Solved in 2 steps with 1 images


