Q: Problem 13.012.c Molar Mass and Gas Constant Determine the average molar mass and gas constant of…
A: Given , A gas mixture consists of O2,N2,CO2
Q: What is the molecular formula of a hydrocarbon that contains 5 carbons, one : ring, and one л bond?
A: 1 pi bond is generally represented by a double bond and the ring is represented by cyclic structure…
Q: which of the following conformation is highest in energy
A: From the given conformation of 2,3-dimethyl butane. to predict which conformed will have more energy…
Q: Draw the Lewis structure for SO₂ (with minimized formal charges) and then choose the appropriate…
A: Given -> SO2
Q: 9.Label the functional groups and identify the correct compound based on the IR spectrum. by by I %…
A: Anlyse ir spectrum.
Q: how do I find which compound is more polar? CF4 or CFCl3
A: Polar molecules are those in which there is a separation of electric charge. Polar molecules are…
Q: A constant current of 0.800A was used to electrolyze copper in an electrolytic cell, where copper…
A: Answer:- This question is answered by using the simple concept of electrolysis of a salt then mass…
Q: c) OMe From
A: The Retrosynthesis of the following compound is given below
Q: The mass percentage composition of O in Ammonium Nitrate (NH4NO3) is:
A: Mass percentage composition is the relative mass of a particular element in one mole of a compound…
Q: You have a mixture of the following compounds dissolved in diethyl ether. Which aqueous system could…
A: Acid-base extraction method is used to separate organic compounds from each other based on their…
Q: Is there commerical resin similar to RP-46 polyimide resin?
A: Ans: widely used thermoset polyimide resin comprises PMR-15 and RP-46
Q: (d) Using the reagents below, list in order (by letter, no period) those CH3 o necessary to prepare…
A: The first step of the formation of the desired product is the benzylic bromination of the toluene.
Q: How many mls of a 27.5% potassium carbonate solution, which has a density of 1.08 g/mi, would a…
A: The concentration of a solution in terms of weight % is defined as: Cwt=MsoluteMsolution×100 ... (1)…
Q: Unit conversion challenge: A local water retention pond was found to have elevated mass…
A: Here We have to change unit of volume from m3 to Liter.
Q: (g) The element whose +2 cation has the same electronic configuration as krypton. (h) The lightest…
A:
Q: How many molecules Of 0₂, are decompose reaction below? OF KC103 of when 47.79 in the 2 KClO32 KCl…
A: Given, 2KClO3 -----> 2KCl + 3O2 mass of KClO3 decomposed = 47.7 g Number of molecules of O2…
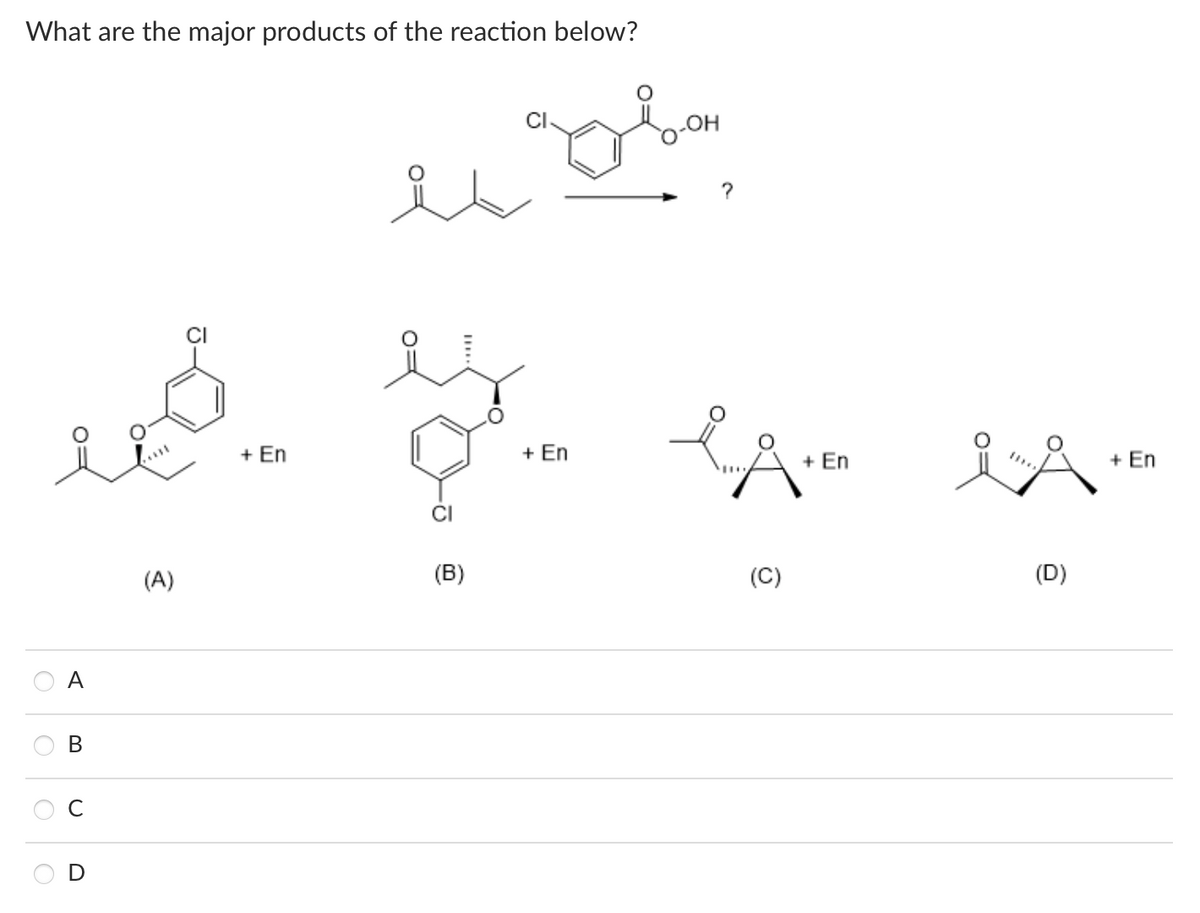
Q: What is the principal product(s) formed when 1 mol of methylmagnesium iodide reacts with 1 mol of…
A: Correct option is I
Q: At high temperature, carbon dioxide gas decomposes into carbon monoxide and oxygen gas. At…
A:
Q: In an experiment when 50 g of C3H8 is combusted, 35 g of carbon dioxide resulted. What was the…
A: The combustion reaction for C3H8 : C3H8 + 5 O2 ==> 3CO2 + 4 H2O Given Mass of propane = 50 g…
Q: 4. Energy flows into or out of the system. What two terms describe the direction of energy flow?…
A: A chemical reaction can be defined as a process in which two or more substances react with one…
Q: Which answer represents the correct balancing coefficients (in order from left to right) & the type…
A:
Q: Absorbance 0.350 0.300 0.250 0.200 0.150 0.100 0.050 Chart Title +Series 1
A: Given :- % transmittance reading of dye = 61.6% Concentration of dye = ?
Q: chemist adds 0.24 L of a 0.0327 g/L magnesium fluoride (MgF2) solution to a flask. Calculate the…
A: Concentration in g/L is equal to the weight of dissolved solute divide by volume of solution given…
Q: the data in the table, calculate the rate constant of -action. A+B → C+D Trial 1 2 3 Units [A] (M)…
A:
Q: Using the data in the table, calculate the rate constant of this reaction. k = A + B C + D Trial [A]…
A: Rate equation is represented by.... Rate = K [A]x . [B]y
Q: A student collected 66.2 mL of hydrogen gas at 22 degrees Celsius. If the pressure of "dry"…
A:
Q: a. Draw the Lewis dot structure of the following atoms/ions: H Sr Al Te c. Using the octet rule,…
A: Lewis dot structure is simplest way to represent any molecule , atom or ions in which the all the…
Q: Many drugs decompose in blood by a first-order process. Two tablets of aspirin supply 0.60g of the…
A: According to the question, The half-life of the reaction is given by = t12 = 90 min The initial…
Q: The thermal decomposition of calcium carbonate produces two by-products, calcium oxide (also known…
A: Solid Calcium carbonate undergoes thermal decomposition to give solid Calcium oxide and gaseous…
Q: HO НО, H3C- 01 C-OH C* C-C=CH CH veen the follow pair of H3C Answer
A: For the stability of chair conformation of cyclohexane. We look at the substituents, if the maximum…
Q: How many litres of water will have to be added to 1125 litres of 45% solution of acid so that the…
A: It is clearly mentioned in question that the amount of acid content in resulting mixture will be…
Q: 2) A cyanide ion selective electrode obeys the equation : E (volts) = constant - 0.05916 log [CN-]…
A: We will first calculate the numerical constant because we have to use this constant for solving the…
Q: What is the IUPAC name of this molecule? о ОН A
A: We need to start counting of the carbon chain which is eight in number in this case. Use the alkane…
Q: Using the reagents below, list in order (by letter, no period) those necessary to prepare the…
A:
Q: Which of the following relationship is correct for a nonvolatile solute? (I-III) I) The vapor…
A: Colligative properties are the properties that depends on the number of particles and not on the…
Q: b Calculate the enthalpy change for this reaction from enthalpies of formation. AH (kJ/mol)…
A: Given data: The enthalpy of formation of CH3CH(OH)CH3(g) is -272.8 kJ/mol The enthalpy of…
Q: You need to make 800 mL of 2.4 M KNO3 solution. What molarity would the potassium nitrate stock…
A:
Q: QUESTION 3 How many moles of CO 2 will be produced if we produce 8.97 grams of H 20 in the reaction…
A:
Q: 6.) Consider the reaction Pb (504) ₂ + 2 Zn → 2 ZnSoy + Pb 2 If 0,411 mol of zinc reacts with excess…
A: Given balanced equation: Pb(SO4)2 + 2Zn → 2ZnSO4+ Pb We have to find the mass of zinc sulfate…
Q: What is the approximate bond angle between 2 of the nitrogen hydrogen bonds in NH3?
A: We have been given a molecule NH3 and we have been asked the approximate bond angle between 2 of…
Q: 1. Identify the charge balance equation for a solution containing HO, OH, Ca, K, CH,OH, CrO, and Cl…
A: The charge balance equation is the equation where the sum of positive and negative charges are equal…
Q: eaction at 11.0 °C evolves 769. mmol of sulfur hexafluoride gas. culate the volume of sulfur…
A: Ideal gas equation. PV = nRT
Q: 1) Convert the following skeletal structure to a condensed structure. 2) what is the molecular…
A: Given that, a skeletal line structure of a compound 1) We have to give the condensed structure of…
Q: Using the procedure above, please annswere the below question: a. In step 1 of the experiment, you…
A: To explain the effect on the calculated molar volume, if the layer of the MgO deposited over the Mg…
Q: You dissolve a 0.553 g sample of an unknown monoprotic weak acid in water to a total volume of 20.0…
A: At equivalence point models of acid are equal to moles of base . So first we would calculate models…
Q: What is the IUPAC name of this molecule? OH A/ 신
A:
Q: A 667 mL sample of Ar gas was collected at 489 mmHg. What would be the pressure in mmHg if the gas…
A: At constant temperature, gas follows Boyle's law. According to Boyle's law pressure is inversely…
Q: Using Le Châtelier's principle, explain what happens to this system if Fe^3+ (aq) ions are added to…
A: Le Chatelier's principle states that if a dynamic equilibrium is disturbed by changing the…
Q: In an organic chemistry lab how do you know which solvents are more polar, less polar, or not polar?
A: We have been asked In an organic chemistry lab how do we know which solvents are more polar, less…
Q: The formal charge is the "charge" an element would have in a molecule or ion if all of the bonding…
A: Given that, three inequivalent lewis structures of CO2. (1) We have to determine the formal charges.…
Step by step
Solved in 2 steps with 1 images

- How do you decompose KHS2O3What substrates are needed to synthesize the conjugate addition product below? a) 1 and 3 b) 2 and 3 c) 1 and 4 d) 2 and 4One of the steps in fat metabolism is the hydration of crotonate to yield 3-hydroxybutyrate. This reaction occurs by addition of —OH to the Si face at C3, followed by protonation at C2, also from the Si face. Draw the product of the reaction, showing the stereochemistry of each step.
- Calculate the Ka's for the following acids: (a) Citric acid, pKa = 3.14 (b) Tartaric acid, pKa = 2.98Cyclohexene (an alkene) is mixed with potassium permanagnate solution (KMnO4). What do you expect to observe? A. The mixture will turn purple as the permanganate is reduced by the alkene. B. The purple colour will disappear as the permanganate is reduced by the alkene. C. Nothing. Permanganate does not react with alkenes. D. The purple colour will disappear as the permanganate is oxidised by the alkene.Draw the structure of the predominant form of CF3CH2OH (pK a = 12.4) at pH = 6.
- What are the products of the ionization of HSO3? A. H3O+ B. SO32- C. H2O D. HSO4 a. C&D b. A & B c. B&C d. A&Da) Identify H and predict the products b) Give the mechanism of the following reaction c) Give the productPredict the products of each of the following reactions. (a) MnCO3(s) + HI(aq) ⟶(b) CoO(s) + O2(g) ⟶(c) La(s) + O2(g) ⟶(d) V(s) + VCl4(s) ⟶(e) Co(s) + xsF2(g) ⟶(f) CrO3(s) + CsOH(aq) ⟶

