What are the structures of the products below? Type the letter labels of your answers. Type NONE if there is no product or if the reaction is not feasible. H 想 alcoholic AgNO, Product C CHOICES: MAN 2 mol Bra CHCL Na in Liq ammonia 学 BAN PAN Product B Ag+ Product A ^^ … VAN CAN
What are the structures of the products below? Type the letter labels of your answers. Type NONE if there is no product or if the reaction is not feasible. H 想 alcoholic AgNO, Product C CHOICES: MAN 2 mol Bra CHCL Na in Liq ammonia 学 BAN PAN Product B Ag+ Product A ^^ … VAN CAN
Chapter5: Stereochemistry At Tetrahedral Centers
Section5.SE: Something Extra
Problem 58AP: One of the steps in fat metabolism is the hydration of crotonate to yield 3-hydroxybutyrate. This...
Related questions
Question
Predict product A, B, and C
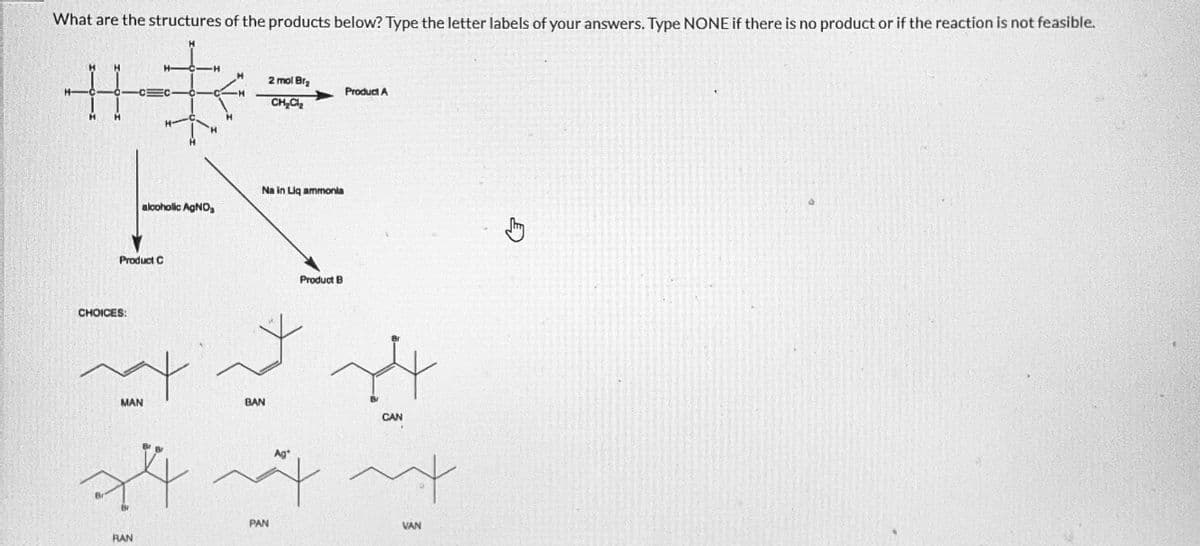
Transcribed Image Text:What are the structures of the products below? Type the letter labels of your answers. Type NONE if there is no product or if the reaction is not feasible.
1_H
H
编
O
Product C
CHOICES:
MAN
alcoholic AgNO,₂
Br
RAN
Na in Liq ammonia
BAN
2 mol Br₂
CH₂Cl₂
+44
PAN
Product B
Product A
4
CAN
m
VAN
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

