For each system listed in the first column of the table below, decide (if possible) whether the change described in the second column will increase the entropy S of the system, decrease S, or leave S unchanged. If you don't have enough information to decide, check the "not enough information" button in the last column System A few moles of carbon dioxide (CO₂) gas. A few moles of carbon dioxide (CO₂) gas. A few moles of helium (He) gas. Change The carbon dioxide is heated from 10.0 C to 48.0 °C and is also compressed from a volume of 7.0 L to a volume of 1.0 L The carbon dioxide is compressed from a volume of 9.0 L to a volume of 4.0 L while the temperature is held constant at -3.0°C. The helium is cooled from 88.0 °C to -11.0 G while the volume is held constant at 14.0 L AS AS O O ASHO O ASSO not enough information O ASCO AS-0 O ASSO not enough information ASCO O AS O As>0 not enough information
For each system listed in the first column of the table below, decide (if possible) whether the change described in the second column will increase the entropy S of the system, decrease S, or leave S unchanged. If you don't have enough information to decide, check the "not enough information" button in the last column System A few moles of carbon dioxide (CO₂) gas. A few moles of carbon dioxide (CO₂) gas. A few moles of helium (He) gas. Change The carbon dioxide is heated from 10.0 C to 48.0 °C and is also compressed from a volume of 7.0 L to a volume of 1.0 L The carbon dioxide is compressed from a volume of 9.0 L to a volume of 4.0 L while the temperature is held constant at -3.0°C. The helium is cooled from 88.0 °C to -11.0 G while the volume is held constant at 14.0 L AS AS O O ASHO O ASSO not enough information O ASCO AS-0 O ASSO not enough information ASCO O AS O As>0 not enough information
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter13: Spontaneous Processes And Thermodynamic Equilibrium
Section: Chapter Questions
Problem 20P
Related questions
Question
4
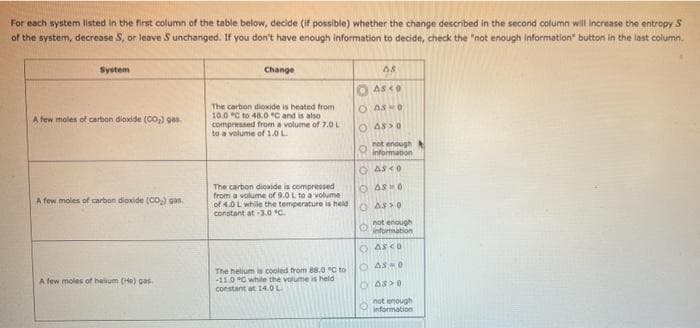
Transcribed Image Text:For
each system listed in the first column of the table below, decide (if possible) whether the change described in the second column will increase the entropy S
of the system, decrease S, or leave S unchanged. If you don't have enough information to decide, check the "not enough information" button in the last column.
System
A few moles of carbon dioxide (CO₂) gas.
A few moles of carbon dioxide (CO₂) gas.
A few moles of helium (Ho) gas.
Change
The carbon dioxide is heated from
10.0 C to 48.0 °C and is also
compressed from a volume of 7.0 L
to a volume of 1.0 L
The carbon dioxide is compressed
from a volume of 9.0 L to a volume
of 4.0 L while the temperature is held
constant at -3.0 °C.
The helium is cooled from 88.0 °C to
-11.0 G while the volume is held
constant at 14.0 L
AS
ASCO
O ASKO
O ASSO
not enough
information
ⒸASCO
OAS=0
O ASSO
not enough
O information
O ASCO
- AS-0
AS O
not enough
information
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning