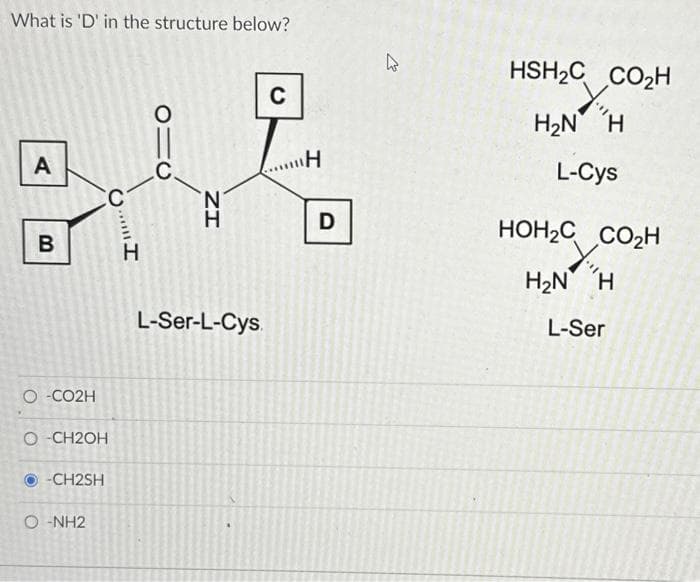
What is 'D' in the structure below? A B O-CO2H CC O-CH2OH Ⓒ-CH2SH O-NH2 Il*** H 010 C IZ H L-Ser-L-Cys. C "Η D W HSH₂C CO₂H H₂N H L-Cys HOH₂C CO₂H H₂N H L-Ser
Q: Ignoring the possibility of stereoisomers, what is the IUPAC name of the following substance?…
A: Rules for IUPAC naming:- Select the longest chain which contains carbon-carbon single bonds. Given…
Q: Draw the alkene that can be used to prepare the following alcohol as the exclusive product of a…
A:
Q: 10. A C Which of the compounds below can exist as a pair of stereoisomers (optical isomers)? Choose…
A: When any compound contain Chiral centre it exhibit optical isomerism & when a carbon is…
Q: Balance the following reaction in your notes, assuming basic conditions (use the smallest whole…
A: Recall the given reaction, MnO4- aq + C2O42- aq → CO2 g + MnO2 s…
Q: A gap junction is a channel that connects adjacent cells. What kinds of substances can pass through…
A: •Gap junctions are intercellular connections between adjacent cells. •These gap junctions permits…
Q: Classify each of the following hydrocarbons as saturated or unsaturated. Н Н- H Н Н Н H Н Н Н Н Н Н…
A:
Q: Arrange these three compounds in order of increasing ability to form intermolecular hydrogen bonds:…
A: The ability to form intermolecular hydrogen bonds depends on the presence of hydrogen bond donors…
Q: Predict the major product for the following reaction. . O IV II III IV V NaOH A || ...
A: This reaction is known as Robinson Annulation which consists of two steps - A. Michael addition B.…
Q: 1. NaNH, 2. Br 1. NaNH, 2. C HH 1. NaNH2 2. 3. NaNH, 4. Br Br
A: Sodamide reacts with terminal alkyne to produce sodium salt of alkyne which reacts with alkyl halide…
Q: Give the expected products for the following transformations. Indicate stereochemistry when…
A: Introduction The three-dimensional arrangement of atoms or groups in molecules and how this…
Q: Explain the principles and mechanisms underlying the transition metal catalysis of C-H activation…
A: Transition metal catalysis of C-H activation reactions is a rapidly evolving field in chemistry,…
Q: What is the product of the following reaction? 1) н 2) OH ОН 1. SOCl2/pyridine 2. LiAI[OC(CH3)3]H…
A:
Q: 2. H3C-CH=CH-CH3 + 3. ,CH=CH₂ Br₂ + H₂O H₂SO4
A:
Q: Draw the structure of the major organic product(s) of the reaction. OH + • You do not have to…
A: Anhydrides react with alcohols to form esters and Carboxylic acids.
Q: Be sure to answer all parts. Draw the products that form when the thioester CH3CH₂CH₂COSCoA is…
A: We will establish the products that form when the thioester CH3CH2CH2COSCoA is hydrolyzed with…
Q: Which of the following is the most acidic?(assume that Ha is the hydrogen that will be released)…
A: In a cyclic conjugated system, the hydrogen atom attached to sp3 hybridized carbon will be more…
Q: Which of the following are constitutional isomers of cyclobutane? (Select all that apply.)…
A: Given, The molecule: Cyclobutane It is a cyclic ring containing 4 carbon groups.
Q: Choose the most appropriate reagent(s) for the conversion of toluene to benzyl bromide. Br CN A NBS,…
A: N-Bromo succinimide (NBS) is the best reagent for allylic bromination. The reaction proceeds via a…
Q: Draw the product of the reaction CH3CH₂CH₂CH₂C=CCH3 + 2Br2 →? Draw the molecule by placing atoms on…
A:
Q: The reaction: H2(g) + I2(g) → 2HI(g) is a second order reaction (first order with respect to each of…
A: Given,The reaction,H2(g) + I2(g) →2 HI(g)at,H2=0.200 mol/LI2=0.200 mol/L The rate -dH2dt = 0.00097…
Q: O a. O b. O O Part long Finish Time le
A: A system with alternate single and double bond is called conjugated system.
Q: 1. Assuming that a 400. J of energy is put into 7.50 g of iron, what will be the change in…
A:
Q: 5) Which of the following compounds gives a single E2 product on reaction with sodium ethoxide,…
A: Elimination reaction of alkyl halide: Alkyl halide gives an elimination reaction by reacting with a…
Q: 3. What is the product of the following sequence of reactions? CI CI NaOH || A) I B) II C) III NaNH,…
A: Given that the following sequence of reactions:
Q: how to use the equation of the line to calculate the arrhenius pre-exponential factor a y =…
A: Equation : y=-1.3376×104 x+ 34.073
Q: Predict the product formed when the compound shown below undergoes a reaction with CH₂ONa in CH₂OH.…
A: CHO3Na is the chemical formula for sodium formate. Sodium formate is the sodium salt of formic acid…
Q: Two reactions of the same order have identical activation energies and their entropies of activation…
A:
Q: What is the order of increasing reactivity in nucleophilic addition of methanol (least to most…
A: Nucleophilic addition to carbonyl compounds depend on two factors: 1. Electronic factors: Higher…
Q: What do the nodes and branch points on a phylogenetic tree represent? a) ancestral groups that split…
A: In a phylogenetic tree, the nodes or branch points represent ancestral groups or common ancestors…
Q: 9. 8. 3 на фон CH 3 двое OH CH₂ H3C CI + 60₂ CH HẠCH3 CH₂ CH3
A: The question is based on organic reactions. we need to identify the product formed and explain…
Q: Match each particle involved in nuclear decay with the associated changes in A and Z numbers: Alpha…
A: Alpha, beta, etc. elimination can decrease or increase Z (atomic number) and A (mass number) of a…
Q: MORE PRACTICE: Balance by oxidation Number Method. 1. MnO, + SO, Mn² + SO² ... in acid. -
A: To solve this problem we have to balance the given chemical equation in acidic medium by oxidation…
Q: art B Construct the Lewis structure for H₂PO4. If necessary, expand the octet on any appropriate…
A: Lewis Structure show the arrangement of electrons and bonds around the atoms.
Q: An analytical chemist is titrating 181.5 mL of a 1.000M solution of cyanic acid (HCNO) with a…
A: To calculate the pH of the acid solution after adding the KOH solution, we need to determine the…
Q: Predict the major product for the following reaction. a) I b) II III d) IV e) V IV NH₂ OH 1. excess…
A:
Q: Four Fe²+ ions are key components of hemoglobin, the protein that transports oxygen in the blood. If…
A: In an atom three fundamental particles are present. Positively charged particle proton and neutral…
Q: In a molecule of hydrogen iodide HI (HI is used in organic and inorganic synthesis as one of the…
A: To determine the expected value of the potential energy for the hydrogen atom in a molecule of…
Q: What is the predicted product of the reaction shown? 0 000. . = = 2 IV ОН ОН ||| OCH3 NaBH4/CH3OH…
A: Sodium borohydride, NaBH4 is a reducing agent and it can selectively reduce a carbonyl functional…
Q: synthesized compound and wants to determine its molecular structure. However, due to the compound's…
A: The computational simulation approach for predicting the molecular structure and properties of…
Q: 5. An unbalanced reaction of iron (III) chloride and ammonium hydroxide is shown below. When 50.0 g…
A:
Q: Why is water such a good solvent? O a) Most polar and charged substances dissolve in it.
A: Water's exceptional ability to act as a solvent is attributed to a combination of distinct…
Q: Predict the final product for the following reaction sequence. HO. مله HO. HO 1. NaOCH2CH3 NaOCH2CH…
A:
Q: ____C3H8 + _5_O2 ➞ _3_CO2 + _4_H2O Reaction Type:______ How many moles of oxygen are required to…
A:
Q: What is the theoretical explanation for the variation in ionization energy across a period in the…
A: The ionization energy across a period in the periodic table varies due to factors such as effective…
Q: How many non-equivalent signals would you expect in the ¹H NMR spectrum of the compound shown below?…
A: 1 H NMR spectroscopy is useful to determine types of hydrogens, their signals, splitting etc hence…
Q: d Name each of the following compounds: structural formula CH₂ CH₂ CH₂ CH₂ CH₂ CH4 CH₂ CH₂ CH₂ -CH₂…
A: Detail
Q: What is the IUPAC name of the following substance? CH3 CH3CH₂CH₂C=CCCH3 CH3
A: We need to write the IUPAC name of the given compound. The given substance contains a triple bond…
Q: TEXT ANSWER You are asked to balance the following redox equation. What characteristics does this…
A: The reaction given : Cu(s) + NO3-(aq) →NO(g) + Cu2+(aq)
Q: ___Cu + ____AgNO3 ➞ ____Cu(NO3)2 Reaction Type:______ If 2.50 mol of copper and an excess of…
A: A balanced chemical equation contain equal number of atoms of each elements on both sides of the…
Q: Which is not a role proteins play in organisms? O a) structural support O b) chemical signaling Oc)…
A: Proteins play essential roles in organisms, but they do not store genetic information. Genetic…
Step by step
Solved in 3 steps with 1 images

- up an example (not appearing in this ChemActivity) of a pair of molecules that are a)constitutional isomers, b) conformers. c) configurational stereoisomers.Rank the following groups in order of decreasing priority. −CH=CH2, −CH3, −C≡CH, −HRank the following groups in order of decreasing priority. −H, −CH3, −Cl, −CH2Cl
- What is the value in kJ/mol, of the lowest energy trnasition above? a.) 545 b.) 444 c.) 320 d.) 266Consider the structure that results from the ring inversion of the chair conformation below. Add the substituents required to the given cyclohexane ring to show this structure.1. Consider the following eqm: SO2(g) + NO2(g) ⇌ NO(g) + SO3(g)What effect will adding SO3 have on the rxn?What effect will removing NO2 have on the rxn? 2. Consider the following exothermic rxn at eqm: C3H8(g) + 5O2(g) ⇌ 4H2O(l) + 3CO2(g)What effect will increasing the temperature have on the rxn?What effect will increasing the pressure have on the rxn?
- Rings + Unsaturation --- Hydrogenation If compound A C51H81BrN5O3P3 is hydrogenated to give compound B C51H101BrN5O3P3. How many rings does compound A have? Assume that P has a valency of 5. Would the answer be 4 rings? Formula -> unsat + rings = 1+C +N/2 - H/2 - X/21.What is the absolute configuration of this isomer of 2,3-dihydroxybutanal? pls explain.2. What is the absolute configuration of the enantiomer of the isomer?What is the most stable chair conformation of the 4-methoxy-1,2-dimethylcyclohexane structure given below ?
- Rank the following groups in order of decreasing priority. a.−F, −NH2, −CH3, −OH b.−CH3, −CH2CH3, −CH2CH2CH3, −(CH2)3CH3 c.−NH2, −CH2NH2, −CH3, −CH2NHCH3 d.−COOH, −CH2OH, −H, −CHO e.−Cl, −CH3, −SH, −OH f.−C≡CH, −CH(CH3)2, −CH2CH3, −CH=CH2Name the following and include any E/Z stereochem in the name .Arrange each set in order of decreasing priority using Cahn-Ingold-Prelog priority rules (1 for highest priority). d) _____-Cl, _____-OH, _____-H, _____-NH2 e) _____–CO2CH3, _____-CO2H, _____-OH, _____-OCH3

