what is the amount of θ phase
Welding: Principles and Applications (MindTap Course List)
8th Edition
ISBN:9781305494695
Author:Larry Jeffus
Publisher:Larry Jeffus
Chapter26: Welding Metallurgy
Section: Chapter Questions
Problem 12R: What is a eutectic composition?
Related questions
Question
Using the Al−Cu phase diagram in (b), what is the amount of θ phase (in %) that would precipitate at the grain boundaries in the equilibrium microstructure after slow cooling shown in (a)?
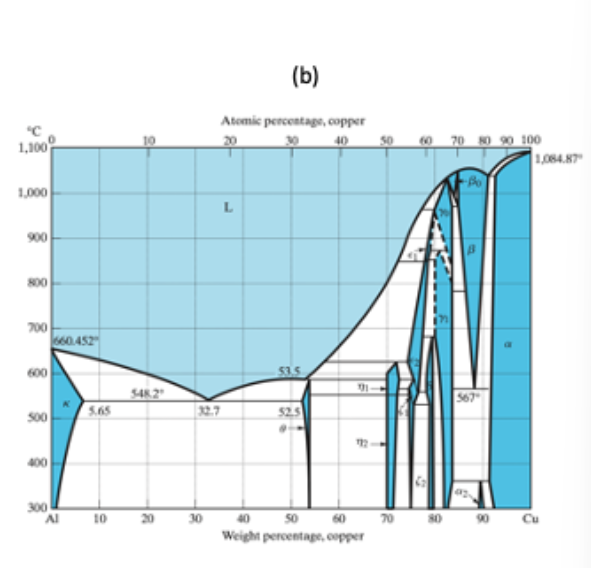
Transcribed Image Text:(b)
Atomic percentage, copper
0 20 80 90 100
1,084.87
1,100
20
40
s0
1,000
L
900
800
700
660.452
600
53.5
548.2
567
s0
5.65
32.7
525
400
300
Al
40 50 60
Weight percentage, copper
10
20
30
70
80 90
Cu
![(a)
700
100%
(95.5% Al, 4.5% Cu)
600
500
400
Page 347
Slow cool
300
200
e precipitates at
]« grain boundaries
100
90 95 100
Time
wt % AI](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F1f969d67-e111-4e40-b575-7702662e30f7%2F38c7ee5e-c9ec-4b19-a9ae-a25cae5012d4%2F57nzypc_processed.png&w=3840&q=75)
Transcribed Image Text:(a)
700
100%
(95.5% Al, 4.5% Cu)
600
500
400
Page 347
Slow cool
300
200
e precipitates at
]« grain boundaries
100
90 95 100
Time
wt % AI
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

Welding: Principles and Applications (MindTap Cou…
Mechanical Engineering
ISBN:
9781305494695
Author:
Larry Jeffus
Publisher:
Cengage Learning

Welding: Principles and Applications (MindTap Cou…
Mechanical Engineering
ISBN:
9781305494695
Author:
Larry Jeffus
Publisher:
Cengage Learning