Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter10: Fuels, Organic Chemicals, And Polymers
Section: Chapter Questions
Problem 119QRT
Related questions
Question
What is the balanced chemical equation of Calcium Carbonate above?
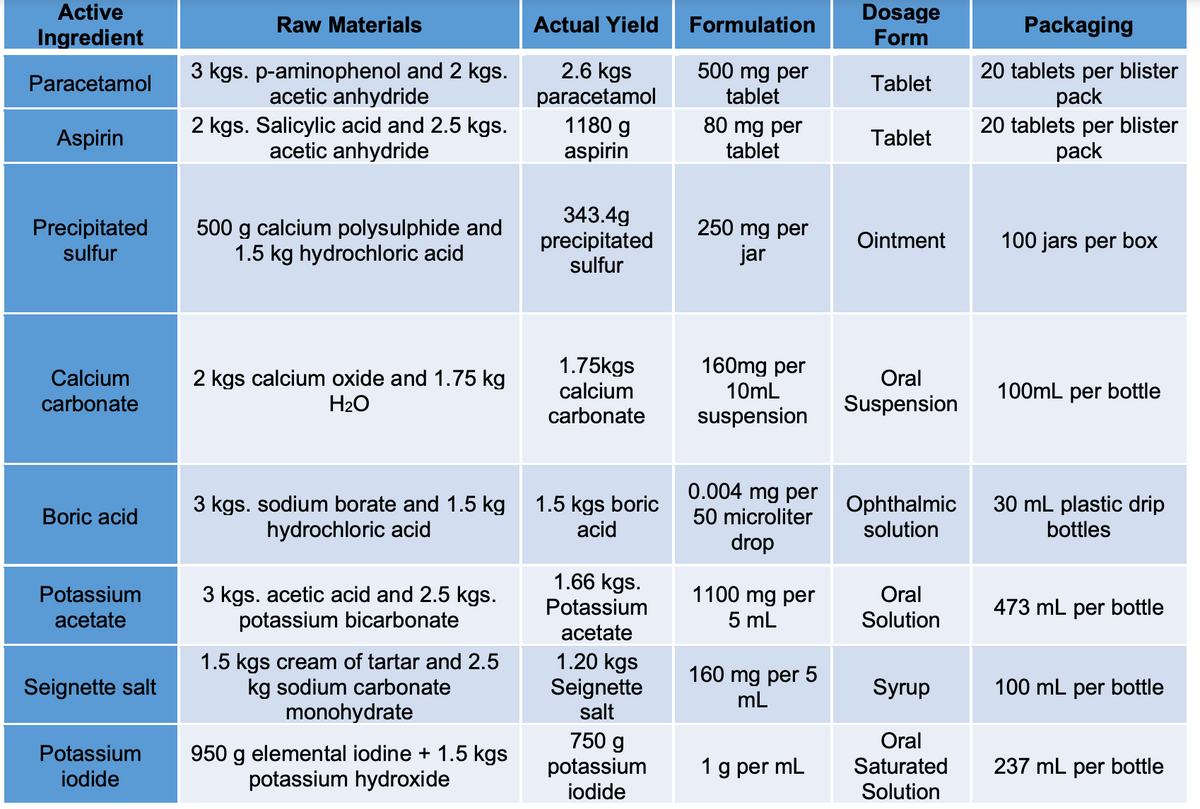
Transcribed Image Text:Active
Dosage
Form
Raw Materials
Actual Yield
Formulation
Packaging
Ingredient
500 mg per
3 kgs. p-aminophenol and 2 kgs.
acetic anhydride
2 kgs. Salicylic acid and 2.5 kgs.
acetic anhydride
2.6 kgs
paracetamol
1180 g
20 tablets per blister
pack
20 tablets per blister
рack
Paracetamol
Tablet
tablet
Aspirin
80 mg per
tablet
Tablet
aspirin
Precipitated
sulfur
500 g calcium polysulphide and
1.5 kg hydrochloric acid
343.4g
precipitated
sulfur
250 mg per
jar
Ointment
100 jars per box
Calcium
carbonate
1.75kgs
calcium
carbonate
160mg per
10mL
Oral
2 kgs calcium oxide and 1.75 kg
H2O
100mL per bottle
Suspension
suspension
0.004 mg per
1.5 kgs boric
acid
Ophthalmic
solution
30 mL plastic drip
3 kgs. sodium borate and 1.5 kg
hydrochloric acid
Boric acid
50 microliter
bottles
drop
1.66 kgs.
Potassium
1100 mg per
5 mL
Potassium
Oral
3 kgs. acetic acid and 2.5 kgs.
potassium bicarbonate
473 mL per bottle
acetate
Solution
acetate
1.5 kgs cream of tartar and 2.5
kg sodium carbonate
monohydrate
1.20 kgs
Seignette
salt
160 mg per 5
mL
Seignette salt
Syrup
100 mL per bottle
Potassium
jodide
950 g elemental iodine + 1.5 kgs
potassium hydroxide
750 g
potassium
iodide
Oral
Saturated
1 g per mL
237 mL per bottle
Solution
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry In Focus
Chemistry
ISBN:
9781305084476
Author:
Tro, Nivaldo J., Neu, Don.
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry In Focus
Chemistry
ISBN:
9781305084476
Author:
Tro, Nivaldo J., Neu, Don.
Publisher:
Cengage Learning