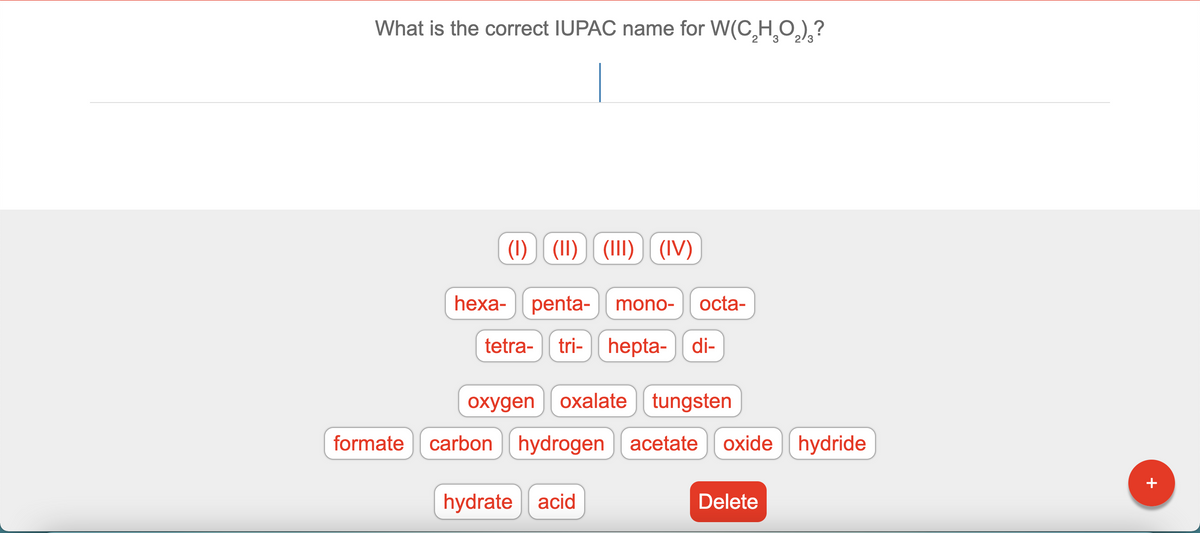
What is the correct IUPAC name for W(C,H¸O,),? (1) (11) (1II) ) (IV) hexa- penta- mono- octa- tetra- tri- ) hepta- di- oxygen oxalate tungsten formate carbon hydrogen acetate oxide hydride hydrate acid Delete
Q: CN Vs Vs CN CI OH Vs CI 'HO, Vs ОН ОН OH
A: Solution We know that Identify the relationship between each pair of…
Q: CH3CH2SH is more reactive than CH3CH20H towards sodium. A True False
A: Sodium replaces a hydrogen atom from the compound and gives an acid-base reaction.
Q: 3. How many molecules of ethylene diamine will react with Ni(H,O),"? Explain your answer.
A: We have find out how many molecules of ethylene diamine will react with Ni(H₂O). Note -As per our…
Q: In an essay provide written explanation, hossa In the sunthesis of Dibenzalacetone why must there be…
A:
Q: MATCHING TYPE: In column A are the artificial flavor/odor produced from the reaction between…
A: Esters are responsible for the The sweetness of the compounds.
Q: In column A are the artificial flavor/odor produced from the reaction between alcohols in column B…
A: For 7 : esters with smell of flowers is methyl pentanoate and is prepared by esterification of…
Q: Compound X has the formula C30H50BrCIO2. (There are no triple bonds in X.) It reacts with excess H2…
A: Double Bond Equivalent (DBE). One DBE = one ring or one double bond. DBE can be calculated using the…
Q: Calculate straln energy for the conformer plctured below, using straln energy Increments from the…
A:
Q: Pure acetic acid is a viscous liquid, with high melting and boiling points (16.7 °C and 118 °C)…
A: Melting point of a substance is the temperature at which the substance changes from solid to liquid…
Q: A small quantity of an unknown organic compound 'X' was burnt in a flame. The comp. burnt with a…
A: The correct option with explanation is given below:
Q: Anant Gyan Date Page below. over comp0unds A annd Bane arawn which one has ahigher boiding point end…
A:
Q: The next set of questions are assertion type: There are two statements in a sentence seperated by…
A: The given statement is Pyridine is more basic than piperidine BECAUSE pyridine is resonance…
Q: -C=C H H₂ (excess) Pd/C HgSO4, H₂O H₂SO4 i. BH3/THF ii. NaOH, H₂O₂, H₂O
A: Answer : A) The terminal alkynes produce a ketone under these conditions.
Q: В
A: The basic rules of IUPAC naming are- 1. Find the longest C chain containing the functional group. 2.…
Q: The pk, of phenol (C,H5OH) is 10.0. When a methyl (CH3) group is attached to the ring, the pK,…
A: (a)
Q: 100 43 80 60 27 29 28 40 41 42 39 15 58 10 20 30 40 50 60 Mass/charge (mle) Relative abundance (%)…
A: Given: Mass spectrum To find: Which compound will show this mass spectrum . Solution: Mass…
Q: reaction the 1- butene with one molecule chlorine by using water as catalyst result
A: We have to tell which option is correct from given choices of options
Q: What is the value of n in Hückel's rule for the following compound? IZ
A: Please find your solution below : In Huckel's rule, n represents the number of pi-electrons present…
Q: F
A: Select the longest principal carbon chain And numbering should be given from that side where…
Q: Pentaborane BgHgls) burns vigorously in O2 to give B203s) and H2O0. Calculate AHren for the…
A: Moles of B5H9 = 5 mole
Q: NH2 0
A: In the given compound 1) The first priority is to a ketone functional group 2) The second priority…
Q: How many pi electrons does this moleate have that are delocalized in closed yclic array ? :o:
A: Aromatic : (4n+2) pi electrons, conjugated, cyclic. Therefore the compound is having 10 pi…
Q: The [a] of pure quinine, an antimalarial drug, is -165. question d and e
A: In chemistry, an enantiomer is also one of two stereoisomers that are non-superposable mirror copies…
Q: How many H-H eclipsing were planar.assume energy cost of 4.0 interaction would be present f…
A: a) If cyclopentane was planar, there would be 10 H-H eclipsing interactions.
Q: (b) For the step in which 4 is transformed into 5, identify the nucleo- philic center and the…
A: Answer (b): Oxygen of carbonyl (C=O) is electronegative atom, it will pull the electron from…
Q: While (S)-ibuprofen is the active ingredient, commercial ibuprofen is sold as a racemic drug because…
A: The research's lab product contains 94 % enantiomeric excess of the S product. Therefore, the…
Q: Predict theλmax of the following compound:
A: It is given a molecule hex-1-en-3-one and its λmax value needs to be determined using UV-visible…
Q: 며. b. it C.
A: For the IUPAC nomenclature of alkene we follow certain rules ; Select the parent chain having long…
Q: b. of butane-2,3-diamine of which the semi-developed formula is: NH2 13C-CH-CH-CH3 NH2 in…
A: In hashed-wedged projection, there is the representation of bonds in the form of a backline called a…
Q: b. Calculate the max of the given compounds with the help of Woodward's Fiesher rule.
A:
Q: 11:33 AM Sat Feb 5 12% Question 6 of 7 Submit In this sixth exercise, you will learn how to add a…
A: First Try making the inner section or say cyclo pentane in that. Then go for the Different branching…
Q: Compound X has the formula C30H50BrCIO2. (There are no triple bonds in X.) It reacts with excess H2…
A: The answer is as follows:
Q: olecular formula C4H10. mula with lines to represer
A: In structural formula of C4H10 As it is Alkane(Butane). So. We use two isomers of butane.
Q: In the structure of 2,3,3-trimethylpentane, identify the primary (p), secondary (s), tertiary(t),…
A: Given compound is alkane as it contain single bonds between carbon atoms. Alkanes are organic…
Q: What is a glysodic bond 1,4 mean and 1,6
A: In oligosaccharides and polysaccharides the monosaccharide units are joined by an -O- linkage it is…
Q: Which group is of lower priority than –CH=CH2 according to Cahn-Ingold-Prelog rules. –CH=C(CH3)2…
A:
Q: Hydrocarbon A possesses a significant dipole, even though it iscomposed of only C—C and C—H bonds.…
A: The dipolar structure is
Q: он HO
A: Given: To find: water solubility highest Melting point Van der Waal interaction dipole-dipole…
Q: Complain it ketule's model Suffarts blowing properties of benvine's (1) The shape and brand angles…
A: Here we have to explain the shape ,bond angle and bond length of benzene by using kekule structures…
Q: Answer the follow ing problems in the space provided and show all work for partial credit. 1.…
A: IUPAC Nomenclature: Count the number of carbons in the ring compound. 1 position is given to the…
Q: 玩,唱 2 / 2 85% Q3. Provide IUPAC name for each of the following compounds a b Br Me с. d.
A:
Q: Using differences in C-H bond dissociation energies, calculate approximate selectivities secondary…
A: Primary C-H bond dissociation = 412 kJ/mol Secondary C-H bond dissociation = 396 kJ/mol Bond…
Q: Explain why alkene A is more stable than alkene B, even though Bcontains more carbon atoms bonded to…
A: Given compounds,
Q: CH-CH-CHCH, -2 -1
A: According to IUPAC nomenclature system, 1. Select the longest no. Of Carbon chain in the structure…
Q: Write a three-dimensional (wedge-dashed wedge-line) formula for each compound given below.…
A: The given model (ball snd stick) model can be converted into a three-dimensional formula by…
Q: Pyrethrin II and pyrethrosin are two natural products isolated from plants of the chrysanthemum…
A: The given compound is, The molecular formula of pyrethrin II is C22H28O5.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Compound K, L and M are three isomers with the molecular formula C5H10O.Compound K cannot be oxidized, while compound M and L can be oxidized.Oxidation of compound L with hot acidified potassium permanganate,KMnO4 solution yields 2-methylbutanoic acid. When treated with Iodoformreagent, yellow precipitation only occurs in compound K. Fehling test onlyyielded positive results for compound L and negative for compound M andK. Compound M is then reacted with hydrogen chloride, HCl produceschlorocyclopentane (i) Draw the structural formula of compounds K, L and M.Compound K, L and M are three isomers with the molecular formula C5H10O.Compound K cannot be oxidized, while compound M and L can be oxidized.Oxidation of compound L with hot acidified potassium permanganate,KMnO4 solution yields 2-methylbutanoic acid. When treated with Iodoformreagent, yellow precipitation only occurs in compound K. Fehling test onlyyielded positive results for compound L and negative for compound M andK. Compound M is then reacted with hydrogen chloride, HCl produceschlorocyclopentane. (ii) Name the type of chemical reaction of compound M when reacted withhydrogen chloride, HCl .An unkown solvent has a formula of C4H8O and molecular weight of 72.11 g/mol. Determine the ff: a.) index of hydrohgen deficiency b.) all possible functional group present base on IHD
- Which compound would be more basic or weaker, a compound with a pka of -3.5 or a compound with a pja of -1.7Compound X is insoluble in cold KMnO4, Br2 in CCl4, and conc. H2SO4. Compound X is most likely: a. an alkane b. none of these c. an alkene d. an alcohol e. an alkyl halide Indicate which of the ff. statements regarding nucleophilicity is incorrect. F- is more nucleophilic, hence, more reactive towards methyl iodide than Cl-. Second row elements are more nucleophilic than first row elements of comparable basicity. The rate of SN2 reaction may be markedly affected by the nucleophilicity of the attacking atom. Nucleophilicity is the affinity of a nucleophile to an electrophilic carbon Which of the following alkynes can be deprotonated by NaNH2 in liquid NH3? 3-Methylhex-2-yne Pent-2-yne 3-Methylbutyne none of these Hex-3-yne(i) Arrange the following compounds in an increasing order of basic strength :C6H5NH2, C6H5N(CH3)2, (C2H5)2NH and CH3NH2(ii) Arrange the following compounds in a decreasing order of pKb values :C2H5NH2, C6H5NHCH3, (C2H5)2NH and C6H5NH
- Compounds Mass 3-nitrophthalic acid used 200 mg = 0.2g 8% aqueous hydrazine used 0.4mL 3-nitrophthalhydrazide obtained 130 mg = 0.13 g sodium hydrosulfite dihydrate 0.6 g luminol obtained 70 mg = 0.07 g compute yield for nitrophthalhydrazide in the first step (assume nitrophthalic acid is limiting reagent) compute yield for luminol in the second step (using nitrophthalhydrazide as limiting reagent) compute yield for the overall reactionWhat is a glysodic bond 1,4 mean and 1,6Compound 1 is an anticoagulant that is extracted from, among other plants, the sweetclover plant, Melilotus Officinalis. However, the compound is moderately toxic and alsofinds use as a rodenticide. Name this compound
- In the following cases rearrange the compounds as directed : (Delhi 2010)(i) In an increasing order of basic strength :C6H5NH2, C6H5 N(CH3)2, (C2H5)2NH and CH3NH2(ii) In a decreasing order of basic strength :Aniline, p-nitroaniline and p-toluidine(iii) In an increasing order of pKb values :C2H5NH2, C6H5 NHCH3, (C2H5)2NH and C6H5NH2Experimental observations show that thiocyanate (SCN- ), the conjugate base of thiocyanic acid (pKa = 1.1 at 25 °C), is quite soluble in neutral water at pH > 2. In the space provided below, explain this experimental observation. That is, why does SCN– readily dissolve in water when the pH > 2?The ionization of monoprotic organic acid with a Ka of 6.7 × 10 -4 is 3.5%.Calculate the molecular weight of this monoprotic organic acid if 100 g of it is dissolved in 1 L of water. Hence, suggest and draw ONE (1) possible chemical structure of monoprotic organic acid having the calculated molecular weight.

