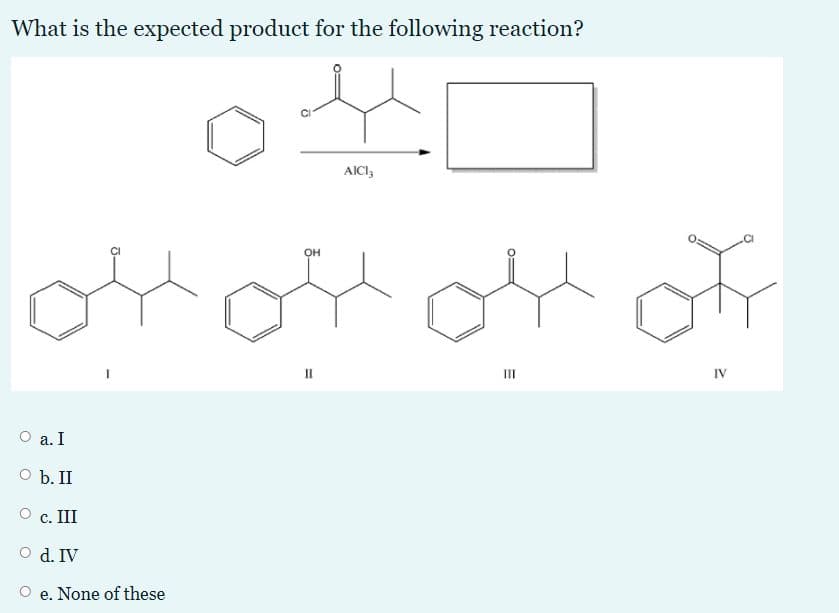
Q: Give the major product(s) of the following reaction. KMN04, H* heat HO, CN OH 8. There is no…
A: The reagent KMnO4 , H+ in presence of heat acts as a strong oxidising agent.
Q: Predict the products: K2S (aq) + BaCl2 (aq) O KC12 (aq) + BaS (aq) O KCI (aq) + BaS (aq) O K2CI (aq)…
A:
Q: elect the major product(s) of the following reactions. OH 1. CrO,/ H* 2. SOCI, OH II III IV D IV II
A: ->CrO3/H+ is oxidizing agent which can oxidize primary alcohol to carboxylic acid. ->SOCl2…
Q: Predict the product(s) of the reaction below: AGNO, + Na(OH) :: (OH)Ag : Ag(OH) : Na(NO3) : (NO3)Na
A: The given reaction is between silver nitrate and sodium hydroxide.
Q: Consider the following reaction: 1. LIAIH4/H+ 2. H2/Pt 3. CrОЗ/Н+ OCH3 Which of the following is the…
A: LiAlH4 is a strong reducing reagent. It reduces ketone to secondary alcohol and ester to primary…
Q: Identify the most likely product produced from this series of reactions. ONa H,O/H Heat HO. HO A D.…
A:
Q: Which is the expected major product under thermodynamic control? do HBr ? Br Br Br Br A B. C OA OB
A: In this First Question, we will Identify the major thermodynamic Product. You can see details…
Q: (g) 1) OsO4 2) NaHSO3
A: Alkenes can be converted to diols by treating with OsO4 and KMnO4 . Both reagents gives cis-diols.…
Q: What are the predicted products for the reaction shown? N' H3O* II
A: Let us discuss both the reactions.
Q: For the following Sn1 reaction what are the final product(s). Br CH;OH OCH, HO- 3. O 2 0 1 O 1 and 2
A:
Q: Predict the products of the following chemical reaction: note the reaction is not balanced. Mg) +…
A:
Q: 27. What is the major organic product of the following reaction? Ag,0, NH,OH он HO, но. но. но. II…
A:
Q: What would be the product(s) of following reaction? 3 FeO + 2 AIC13 → ? 3FECI, + Al,03 FeCl3 + AlO O…
A: Following is the product of the given chemical reaction.
Q: For which one of the following reactions is the value of ΔH°rxn EQUAL to ΔH°f for the product? (a)…
A: Enthalpy of formation It is the the enthalpy of formation of one mole of compound in its standard…
Q: Prove the product of the reaction in the given image. 1. m-CPBA 2. Cold-Dilute KMnO4 3. a. O3; b.…
A:
Q: Predict the products for the following sets of reactants: a. Ba(OH), (aq) + HC,H;O, (aq) → b.…
A: We have given that Predict the product of the following set of reactant: a. Ba(OH)2(aq) +…
Q: What is the expected product of this reaction? NaOCH, CH O`Na* H,CO H,C O`Na *Na'o OCH, H,CO
A: Given reaction is an example of esterification reaction where ester reacts with sodium methoxide to…
Q: Choose the correct answer: Predict the product for the following reaction. 1. NaH, 25°C 2. CH,CH,Br…
A: Base can abstract most acidic hydrogen from molecules.then there form nucleophile which give…
Q: B. Predict the products formed based from the lesson, Types of Chemical Reactions. 1. KCI 2. Mg 3.…
A: Note: We'll answer the first question since the exact one wasn't specified. Please submit a new…
Q: Predict the products (if any) that will be formed by the reaction below. If no reaction occurs,…
A: Complete the given reaction--- FeSO4(aq) + Zn(s) -----> ?
Q: Predict the products (if any) that will be formed by the reaction below. If no reaction occurs,…
A: This is a displacement reaction in which a more reactive metal displaces a less reactive metal. Zinc…
Q: 120 loo 60 40 20 Reaction progiess whet is the activadion energa for this reaction kj lmol? in 132…
A: - Activation energy is the energy which is inversely proportional to rate of reaction .If activation…
Q: Write the main products of the following reactions: g) KI H;PO4 peroxide h) HBr
A: alkene in presence of H3PO4, KI form alkyl iodide through the formation of carbocation intermediate.…
Q: Predict the product of the following Heck reactions. Part C Br Pd(PPh,)4 Et,N Draw the molecule on…
A: This reaction in cross coupling reaction called Heck reaction. In this reaction, HBr removed from…
Q: LAH (4) Pyidne a LANOR (e)
A: Organic reactions are carried out with help of reagents and thermodynamic parameters like…
Q: What are the products of the following reaction?
A: When cyanide undergoes through catalytic hydrogenation,it results into the formation of primary…
Q: What is the product of the following reaction? KMNO, ОН (сold) он OH HO II IV II O II O IV
A: Oxidation means loss of electron or the gain of Oxygen.
Q: Predict the products (if any) that will be formed by the reaction below. If no reaction occurs,…
A: Recall the reaction--> Zn(s) + Au(NO3)2(aq) ---> ?
Q: Select the productts) that are most likely to be produced in the following reaction. HO,S ŠOH HOS D…
A: The reaction taking place is given as,
Q: What is the predicted product of the reaction shown? 1. O3 2. H20 OH HO ОН II II H. IV V
A:
Q: 6.) which of the following will have a product? Feo + Ni²". (aq) Au + Al³ , (aq) Nao + Zn" (aq) Al…
A:
Q: 35. The major product formed when H,C is oxidized with K.Cr.O, Br (A) (C) H,C OH Br (B) (D) No…
A: We will answer the first one since the exact one wasn't specified. Please submit a new question…
Q: Predict the products for the following reactions CN 1) Et CuLi 2) H3O* 1) КОН 2) 3) H,O* OH OEt
A: Gilman reagent adds to the α, β-unsaturated carbon. Active methylene compounds also adds to the α,…
Q: Predict the product/s of the following reactions AND identify the type of reaction taking place.…
A: The various types of reactions are:- Combination Reaction Displacement Reaction Decomposition…
Q: What is (are) the product(s) of the following reaction? Br A в O A. A and B В. В and C C. C and D O…
A: Mechanism of E2 reaction.
Q: Use the curved arrows to predict the product(s) of the following reaction. Draw the product(s) in…
A: As Nitrogen is more electronegative element,having +ve charge very unstable. That's why arrow is…
Q: Which of the following is one of the products formed in the reaction below? IV OA V O B. II OC. I O…
A:
Q: Predict the products (f any) that will be formed by the reaction below. If no reaction occurs, write…
A:
Q: Br Mg Ether 1. LDA 2 1 'Br MgBr Br 0:00 i. Product 2 was never obtained from the reaction shown…
A:
Q: _9. What is the product of the following reaction? 1.0, 2. H,O, OH OH HO II III IV V d. IV а. I b. П…
A:
Q: Predict the product(s) for the following reaction. HBr H. он Br Br Bi II Br IV O a 1 O b.|| OC. II O…
A: This question is checking the basic concept of protonation and nucleophilic attack on epoxide. So,…
Q: Pd(PPH3)4 CI + -B(OH)2 КОН
A: To know deep about this reaction please read suzuki cross coupling and then try to solve. I have…
Q: the products shown below is the most abundant in the mixture? a. b. c. b. O O
A: Electrophilic aromatic substitution reaction
Q: Choose the correct answer: Predict the product for the following reaction. OH Na,Cr,07 H2SO4, H2O CI…
A: Option C will be correct.
Q: Which of the compounds below is/are possible product(s) for the following reactions? Br CH;CH;OH IV…
A:
Q: 1) H,0* 2) CH,COCI, pyridine (d) OMe
A:
Q: 6. Identify compounds X and Y from the options depicted below? LIAIH, EL,O then workup NABH, EIOH Y…
A: Since you have posted multiple questions, we are entitled to answer the first only.
Q: H20 H;0*
A:
Q: Provide the organic product(s) of the following reaction. .CI 1. Mg, ether 2. CO2 3. HС, Н20
A:
Q: OH a) HO. CI b) OH c) CI H d)
A: From given For each reaction given its reaction type is mentioned along with explanation as follows
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

- Halogenation is typically done with symmetric dihalides (Cl2 or Br2 ), but mixed dihalogens can also be used. Predict the product of the following reaction and explain why a single product results.Predict the product/s of the following reactions AND identify the type of reaction taking place. CO2(g) + H2O(l) MgO(s) + SO3(g) CuSO4(aq) + Zn(s) AgNO3(aq) + NaCl(aq) Zn(s) + N2(g)For the reaction 5Ce4+ + Mn 2+ + 4H2 O → 5Ce3+ + MnO4- + 8H+ , given E0 (Ce4+ /Ce3+ ) = 1.70V,E0 (MnO4- /Mn2+ ) = 1.507V, calculate ΔG0 for this reaction ?
- Which are optimum conditions for an SN2 reaction? I, II I,IV III, IV II, IIIThe Palladium complex shown in the reaction scheme below will catalyze the addition of SiH4 to an alkene. Identify the reaction types. A. B. C. .D. E.?What is the role of Na2CO3(aq) in the conversion of D to E? How would you test for the presence of compound H? Which other reducing agent could be used in place of Iron in the conversion of B to C?
- Which group elements show maximum catalytic activity for hydrogenationreactions?For the powder with composition 9wt.% Al, 3wt.% Ni and 88wt.% Mg Calculate the possible Gibb's Free energy (which reactions are most favorable) in a closed system of 610 °CGiven A, B, and C… [A] U(VI) as uraninite; UO2 (where Fe2+= reductant; Fe(OH)3 ferrihydrite= product): 2Fe2+ + UO22+ +3H2O + H+ ---- > 2Fe(OH)3 + U4+ +2H2O [B] U(VI) as uraninite; UO2 (where Mn2+= reductant; MnO2 pyrolusite= product): 2H2O + UO22+ + Mn2+ ---- > UO2 + B - MnO2 + 4H+ [C] U(VI) as as uraninite; UO2 (where HS-= reductant; S0= product): UO22+ + Hs- ---- > UO2 + S + H+ QUESTION: Use thermodynamic calculations [use redox potential (Eh)] to predict which of the three reductants below is most favorable at pH 3 ? (1) Fe2+ (2) Mn2+ (3) HS-

