Q: Is Empirical or Molecular. W
A:
Q: What is the pH (answer in 2 decimals) of a solution containing 0.25 M sodium formate (HCOONa) and 0....
A:
Q: Complete the data sheet with relevant solutions in solving for the amounts needed in the experiment
A: Here we have to keep following points in mind 1. We are making homogeneous solution, in homogeneous...
Q: For weak electrolytes, such as acetic acid, at very low concentrations the molar conductivity drops ...
A: Weak electrolytes are the electrolytes that are not completely dissociated into water. Strong electr...
Q: A 9.50 % by mass solution of acetone (C3H6O) in water has a density of 0.9849 g/mL at 20°C. What is ...
A: Given, Percent by mass of acetone = 9.50 % Density of the solution = 0.9849 g/mL Molarity of the sol...
Q: How did you determine the values of pka1 and pka2 from the titration curve?
A: For the titration of a polyprotic acid with a base, we get more than one equivalence points. We can ...
Q: PROCEDURE 1000 mg L-asocrbic acid tablet (pulverized into fine powder) Molar mass of tablet = 176....
A:
Q: Define and explain the difference between molecular weight and atomic weight. With examples. 5. Cal...
A: Atomic mass is the mass of an atom.
Q: few hydrogen and oxygen molecules are introduced into a container in the quantities depicted in the ...
A:
Q: The reaction of hydroxide ion with chloromethane to yield methanol and chloride ion is an example of...
A:
Q: The vapor pressure of pure CCI4 liquid is 504 mm Hg at 65°C. Now we prepare several 12/cCl4 solution...
A: The correct option is option B.
Q: Which of the following is consistent with a spontaneous process in the forward direction? a.ΔSunive...
A:
Q: A student dissolves 5.0g NaOH in 100mL of water in a calorimeter at 20.5C as the NaOH dissolves, the...
A:
Q: Arrange the following in order of decreasing acidity, i.e., strongest acid first. HF (KA = 6.8 x 10*...
A: Greater is theKA value , more will be the acidity of the species. So, let's arrange in the decreasi...
Q: In the gas phase reaction 2 A(g) + B(g) = 3 C (g) + 2 D (g), it was found that when 1.00 mol A, 2.00...
A:
Q: Using Brønsted-Lowry definitions, designate each of the following as a strong base (SB), a weak base...
A:
Q: Please explain why this is non aromatic with 5 pi electrons Me, B.
A:
Q: na polarographic analysis in which each compound is reduced at an electrode urface. A mixture contai...
A: Solution - According to the question - Given - The equation for response factor is = ADDT/(DDT)/Ac /...
Q: The addition of water to ethylene to yield ethanol has the following thermodynamic parameters: JH= -...
A: Solution is given below in next step
Q: What is the overall order of the following reaction, given the rate law? 2 X + 3 Y → 2 Z Rate = k[X]...
A: For the given reaction., 2X + 3Y -----> 2Z According to Rate law Rate of the reaction i...
Q: What volume of 1.44 M KMN04, in liters, contains 292 g of solute? Volume = L. %3D
A: Given that, Molarity of KMnO4 = 1.44 M Molar mass = 158.034 g mol-1 Mass of KMnO4 = 292 g Volume (...
Q: Calculate E° for the reaction: CIO4 + 6 CI` + 6 H3O* →CIO + 3CI2 + 9 H2O E°CIO,/CIO", H30* = +1.360,...
A:
Q: In a voltammogram, which of the following statements is correct a. The limiting current i, is used f...
A: Among the options given option a) the limiting current used for quantitative analysis is correct . T...
Q: Briefly explain why Fe* is a Lewis acid, but Fe" is not a Brønsted-Lowry acid.
A:
Q: According to Kolrausch's law of independent ionic migration, the molar conductivity of an electrolyt...
A: Please find your solution below : Kohlrausch law of independent Migration of Ions states that the mo...
Q: Consider the following reaction: SO2 (g) + NO2 (g) = SO3 (g) + NO (g) Keg = 0.33 A reaction mixture ...
A: Q is the reaction Quotient ,which measure the relative amount of product and reactant at any time . ...
Q: In image
A: The reaction as, N2 + 3 H2 —> 2 NH3
Q: Name of Experiment: The identification of an unknown weak acid by potentiometric titration. Q1: Purp...
A: The purpose of experiment is to identify the unknown weak acid and with the help of potentiometric t...
Q: For the reaction A+B+C→D+EA+B+C→D+E, the initial reaction rate was measured for various initial conc...
A:
Q: Given the following experimental data, calculate the molarity and the pKa of the acid analyte (HA) i...
A: Solution is given below in next step in jpeg format
Q: What volume of 0.172 M NaOH, in militers, contains 200g of NaOH? Volume mL
A:
Q: molarity
A:
Q: Write down the nuclear equations for the following: alpha emission by plutonium-239 beta em...
A: The nuclear reaction takes place by the emission or accepting the radioactive particles like α, β, γ...
Q: 3A + 8B → C + 5D [D] (M) [A] (M) 55.3 41.2 35.8 21.9 11.6 Time (sec) 0.74 1.88 7.16 19 32 12.04 61 1...
A:
Q: he activation energy, Ea, for a particular action is 13.6 kJ/mol. If the rate constant at 54 °C is 2...
A: Activation energy = 13.6kJ/mol
Q: Why does the presence of an unpaired electron make a molecule paramagnetic
A: The answer is as follows:
Q: Calculate the degree of unsaturation. Assign the relevant peaks in the IR spectrum. Assign the peaks...
A: The structure of the following is:
Q: Report the photon energy (in kJ/mol) for the single Li emission and the two Na emission wavelengths....
A: i) To Calculate photon energy (in kJ/mol) for the single Li emission and the two Na emission wavele...
Q: A. Predict the polarity of CF, and CHCI, given the structures below. Using arrows, illustrate the po...
A: Polarity is developed in bonds dueto electronegative difference between two atoms
Q: 1. At a certain temperature, the value of K, for the following reaction is 4.2 x 10: Na) + Ozce) = 2...
A:
Q: You prepared a 0.250 M NH3 solution by diluting 8.45 (±0.04) mL of 28.0 (±0.5) wt% NH3 [density g/mL...
A: Solution -
Q: Aqueous solutions of tin (IV) nitrate and Iron (III) lodide react. Whlch of the following Is the cor...
A:
Q: a) You are given a solution and asked to determine what cations are present in it. the solon of HCl ...
A: Group I cations, Hg22+, Pb2+ and Ag+ precipitate on addition of HCl solution. The reactions are, Hg2...
Q: 1.Consider the complete combustion of methane gas, CH4(g): Which of the following BEST describes the...
A: Entropy is defined as measure of randomness or disorder of system.
Q: .Estimate the critical constants of a gas with the van der Waals constants: a = 0.761 bar dm mol and...
A:
Q: Predict Mat which chenical species of Phe to llowing poirs is hig her in en tropy at 298 k (gases Or...
A: Entropy is the measure of randomness and thus gases have the highest entropy among the solids, liqui...
Q: Which alkyl halide could react faster with NASH in DMSO (dimethyl sulfoxide)? QUESTION 19 ¿Cuál halu...
A:
Q: Question 23 The equilibrium constant Kc for the reaction fructose-1,6-diphosphate = glyceraldehyde-3...
A:
Q: Question 25 The standard cell potential of the reaction is 1.10 V for the cell Zn (s)/Zn2+//Cu²+/Cu ...
A:
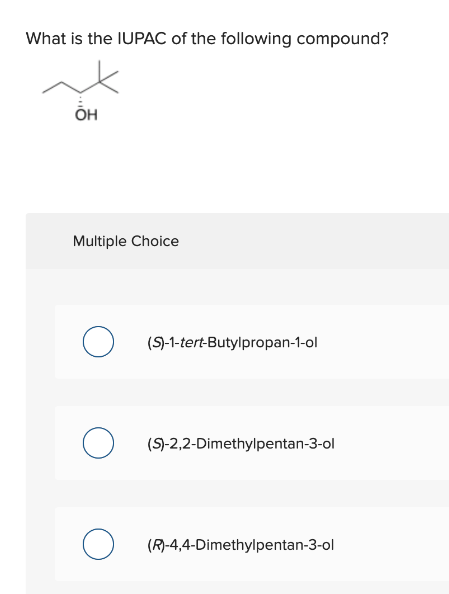
Step by step
Solved in 4 steps with 2 images

- What is the IUPAC name for the following compound? A) 3,5-dimethylhex-4-en-2-ol B) 2,4-dimethylhex-2-en-5-ol C) 1,2,4-trimethylhex-3-en-1-ol D) 1,1,3-trimethylhex-1-en-4-olExplain why each of the following names is incorrect: (a) 2,2-Dimethyl-6-ethylheptane (b) 4-Ethyl-5,5-dimethylpentane (c) 3-Ethyl-4,4-dimethylhexane (d) 5,5,6-Trimethyloctane (e) 2-Isopropyl-4-methylheptaneConsider 3-methylpent-2-ene, if it undergoes hydroboration, which of the following final product is formed? a. 3-methylpent-2-en-2-ol b. 3-methylpentan-3-ol c. 3-methylpentan-2-ol d. 2-methylpentan-3-ol
- What the correct IUPAC name for the compound shown? CH2 I CH3-C-CH-CH2-CH3 I Br What the correct IUPAC name for the compound shown? CH3-CH2-CH-CH2-CH3 I CH2OH What the correct IUPAC name for the compound shown? CH3 I CH3-O-CH2-CH2-CH-CH3How do you draw the structure corresponding to the following IUPAC names? 1) 6-bromo-3-ethyl-1-hexene 2) 4-bromo-3-methyl-1-pentyne 3) 4-isopropyl-1, 1-dimethylcyclohexane can I get an explanation how its done? ThankyouWhat is the IUPAC name for the compound below? choices: 7-tert-butyl-6-isopropyl-5-methyl-2,4,6,8-decatetraene 4-sec-butyl-5-isopropyl-6-methyl-2,4,6,8-decatetraene 6-isopropyl-7-propene-5,9-dimethyl-2,4,6-decatriene 4-isobutyl-5-isopropyl-6-methyl-2,4,6,8-decatetraene
- Which of the following is a correct IUPAC name? 2-chloro-3-bromotoluene 3-bromo-2-chlorotoluene 2-chloro-4-bromotoluene 6-bromo-4-chlorotolueneEach IUPAC is incorrect, what is the correct IUPAC name? -3-butyl-2-isopropylpentane -5-butyl-6-ethyl-4-methylhex-1-ene -3,4-dimethylhex-6-ene-1-yne -3-bromo-3-methyl-6-chloroheptan -1,2,4-trimethyloct-3-ene-7-yneWhat is the correct common IUPAC name for the following structure: CH3-CH2-CO-CH3
- What reagent is necessary to complete the reaction? CH3-CH2-CH-C-OH ? CHỊCH,CH C-0- Na* CH3 CH3 NaO O NaCl O Na O NaOH + H₂OIdentify the correct IUPAC name for the structure shown below. A) 1-tert-butyl-2-chloro-5-bromocycloheptaneB) 5-bromo-1-tert-butyl-2-chlorocycloheptaneC) 1-bromo-4-chloro-5-tertbutylcycloheptaneD) 1-bromo-4-tert-butyl-5-chlorocycloheptaneE) 1-tert-butyl-4-bromo-7-chlorocycloheptaneWhat is the IUPAC name for the following compounds in the image. Please explain if it is cis or trans and illustrate the compounds in their corresponding drawings. For compound 1: a. cis-4-isopropylcyclohexanol b. trans-4-hydroxy-1-isopropylcyclohexane c. trans-4-isopropylcyclohexanol d. cis-4-hydroxy-1-isopropylcyclohexane e. p-isopropylcyclohexanol f. cis-1-hydroxy-4-isopropylcyclohexane For compound 2: a. 3-ethylpent-3-ol b. 2,2-diethylbut-1-ol c. 2-ethylpent-3-ol d. 2-ethylpent-2-ol

