What is the Keq of Reaction #3? Show your work & clearly label your answer. Is Reaction #3 favorable under mammalian cellular conditions when the concentration of dGTP is 0.2 M, dCMP is 20 mM, dGMP-dCMP is 7 mM, and PPi is 10 mM? Use △G to support your answer
What is the Keq of Reaction #3? Show your work & clearly label your answer. Is Reaction #3 favorable under mammalian cellular conditions when the concentration of dGTP is 0.2 M, dCMP is 20 mM, dGMP-dCMP is 7 mM, and PPi is 10 mM? Use △G to support your answer
Principles Of Radiographic Imaging: An Art And A Science
6th Edition
ISBN:9781337711067
Author:Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:Richard R. Carlton, Arlene M. Adler, Vesna Balac
Chapter6: X-ray Equipment
Section: Chapter Questions
Problem 6RQ
Related questions
Question
What is the Keq of Reaction #3? Show your work & clearly label your answer.
Is Reaction #3 favorable under mammalian cellular conditions when the concentration of dGTP is 0.2 M, dCMP is 20 mM, dGMP-dCMP is 7 mM, and PPi is 10 mM? Use △G to support your answer.
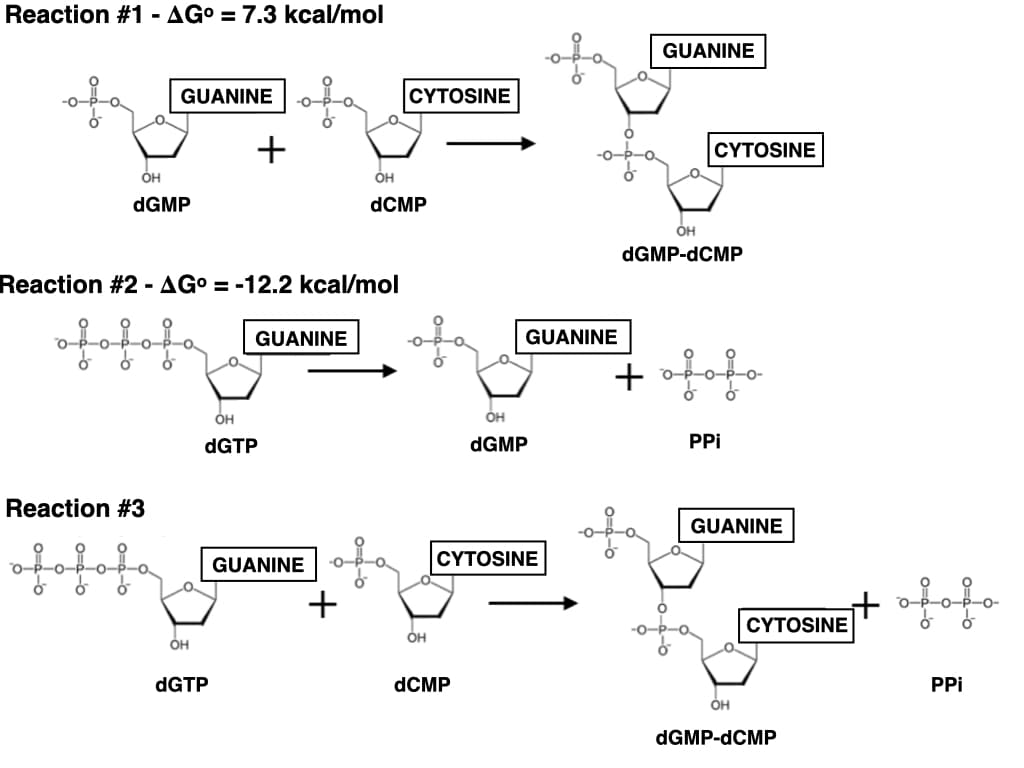
Transcribed Image Text:Reaction #1 - AG° = 7.3 kcal/mol
GUANINE
OH
dGMP
Reaction #3
ofofofo
Reaction #2 - AG° = -12.2 kcal/mol
ofofa
OH
+
dGTP
GUANINE
OH
dGTP
GUANINE
CYTOSINE
OH
dCMP
OH
dCMP
GUANINE
OH
dGMP
CYTOSINE
0=4-0
GUANINE
CYTOSINE
OH
dGMP-dCMP
+ ofofo
PPi
GUANINE
CYTOSINE
OH
dGMP-dCMP
ofofo
PPi
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Recommended textbooks for you

Principles Of Radiographic Imaging: An Art And A …
Health & Nutrition
ISBN:
9781337711067
Author:
Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:
Cengage Learning

Human Physiology: From Cells to Systems (MindTap …
Biology
ISBN:
9781285866932
Author:
Lauralee Sherwood
Publisher:
Cengage Learning

Principles Of Radiographic Imaging: An Art And A …
Health & Nutrition
ISBN:
9781337711067
Author:
Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:
Cengage Learning

Human Physiology: From Cells to Systems (MindTap …
Biology
ISBN:
9781285866932
Author:
Lauralee Sherwood
Publisher:
Cengage Learning