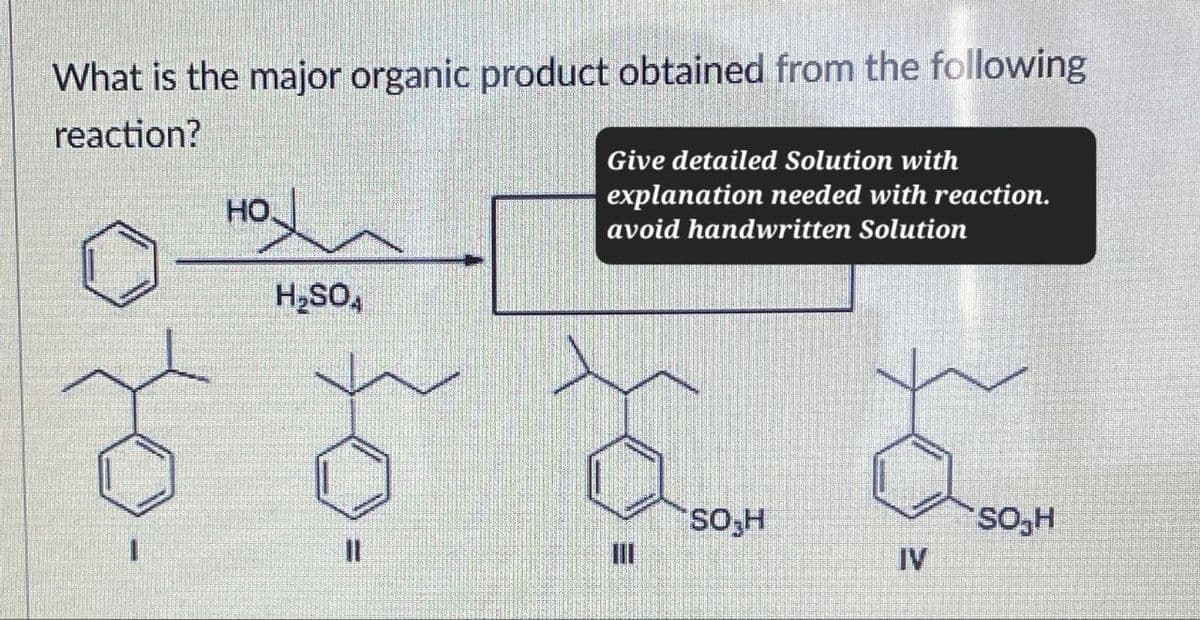
What is the major organic product obtained from the following reaction? HO H₂SOA Give detailed Solution with explanation needed with reaction. avoid handwritten Solution SO₂H SO₂H 11 IV
Q: What is the pH of a solution prepared by mixing 100.00 mL of 0.020 M Ca(OH)2 with 50.00 mL of 0.300…
A: Step 1: Step 2: Step 3: Step 4:
Q: Identify and provide an explanation of what “Separation Science” entails and its importance with the…
A: Seperation science involves the separation of mixtures into their individual components for further…
Q: None
A:
Q: Enter your answer in the provided box. Hydrogen sulfide decomposes according to the following…
A:
Q: 17.43 Draw the products of each reduction reaction. NaBH4 CH3OH OH [1]LIAIH [2] H₂O [1]LIAIH4 OH d.…
A: (a)(b)(c)(d)
Q: None
A: Here's a breakdown of the balancing process using the half-reaction method:1. Split the reaction…
Q: The Ksp of Al(OH)3 is 3.0 x 10-34. Assuming no other reactions (like metal-ligand complex formation)…
A:
Q: 5) Provide the expected mechanism for the following reaction. Please show all intermediates and use…
A: Step 1:
Q: Describe the energy in natural gas and the way in which it's converted to electrical energy?
A: Step 1: Natural gas is primarily composed of methane, with smaller amounts of other hydrocarbons…
Q: None
A: Step 1:Step 2:
Q: Please don't provide handwritten solution ...
A: Step 1:
Q: Gold is frequently purified by electroplating. Typically the gold is dissolved in a cyanide solution…
A:
Q: HO 义 H2SO4 E + F major minor G Identify and label the nucleophile and electrophile
A: Approach to solving the question:Detailed explanation:Examples: Key references:
Q: OH H H Na2Cr2O7, H, H₂O NaBH4, CH3OH + LiAlH4, H3O
A: Step 1: Step 2: Step 3: Step 4:
Q: Draw the product of the reaction shown below at physiological pH (pH = 7.4). Ignore inorganic…
A: Step 1: Step 2: Step 3: Step 4:
Q: please answer in text form and in proper format answer with must explanation , calculation for each…
A: The solubility product expression for aluminum chlorate, Al(CIO₃)₃, can be represented by the…
Q: = O Kinetics and Equilibrium 2/5 Bi Calculating an equilibrium constant from a heterogeneous…
A: Given: V=8.3Lmass,gMolarmass,g/mol2Hg(l)7.2200.59+O2(g)15.432.00→2HgO(s)11.7216.59Step 1:…
Q: For each of the molecules or ions in the chart, do the following • Draw the shape predicted by VSEPR…
A: For CO32-, it has no dipole moment, because all Oxygen atoms are pulling electron density towards…
Q: Which statement about the factors that affect reaction rates is false? Question 8 options:…
A: Option a: This option is true because the higher the concentration of the reactants, the higher the…
Q: Step 2 Add any remaining curved arrow(s) to draw step 2 of the mechanism. Modify the given drawing…
A:
Q: During your first day on the job as a research assistant, you are preparing 50 mL of hybridization…
A: Dilution is a process during which the addition of a solvent, generally water, to a solution causes…
Q: 4. Propose a synthesis for the transformations shown below (no mechanisms needed, just…
A: Synthesis 1: This reaction is completed in three steps first, the deprotonation by a strong base,…
Q: Give the major product for the reaction. า Br2 (low conc.)
A: Step 1: Step 2: Step 3: Step 4:
Q: 7. Give the final product 1) 480+ 2) H2Cr2O7 3) RCO₂H 4) excess LAH 5) H₂O он b) HO- он <) HO- но он
A: in case of any doubt feel free to ask
Q: 79 3. Complete the following reaction and write a mechanism for both the substitution and…
A: The objective of the question is to determine the products of a reaction involving a chiral…
Q: O Kinetics and Equilibrium Calculating an equilibrium constant from a heterogeneous equilibrium...…
A: Given:…
Q: Q1: The following are relative peak areas for chromatograms of standard solutions of methyl vinyl…
A: MKV concentration, mmol/LRelative peak area…
Q: a.)Identify structure of the compound and draw the moleculeb.)Analyse HNMR peaks for (A AND B)C.…
A: HNMR Spectra AnalysisThere are only two types of protons so only two singlet are obtained as we…
Q: Determine the pH change when 0.099 mol KOH is added to 1.00 L of a buffer solution that is 0.359 M…
A: The objective of this question is to calculate the change in pH when a certain amount of KOH is…
Q: Use measured cell potential to calculate concentration. When [Cu2+] = 1.32 M, the observed cell…
A: Step 1: Step 2: Step 3:Step 4:
Q: None
A: The IUPAC nomenclature of organic compounds follows this naming format:Writing the name of organic…
Q: From the data given below calculate the value of ΔS° for the reaction. T = 298.15 K.…
A:
Q: H+ (aq) + HCO2 (aq) -- H2CO (aq) + H2O(l) What is the equilibrium expression.
A: Step 1: The equilibrium expression represents the ratio of the concentrations of the products to the…
Q: Using the information in the table, the value of the rate constant for the reaction A(g) + 3 B(g) →…
A:
Q: Be sure to answer all parts. Enter your answer in scientific notation. Calculate Ke for the…
A: Step 1:-Given the equilibrium equation: 2SO₂(g) + O₂(g) ⇌ 2SO₃(g)We have Kp = 2.5 × 1010 at T = 4.5…
Q: Please provide explanations regarding IUPAC rules
A:
Q: 24. Von Franz's work on alchemy focuses on: A) "The symbolic nature of alchemical texts and…
A: A) "The symbolic nature of alchemical texts and practices." is correct because:Marie-Louise von…
Q: 17. What can you say about the reaction below? 1. OsO4 2. NaHSO3 ? A. This reaction will generate…
A: Step 1: Step 2: Step 3: Step 4:
Q: 8.) Consider the following equilibrated system: 2NO2(g)=2NO(g) + O2(g). If the Kc value is 0.918,…
A:
Q: The weak acid hydrofluoric acid, HF, and the strong base lithium hydroxide react to form the salt…
A:
Q: Draw the product of this reaction. Ignore inorganic byproducts. HO H HO- ・H HO ・H HO ・H CH2OH H2 Pt…
A:
Q: No hand written solution please. Give proper explanation please.
A: Given: [F]=2.00M3F(g)⇌G(g)+2H(g);Kc=3.00Step 1: Fill out the ICE…
Q: A bubble of methane gas, CH, is released from a deep bog. The temperature at the bottor the bog is…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: esc Organic Functional Groups Naming and drawing aldehydes Write the systematic name of each organic…
A:
Q: Retro aldol: H. NaOH H₂O H. HTH "j"82f NaOH
A:
Q: Please correct answer and don't use hend raiting
A: I provided the steps in an image below on how I arrived at a particular answer.
Q: Draw one of the possible diastereomers of the molecule shown below. Use a dash or wedge bond to…
A: A diasteromer of a compound is the isomer that unlike enentiomer is not a mirror image reflection of…
Q: Draw the curved arrow mechanism for the first step in the formation of one repeat unit of this…
A:
Q: 1.3g of copper (II) chloride was dissolved in water and a 0.50g of Al was placed in the solution.…
A: Step 1:Given , Mass of (CuCl2) reacting= 1.3 g Mass of Al reacting = 0.50 g Actual yield of Cu…
Q: Draw a plausible mechanism for the following transformation: 19.70a1 NH₂ N NH₂ [H₂SO] (-H₂O) X…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Step by step
Solved in 2 steps with 1 images

- What is /are organic product(s) expected when 1-butyne is first treated with sodium amide and the resulting acetylide anion reacts with tert-butyl chloride before aqueous acid workup?w how enols, enolate ions, andenamines act as nucleophiles. Predictthe products of their reactions withhalogens, alkyl halides, and otherelectrophiles. Show how they areuseful in synthesis.Compounds A, B, and C have the same molecular formula C4H8. They all react wotu H2/PtO2 to give the same compound. The reaction of A or B with H2O/H2SO4 or with BH3-THF, followed by treatment with a basic solution of hydrogen peroxide, gives the same compound, namely D. The reaction of C with H2O/H2SO4 also gives D. However, the reaction of C with BH3-THF, followed by HO-, H2O2 gives a new compound, E. Provide the identity of A, B, C, D, and E along with explanations of reactivity.
- Ozonolysis of compound D will produce compound E and compound F as products. Hydrationof compound D will produce compound G as major product. Compound D is a major productobtained from dehydrohalogenation of 2-chloro-3-methylbutane. a) Identify the structural formula for compound D, E, F and G.b) Suggest suitable reagent(s) and condition(s) needed for dehydrohalogenation of 2-chloro-3-methylbutaneOne of the products of petroleum refinery is naphtha where, benzene could beobtained via catalytic reforming of naphtha. The obtained benzene can potentiallyto react with Lewis acid to form new carbon-carbon bond. Propose the startingmaterial and stepwise mechanism to produce new chemical structure which consista formula molecule of C11H16.The hydrocrbon B undergoes catalytic hydrogenation (H2, Pd/C) to give 1-isopropyl-4-methylcyclohexane. Using the ozonolysis products for B as a guide, reconstruct the structures of B.
- Draw the structure(s) of the organic product(s) predicted when this compound reacts with acetylide ion then H3O+.Acetoxybenzene (PhOC(=O)OCH3) is much less reactive than ethoxybenzene (PhOCH2CH3) in electrophilic aromatic substitution reactions. Suggest an explanation for this result, based on an analysis of the inductive and resonance electronic effects of the two substituents on the stability of theWheland intermediate for para substitution by an electrophile E+Following is a balanced equation for bromination of toluene.(a) Using the values for bond dissociation enthalpies given in Appendix 3,calculate ∆H0for this reaction.(b) Propose a pair of chain propagation steps and show that they add up to theobserved reaction.(c) Calculate ∆H0for each chain propagation step.(d) Which propagation step is rate-determinin
- Provide a primary organic product(s) for each reaction shownIdentify the pericyclic reactions in the followingreaction schemes. Give the complete reaction name and indicate the course of the reaction with the aid of the arrow notation.Can you please check my answers for the following reaction schemes... The question ask to provide the bond line structures for the major organic product obtained at each step.. Thank you!!!



