Q: Consider the image of a mercury manometer below. Based on the image, if the pressure of the…
A:
Q: What is the main aspect that the enzymes succinyl-CoA synthetase, pyruvate kinase, and…
A: Succinyl coA synthetase is the enzyme involved in the conversion of succinyl coA to succinate.…
Q: Question 18 of 20 hat glycolytic intermediate is glycerol, formed by hydrolysis of triacylglycerols,…
A: Hydrolysis of the lipid molecule triacylglycerols will produce 3 fatty acid molecules and one…
Q: What is the ksp of Silver sulfate
A:
Q: ● Which sequence will produce the following transformation? 1. 2. 3. 4. 5. PhMgBr Et₂O PhMgBr Et₂O…
A: Given reaction: We have to find the correct sequence for this transformation.
Q: What is the [OH-] in a solution that has a [H3O+] = 1.0 × 10-5 M?
A: pH measured the acidity of the solution and pOH measured the basicity of the solution. Recall data,…
Q: Calculate the molarity of the two solutions. The first solution contains 0.200 mol of NaOH in 1.65 L…
A: Molarity is defined as the number of moles of solute present in per litre of solution. It is denoted…
Q: estion 51 Which of the following occurs during Fischer esterification? O a. The -OH from the…
A:
Q: = 5. a) Calculate the pH of a buffer solution which is 0.1 mol/L with respect to HCN (Ka 4.9 x 10-10…
A: a). pH of a buffer solution can be calculated with the help of Henderson - Hasselbalch equation : pH…
Q: 15. Label the following transition state with the applicable following items (not all labels will…
A: This is an example of basic diels Alder reaction.
Q: The product P in the following chemical reaction will be 0= CI 1. NaN3 2. Heat 3. H₂O P
A:
Q: Determine ∆G° for a reaction when ∆G = -122.6 kJ/mol and Q = 0.043 at 298 K. (R = 8.314 J/mol ・ K)
A: The free energy change of the reaction is = -122.6 kJ/mol The temperature of the reaction is = 298 K…
Q: A solution may contain Ag*, Pb²+, Hg₂². A white precipitate forms on addition of 6 M HCI. The…
A: Ag+, Hg2+2 and Pb+2 are group-I cation. They can be separated by group separation technique.
Q: Calculate the pH of a buffer which is 0.2 mol/L with respect to ammonium sulphate and 0.1 mol/L with…
A: Given data is as follows: The concentration of salt (ammonium sulphate) = 0.2 mol/L The…
Q: NO₂ CH3CI, AICI
A: This can be considered as the example of Friedel craft alkylation. The presence of Nitro group in…
Q: How many grams of a 12.3% sugar solution contain 48.0 g of sugar? g solution:
A: Given, The mass of 12.3 % sugar solution that contain 48.0 g of sugar is:
Q: IDENTIFY THE OXIDATION NUMBER OF EACH ELEMENT IN THE GIVEN CHEMICAL FORMULA. -All answers to be…
A: The chemical formula of sodium chloride is NaCl
Q: O₂N -NO₂ H₂O NH₂ NH₂ 2 O-N+
A:
Q: Part A Use average bond energies together with the standard enthalpy of formation of C(g) (718.4…
A:
Q: Calculate the molar solubility of Ag2CrO4 in a solution that is 0.01 M in AgNO3. Ksp of Ag2 CrO4 =…
A:
Q: For the compound shown below, state the number of signals expected the ¹3C NMR and the ¹H NMR. 13C…
A:
Q: I ift ↑ Qlaccds X tab ΤΟΝ C caps lock H fn esc Re → O CHEMICAL REACTIONS Calculating molarity using…
A:
Q: balanced equation for the neutralization reaction of aqueous H₂SO4 with aqueous KOH is shown.…
A: Given Reaction :- H2SO4 + 2KOH -----> K2SO4 + 2H2O Molarity of KOH = 0.220M Molarity of H2SO4 =…
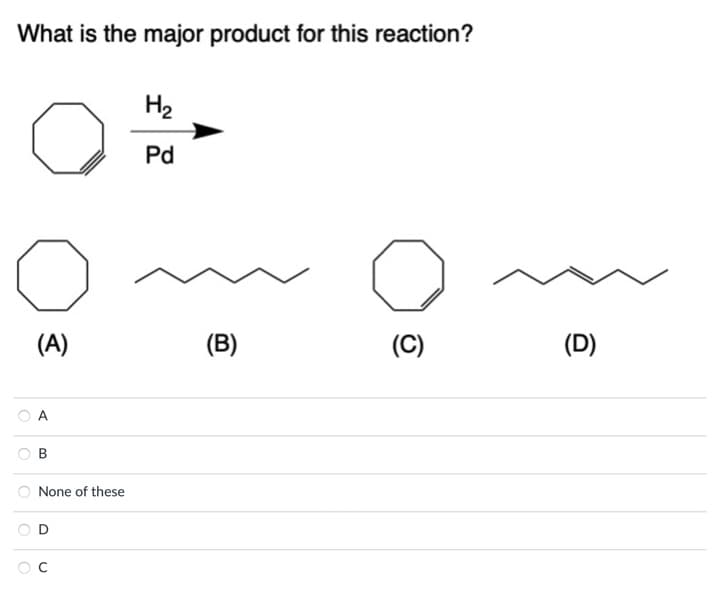
Q: What is the major product for this reaction? (A) H₂ Pd (B) (C) ??? (D) What reagents would you use…
A:
Q: oncisely define and differentiate the Ollowing concepts a. analytical information, analytical…
A:
Q: Criven belove Some Compound Choose the coverend Covalent Compound I (1) (NH4)3 AS03 (11) POL- (TP:)…
A: We need to find the covalent compound.
Q: How many more times basic is something with a pH 13 as compared to a pH of 11?
A: Hydroxide ion concentration measure the basicity of the solution. If pH more then 7 then solution is…
Q: But you didn't make use of the data given. We're supposed to use the data to find the answer.
A: Heat of combustion: It is defined as the amount of heat released when one mole of a given compound…
Q: How many mols of HBr are used to neutralize 18.66 mL of a 0.2299 M Ca(OH)₂ solution?
A:
Q: Assess the validity of each statement and choose X if the statement is CORRECT, Choose Y if…
A:
Q: Stronger bonds will be found where in the infrared spectrum? O Lower molecular ion O Higher…
A:
Q: 3. Examine the following graph. What are the relative strengths of the acid and base that were used…
A:
Q: A 87.0 mL aliquot of a 0.500 M stock solution must be diluted to 0.100 M. Assuming the volumes are…
A: M1 =0.500 M M2 =0.100 M V1 =87.0 ml V2 =to find
Q: Provide the major product of the SO₂H Cl₂ AICI
A: Given reaction is halogenation reaction. Cl substitutes ring hydrogen.
Q: What descriptive term is applied to the type of diene represented by 1,5-octadiene ? O alkynyl diene…
A: Each type of diene is represented by the position of two double bonds present in the compound. a.…
Q: Over-the-counter hydrogen peroxide solutions are 3 % (w/v). You may want to reference (Pages…
A:
Q: Ethyl Mercaptan (C2H5SH) is a colorless or yellowish liquid or a gas with a pungent, garlic or…
A: Given :- Mass of Ethyl Mercaptan, C2H5SH sample= 3.85 g Molarity of I2 = 0.01204 M = 0.01204 mol/L…
Q: Cuien H₂₂ (g) + F₂ (g) = 2HF (g) Find the partial pressure of HF? Crien An empty Steel Container is…
A:
Q: Reaction Type:_ At completion of reactions: Grams of hydrogen peroxide Grams of water Grams of…
A: Decomposition reactions are the reactions in which chemical species breaks into simpler molecules.
Q: Would you please answer this accordingly in the question as it mentioned? D) Finally show how an…
A: As per the Bartley expert guidelines, I am not allowed to attach references. So, unable to solve E…
Q: Calculate the standard Gibbs free energy (in kJ/mol) of the reaction below. (Use R = 8.314 J/mol-K;…
A: Given reaction is : 6Cu (s) + Cr2O72- (aq) + 14H+ (aq) -------> 6Cu+ (aq) + 2Cr3+ (aq) + 7H2O…
Q: Arrange the highlighted bonds in the table below in decreasing order of polarity. That is, pick 1…
A: The polarity of a bond depends on electronegativity difference between the two atoms sharing the…
Q: Predict the Product Provide the major organic product(s) for each of the following reactions shown…
A: The reaction proceed as:
Q: Identify the mechanism by which each of the reactions above proceeds from among the mechanisms…
A:
Q: Calculate the mass percent of solute in each solution. Calculate the mass percent of 3.07 g KCl…
A:
Q: A gas sample containing 12.0 g of He has a volume of 14.0 L. What will the volume be if 1.6 g of H2…
A:
Q: Question 4 What is the approximate boiling point of the liquid in the mixture with a lower boiling…
A: First plateau shows the boiling point of lower boiling point component at 70°C and second plateau…
Q: Consider the electrochemical cell described by the following reaction at 310 K. The measured cell…
A:
Q: Mass of sodium chloride used to create a 100 ml solution at a concentration of 1.5 M
A: We need to find mass of sodium chloride.
Q: QUESTION 10 How many grams of MgO are produced when 40.0 grams of O2 react completely with Mg? 2Mg +…
A: Here we have to determine the mass of MgO produced when 40 g of O2 react completely with Mg in the…
Step by step
Solved in 2 steps with 2 images

- Compound 1 is an anticoagulant that is extracted from, among other plants, the sweetclover plant, Melilotus Officinalis. However, the compound is moderately toxic and alsofinds use as a rodenticide. Name this compound2,3-Dimethylfumaric acid has a molecular formula C6H8O4. It undergoes oxidativecleavage to form two identical compound N. Compound N is then reacted withethylmagnesium bromide to form compound O. Compound O is then hydrolysed inacidic condition to form compound P. Draw the structure of compound N, O and P. PLEASE PROVIDE CLEAR HANDWRITING, and explantionCompound A is an alcohol that undergoes oxidation to produce compound B.Compound B is a ketone that gives positive triiodomethane reaction. Compound B isthen reacted with phenyl magnesium bromide, C6H5MgBr in the presence of aqueousacid to form compound C. Compound C has the molecular formula of C9H12O. Deducethe structure for compound A, B and C. PLEASE PROVIDE CLEAR DRAWINGS AND EXPLANATIONS
- (a) Draw the structures of the following compounds :(i) 4-Chloropentan-2-one (ii) p-Nitropropiophenone(b) Give tests to distinguish between the following pairs of compounds :(i) Ethanal and Propanal (ii) Phenol and Benzoic acid(iii) Benzaldehyde and Acetophenoneprovide the reagents necessary to complete the following information.Compound J, C16H16Br2, is optically active. On treatment with strong base, compounds K and L (each C16H14) are formed; K and L each absorb only 2 equivalents of hydrogen when reduced over a Pd/C catalyst. Compound K reacts with ozone to give phenylacetic acid (C6H5CH2COOH), while similar treatment of L gives 2 products. One product, M, is an aldehyde with formula C7H6O; the other product is glyoxal (CHO)2. Draw the structure of compound L.
- Identify A, B, and C, intermediates in the synthesis of the ve-membered ring called an α-methylene-γ-butyrolactone. This heterocyclic ring system is present in some antitumor agents.Compound A is first treated with ylide Y and then reacted with Rh and hydrogen gas. Using the spectroscopic data provided, what is the structure of Compound A?Identify A, B, and C, intermediates in the synthesis of the five-membered ring called an α-methylene-γ-butyrolactone. This heterocyclic ring system is present in some antitumor agents.
- Quinapril (trade name Accupril) is used to treat high blood pressure andcongestive heart failure. One step in the synthesis of quinapril involvesreaction of the racemic alkyl bromide A with a single enantiomer of theamino ester B. Given the structure of quinapril, which one of these two products isneeded to synthesize the drug?what is the structure of compound B?Although ibuprofen is sold as a racemic mixture, only the S enantiomer acts as an analgesic. In the body, however, some of the R enantiomer is converted to the S isomer by tautomerization to an enol and then protonation to regenerate the carbonyl compound. Write a stepwise mechanism for this isomerization.



