Q: Describe a sequence of reactions by which 3-methyl-1-butyne can be straightforwardly prepared from…
A:
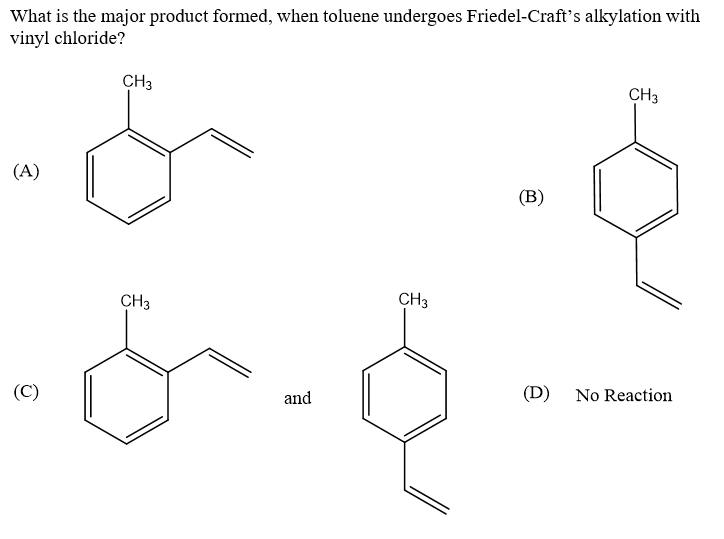
Q: what is the major product formed when toulene react with vinyl chloride?
A: Given: Toluene reacts with vinyl chloride. To find: Major product formed from the above reaction.…
Q: What product is obtained from the reaction of dichlorocarbene with cyclopentene?
A: A simple trick for this reaction is, just insert the dichlorocarbene into the double bond making it…
Q: Which one of these molecules can be a reactant in a Friedel-Crafts reaction? and why? a) Aniline…
A:
Q: What factor seems to be responsible for the Wagner-Meerwein rearrangement of camphene to give…
A: The rearrangement of camphene to give isobornyl acetate. in general carbocations will rearrange from…
Q: Write the structures of all the alkenes that can be formed by dehydrohalogenation of the following…
A:
Q: Can you explain the process of free radical bromination by using methylcyclohexane?
A: This reaction have done in three step. Step1: Chain Initiation Formation of two free radical Step2:…
Q: Provide the curved-arrow mechanism to account for the following nucleophilic addition- elimination…
A: The curved-arrow mechanism to account for the given nucleophilic addition elimination reaction can…
Q: PLEASE HELP DRAW AND WRITE THE REACTION, EQUATIONS AND MECHANISMS FOR THE REACTION OF CYCLOHEXENE…
A: Alkene reacts with Br2 in presence of water to give halohydrin.
Q: The coupling of an alkyne with an aryl halide in the presence of a palladium catalyst and…
A: Please find below the reactants along with the reaction taking place.
Q: Which of the following is the least reactive to Friedel-Craft Alkylation and Acylation? A.…
A: Friedel Crafts alkylation and Friedel Crafts acylation: Friedel Crafts acylation is the process of…
Q: Describe the way that the following compound could be synthesize from benzene.
A: Ans- 1) synthesis of benzene- Benzene having chemical molecular formula is C6H6 can be…
Q: Solvolysis of 2-bromo-2-methylbutane in acetic acid containing sodium acetate gives three organic…
A: When 2-bromo-2-methylbutane undergoes solvolysis in the presence of acidic acid and sodium acetate…
Q: Because bromocyclohexane is a secondary alkyl halide, both cyclohexanol and cyclohexene are formed…
A:
Q: How many different monosubstituted products are possible when ethylcyclopentane reacts with one…
A:
Q: Draw all the Kekulé structures of anthracene and phenanthrene.
A: Concept introduction: The structure of benzene consists of carbon atoms arranged in a hexagon with…
Q: Which one of the following alkenes is the Zaitsev product formed by the reaction of hydroxide with…
A: Elimination reactions usually produce the more highly substituted alkene, called the Zaitsev…
Q: Which of the following steps is correct to convert Phenol to benzene and vice Versa (a) Reduction…
A: Organic reactions are chemical reactions in which organic compounds are involved. Addition…
Q: Predict the product that forms upon reaction of 1-methylcyclohexene with iodine monochloride, I-Cl,…
A: When alkene or alkyne react with hydrogen halide it gives alkylhalide (haloalkane) as a product.…
Q: Which of the following is the product upon reaction of cyclopentene with cold dilute neutral…
A: Heat of hydrogenation = enthalpy change when catalytic hydrogenation of one mole alkene
Q: Provide Examples of dehydrohalogenation of dihalides to afford alkynes ?
A: Alkynes- Unsaturated hydrocarbon containing carbon carbon triple bond is known as alkyne. Alkynes…
Q: what is the major monoalkylation product you would expect to obtain of bromobenzene from reaction…
A: Welcome to bartleby ! We have to tell the major product
Q: What is the purpose of HCl in the iodination of vanillin. And what is the complete mechanism for…
A:
Q: Describe a sequence of reactions by which 3-heptyne can be straightforwardly prepared from…
A:
Q: What reaction do nitro compounds undergo when treated with reducing agent?
A:
Q: Considering the stereochemistry, what is the name of the product when cyclopentene undergoes…
A: Bromination of alkene is defined as when the alkene is treated with bromine then carbon to carbon…
Q: What aspect of the structure of the alkene does ozonolysis not tell you?
A: During the ozonolysis what happens is the double bond breaks and double bonded O gets attached to…
Q: Give the structure of hydrocarbons that yield CH;CH2COCH3 upon ozonolysis.
A:
Q: An unknown alkyne is treated with ozone (followed by hydrolysis) to yield acetic acid and carbon…
A: An unknown alkyne is treated with ozone (followed by hydrolysis) to yield acetic acid and carbon…
Q: Which set of reagents would be appropriate to synthesize bromobenzene from benzene?
A: Given :- benzene → bromobenzene To identify :- correct set of reagentS for the the above…
Q: Methyl salicylate cannot be produced by using methyl chloride through an SN2 process. Explain. What…
A: This can be explained by SN2 reaction mechanism
Q: in the hydrohalogenation reaction of 2,2,3,5,5-pentamethyl-3-heptene with HCL, what is the minor…
A:
Q: How is alkene produced by vicinal dihalide?.
A: Vicinal dihalides are organic compounds that have two halogen atoms present at adjacent positions.…
Q: Identify the product of the reaction of an alkene with diazomethane or the Simmons-Smith reagent…
A:
Q: Friedel-Crafts acylation of p-xylene,
A:
Q: Provide the curved-arrow mechanism to account for the following nucleophilic addition reaction. H20…
A: Hydrolysis of amine to nitrile is nucleophilic addition reaction. And mechanism for this reaction…
Q: Which of the following pair of reactions were used in the synthesis of 3-chloro-6-popyllbenzoic acid…
A: Given: To synthesis 3-chloro-6-popyllbenzoic acid from benzene.
Q: Provide the name of the product after cyclopropanone reacts with methylmagnesium bromide and…
A:
Q: Nerol, a natural product from lemongrass, can be converted to α-cyclogeraniol upon treatment with…
A: Given reaction,
Q: Provide the systematic name of the major product after (S)-1-bromo-4-methylhexane reacts with sodium…
A: Given reaction conditions NaOH in diethyl ether is of SN2 reaction. So here substitution reaction…
Q: Draw the structure of the major product that would be obtained from the dehydration of…
A:
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

- An unknown hydrocarbon A with the formula C6H10 reacts with 1 molar equivalent of H2 over a palladium catalyst to give B C6H12 (Rxn 1). Hydrocarbon A also reacts with OsO4 to give the glycol C (Rxn 2). A gives 5-oxohexanal on ozonolysis (Rxn 3). Draw the structures of A, B, and C. Give the reactions.Compound A of molecular formula C8H14 is reduced by sodium in liquid ammonia to give compound B of molecular formula C8H16. product (Y).Both A and B undergo hydrogenation in the presence of a platinum catalyst to give 2,5-dimethylhexane. Ozonolysis of B with an oxidative workup produces a carboxylic acid of molecular formula C4H8O2. Reaction of B with a peroxyacid gives a chiral C8H14O product, but reaction with bromine gives an achiral C8H14Br2 product. What are the identities of A and B?2,3-Dimethylfumaric acid has a molecular formula C6H8O4. It undergoes oxidativecleavage to form two identical compound N. Compound N is then reacted withethylmagnesium bromide to form compound O. Compound O is then hydrolysed inacidic condition to form compound P. Draw the structure of compound N, O and P. PLEASE PROVIDE CLEAR HANDWRITING, and explantion
- Draw the product(s) obtained when benzoquinone is treated with excess butadiene. Using wedges and dashes, indicate the stereochemistry.Birch Reduction of toluene leads to a product, X with the molecular formula C7H10. After ozonolysis of X, the two compounds, 3-oxobutanal and malonaldehyde are formed. what is the structure of xCompound J, C16H16Br2, is optically active. On treatment with strong base, compounds K and L (each C16H14) are formed; K and L each absorb only 2 equivalents of hydrogen when reduced over a Pd/C catalyst. Compound K reacts with ozone to give phenylacetic acid (C6H5CH2COOH), while similar treatment of L gives 2 products. One product, M, is an aldehyde with formula C7H6O; the other product is glyoxal (CHO)2. Draw the structure of compound L.
- Alcohol A (C10H18O) is converted to a mixture of alkenes B and C on being heated with potassium hydrogen sulfate (KHSO4). Catalytic hydrogenation of B and C yields the same product. Assuming that dehydration of alcohol A proceeds without rearrangement, deduce the structures of alcohol A and alkene C.Ibufenac, a para-disubstituted arene with the structureHO2CCH2C6H4CH2CH(CH3)2 , is a much more potent analgesic thanaspirin, but it was never sold commercially because it caused livertoxicity in some clinical trials. Devise a synthesis of ibufenac frombenzene and organic halides having fewer than five carbons.An unknown compound A of molecular formula C10H18O reacts with H2SO4 to form two compounds (B and C)of molecular formula C10H16. B and C both react with H2 in the presence of Pd-C to form decalin. Ozonolysis of B forms D, and ozonolysis of C forms a diketone E of molecular formula C10H16O2. Identify the structures of compounds A, B, C, and E.
- Compound AA, C6H14O, reacts very fast and immediately form a cloudy solutionwith Lucas reagent. Compound BB, an alkene is formed when compound AAreacted with concentrated sulfuric acid, H2SO4.Ozonolysis process of compoundBB will produce only propanone. Reduction of propanone with Nickel, Ni givescompound CC. Esterification of compound CC with propanoic acid producescompound DD. Draw the structural formula of compounds AA, BB, CC, and DDCompound A, C11H16O, was found to be an optically active alcohol. Despite its apparent unsaturation, no hydrogen was absorbed on catalytic reduction over a Pd/C catalyst. On treatment of A with dilute H2SO4, dehydration occurred and an optically inactive alkene B, C11H14 was produced as the major product. Alkene B, on ozonolysis, gave two products. Product C, C7H6O, was shown to be an aldehyde while product D, C4H8O, was shown to be a ketone. Draw the structure of compound C. You do not have to consider stereochemistry. You do not have to explicitly draw H atoms. In cases where there is more than one answer, just draw one. HELP PLEASE I DONT UNDERSTAND THE PROCESSCompound A, C11H16O, was found to be an optically active alcohol. Despite its apparent unsaturation, no hydrogen was absorbed on catalytic reduction over a Pd/C catalyst. On treatment of A with dilute H2SO4, dehydration occurred and an optically inactive alkene B, C11H14 was produced as the major product. Alkene B, on ozonolysis, gave two products. Product C, C8H8O, was shown to be a methyl ketone while product D, C3H6O, was shown to be an aldehyde.

