Q: Alright, you have added 800 μL of the buffer to the tube. If I asked you to now add 10 μL of protein…
A: Micropipette is used to transfer small volumes as small as 0.2 microliters.
Q: Add any remaining curved arrow(s) to draw the mechanism for the formation of B. ¹0% NB Be H₂C
A: when alkyne A react with benzyl azide, a [3+2] cycloaddition occurs between the triple bond and the…
Q: In the electrolysis of water, how long will it take to produce 225.0 L of H2 at 1.0 atm and 273 K…
A:
Q: Draw and name all possible products indicating the major species.. α + NaOCH₂CH3
A: The given reaction is an simple example of elimination or E2 reaction to give alkene as final…
Q: ame the organic product that is formed when 2-methylpent-1-ene and HCI react.
A: -> When alkene reacts with HCl it proceeds through formation of carbocation if possible then…
Q: Use the method of initial rates to determine the rate constant, k, given the reaction and rate…
A:
Q: The primary product of the following reaction is which of the following? CH3 H2SO H2O + OH OH H3C OH…
A: This is an example of hydration of alkene
Q: Draw and name all possible products indicating the major species. + H₂O
A: Hydroxy alkanes (alcohols) are formed in the hydration reaction of alkene. This reaction proceeds…
Q: Use the information for the reaction given below to answer the following questions. CH4 (g) + 2H₂O…
A: Decrease in pressure will not have any effect on Kc. In crease in pressure shifts the equilibrium in…
Q: Given the reaction: PCI 5 (g) PC|3 (g) + Cl₂ (g) which has Kc =0.0454 at 261 °C. In which direction…
A:
Q: Name the starting compound, give the Major product(s): HO Ch/CH-Ch EtOH/H,SO4 1. 2 EtOH/H₂SO4 Name…
A: A C=C bond reacts with a chlorine molecule to produce a dichloro compound.In this reaction, a…
Q: How many moles of CH3OH are there in 1 L of 0.500 M CH3OH?
A:
Q: 2. use benzene as one of starting materials)
A:
Q: Which compound would be the least likely to react as a diene in a Diels- Alder Reaction? A. B. C. D.
A: This involves Diels-Alder reaction knowledge.In Diels-Alder reaction, a diene and dienophile react…
Q: Consider the reaction below and answer the question below the reaction. A student performed a…
A:
Q: Mass of H₂O b.o Mass of CO2 g Similarly, determine the mass of CO₂ produced from the mass change in…
A: we have to calculate mass of H2O and mass of CO2.
Q: 30.00mL of a 0.050M unknown base is titrated with HCl After 5.00mL of 0.100M HCl, the pH is 9.2.…
A: Given,Molarity of an unknown base = 0.050 Mvolume of base = 30.00 mLMolarity of HCl = 0.100 MVolume…
Q: Draw the structural formula for 3-chlorohexanenitrile. O O O O O CT CH 3 CH ₂ CH CH ₂ CH ₂ CN J CHB…
A: To make structure of the formula we need to determine the functional group present and the position…
Q: The molecule CCI₂O has A A A double bonds, and it is 9 total valence electrons, the shape is the…
A:
Q: A mixture contains 0.0100 M of both Cut ions and Aut ions. If NaCl is slowly added to the solution,…
A:
Q: In the in the ¹H NMR spectrum of the molecule below, what is the multiplicity of the protons…
A:
Q: Reference the vapor pressure diagram above and the table below, which provides the chemical formulas…
A: The boiling point of a substance is the temperature at which its vapor pressure equals the…
Q: Name the following: OH HO 0
A:
Q: PC15 (s) + H₂O(1) → POCI3 (1) + 2HCl(aq) When 58.83 g of phosphorus pentachloride reacts with water,…
A: Here, in this question balanced chemical equation concept is applied.A balanced chemical equation…
Q: Acetic acid (CH₂COOH, K₂ = 1.80 x 105) is a weak acid, so the salt sodium acetate (CH, COONa) acts…
A: Given,Molarity of sodium acetate = 0.451 MKa of acetic acid ( CH3COOH ) = 1.80 x 10-5
Q: What is/are the most acidic proton(s) in the moluecle below? Select the answer where the most acidic…
A:
Q: =X =Y Use the diagram shown above to answer the following questions. All atoms/molecules are in the…
A: X2 combines with Y2X to produce X3 and Y2Entropy increases if the number of molecules on product…
Q: 85. A solution containing 27.55 mg of an unknown protein per 25.0 mL solution was found to have an…
A: mass of protein (m) = 27.55 mg = 0.02755 gVolume of the solution (V) = 25.0 mL = 0.025 LOsmotic…
Q: Consider the following crossed aldol condensation. Which statement explains why a single cross…
A: We have to select the correct option for the single product.
Q: Which of the statement is INCORRECT? a. The increase in stability of 2,4-hexadiene over…
A: In the field of chemistry, resonance refers to the delocalization of electrons in certain molecules…
Q: How many of the following are aromatic? 2 3 *
A: This question belong to General Organic chemistry that is Aromaticity. Aromatic Compound are…
Q: Write the products for this reactions. a.. b. C. d. ر لحمد سين سند OH 1. 2 OH H* 2 * NH₂ H₂O H* MgBr
A: The hydrolysis of an anhydride involves the reaction of water with the anhydride compound, resulting…
Q: Determine the most appropriate starting material for one of these reactions. b) c) 6 mCPBA, CH₂Cl₂…
A: An epoxide is a cyclic ether made from three atoms such as oxygen connected to two adjacent carbons.
Q: A OH В О CH 3 OH D
A: IR spectroscopy, or infrared spectroscopy, is a technique used in analytical chemistry to study the…
Q: What is the concentration of A after 37.5 minutes for the reaction A → Products when the initial…
A:
Q: QUESTION.25 Consider the Energy Diagram labeled below. Which letter labels the Activation Energy? OA…
A: The activation energy of a reaction is the minimum amount of energy required to start the reaction.
Q: The equilibrium constant, Ke. for the following reaction is 55.6 at 698 K. H₂(g) + 12 (9) 2HI(g)…
A: Kc = 55.6Initial concentration of H2 = moles/volume = 0.287/1 = 0.287 MInitial concentration of I2 =…
Q: When naming an alcohol, the root is based on the parent alkane the number of the carbon which…
A: The IUPAC name of the compound can be written on the basis of the number of carbon atoms in the main…
Q: Which of the following transitions (in a hydrogen atom) represent the absorption of the smallest…
A: Smallest frequency photonn = 3 to n =1n = 1 to n = 5n = 5 to n = 2n = 2 to n = 4n = 5 to n = 6
Q: 3) Complete the following reaction and explain your answer CH3 CH3 + CH3MgBr ?
A: The above reaction is between reactant epoxide and grignard reagent.In this reaction the epoxide…
Q: Example of Isolated diene is: a. Ob. OC. Od.
A: Isolated dienes : in Isolated dienes, two double bonds are separated by more than one single bond.…
Q: 14. At 25°C, consider a solution that contains the following two equilibrium reactions. pK₂ = 6.37…
A: The correct relationship between concentration of conjugate base and acid is to identified for the…
Q: The equilibrium HBrO (aq) + H₂O(l) H3O*(aq) + BrO¯ (aq) forms some products but strongly favors…
A: HBrO (aq) + H2O(l) ⇌ H3O+ (aq) + BrO- (aq)Given that, products are favored.
Q: Consider the reaction of propanol with TBS-CI/NEt3. As the reaction proceeds the pH rises As the…
A: Let us first study the reaction completely to find out the effect on pH.
Q: Which statement describes an esterification reaction? It involves a carboxylic acid and an alkane.…
A:
Q: Use the following information to answer the next question. CH₂ CH, - butane pentane heptane hexane…
A:
Q: Predict the major final organic product for the reactions below
A: In the first reaction is the aromatic necleophilic substitution reaction which proceed through the…
Q: A. Br B. C. AB с D E U Br Br OH xs HBr Heat D. E. F. Br Br OH
A: -> In presence of acid ether gives alkyl halide and alcohol .-> If acid (HX) present in excess…
Q: 1. Give a systematic name for each of the following structures. Indicate relevant stereochemistry if…
A: The carbon containing four different groups/atoms is known as chiral carbon. At chiral center the…
Q: QUESTION 8 Match the following atoms with their formal charges referring to activity 6 on page 7 ✓…
A: Formal charge on the desired atom in given molecules is to be determined.
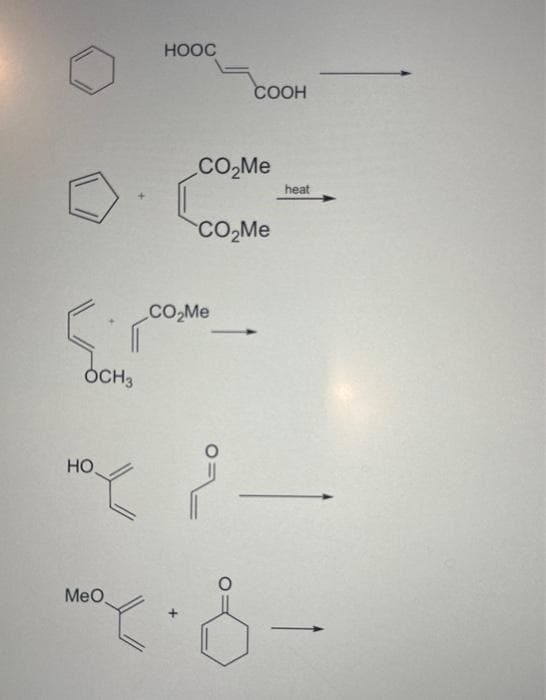
What is the major product of the following Diels Alder reactions?
Step by step
Solved in 3 steps with 1 images


