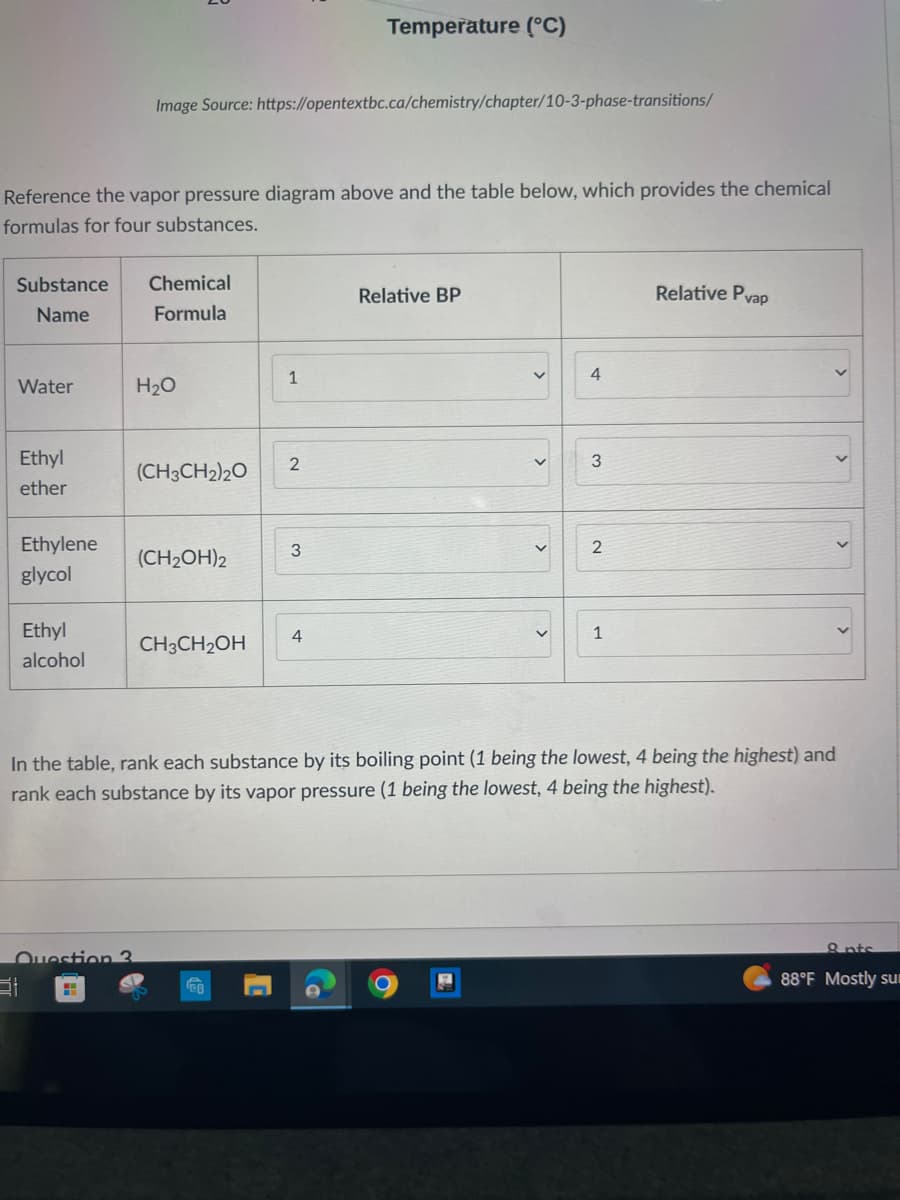
Reference the vapor pressure diagram above and the table below, which provides the chemical formulas for four substances. Substance Name Water Ethyl ether Ethylene glycol Image Source: https://opentextbc.ca/chemistry/chapter/10-3-phase-transitions/ Ethyl alcohol Chemical Formula H₂O (CH3CH₂)20 (CH₂OH)2 CH3CH₂OH 1 2 3 4 Relative BP < < < 4 3 2 1 Relative Pvap In the table, rank each substance by its boiling point (1 being the lowest, 4 being the highest) and rank each substance by its vapor pressure (1 being the lowest, 4 being the highest). >
Reference the vapor pressure diagram above and the table below, which provides the chemical formulas for four substances. Substance Name Water Ethyl ether Ethylene glycol Image Source: https://opentextbc.ca/chemistry/chapter/10-3-phase-transitions/ Ethyl alcohol Chemical Formula H₂O (CH3CH₂)20 (CH₂OH)2 CH3CH₂OH 1 2 3 4 Relative BP < < < 4 3 2 1 Relative Pvap In the table, rank each substance by its boiling point (1 being the lowest, 4 being the highest) and rank each substance by its vapor pressure (1 being the lowest, 4 being the highest). >
Chapter87: Steam Distillation
Section: Chapter Questions
Problem 2P
Related questions
Question
Is this correct?
Transcribed Image Text:Substance
Name
Reference the vapor pressure diagram above and the table below, which provides the chemical
formulas for four substances.
Water
Ethyl
ether
Ethylene
glycol
Ethyl
alcohol
Question 3
Et
Image Source: https://opentextbc.ca/chemistry/chapter/10-3-phase-transitions/
Chemical
Formula
H₂O
(CH3CH2)2O
(CH₂OH)2
CH3CH₂OH
1
TO
2
Temperature (°C)
3
4
Relative BP
>
2
>
>
<
4
3
In the table, rank each substance by its boiling point (1 being the lowest, 4 being the highest) and
rank each substance by its vapor pressure (1 being the lowest, 4 being the highest).
2
1
Relative Pvap
>
8 ntc
88°F Mostly sur
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning