Q: what can we discuss about the graph below about the relationship between water and alcohol in a…
A: Solutions- This graph shows the relationship between water and alcohol.
Q: Draw the structure of the product: H3C- H3C- -CH3 НС
A: Addition of HX follows Markovnikov's addition, i.e., when HX is added to an unsymmetrical alkene,…
Q: Question 4 What is the factor responsible for the separation in column chromatography? O Polarity…
A: chromatography is based on the mobile phase which is solvent and stationary phase which is SiO2.
Q: Question 25 The correct order of increasing strength of adsorption is Alkanes >> Esters >> Ketones…
A: Mixture is formed by mixing different substances physically.
Q: Use the symmetry criteria for polarity and chirality of molecules and refer to the structures of…
A: The compound which doesn't possess plane of symmetry , then this compound is considered as chiral…
Q: If H=-180kJ/mol and S=45J/k, then the reaction will be… a) Spontaneous at all temperatures b)…
A: Reaction spontaneity ∆H ∆S reaction spontaneity + value - value non spontaneous at all…
Q: What is the Ecell (in V) for the reaction below wth the given concentrations at 48.0 oC? 4 H+ (aq)…
A: Here we are required to find the Ecell of the reaction .
Q: estion 18 The relative solubility of solute in both the phases determines the Rate of disappearance…
A: :Mobile phase is a substance that can move through carrier.
Q: How many stereoisomer(s) is/are possible in the structure below: X a.2 O b.3 C. 1 O d.4 но ОН
A:
Q: A solution of toluene (C,H₂).in 301 g of cyclohexane (C₂H₁₂) has a boiling point of 90.30 °C. What…
A: We have to predict the amount of toluene in solution.
Q: The specific Rotation of (R)-carvone (at 20 deg) is -61. A chemist prepare a mixture of (r)-carvone…
A:
Q: Rank the numbers from greatest to least: 5.67 x 102 3.45 x 104 4.56 x 103 1.23 x 106 2.34 x 105
A:
Q: An atomic orbital represents: O the region of electron density for a covalent bond O a fixed path…
A: Here we have to define atomic orbital.
Q: Draw the structure of the monomer from which the polymer can be synthesized.
A: A polymer is a compound that is formed by the repetition of a monomer. The polymer can have one or…
Q: What is the pH of 0.15 M acetic acid to 1.00 L of which 2.70 g of sodium acetate NaCH3CO2, has been…
A: Molarity of acid = 0.15 M Volume of sodium acetate = 1.00 L Mass of sodium acetate = 2.70 g Ka of…
Q: Based on the energy level diagram to the right, which transition corresponds to emission of a photon…
A: From the given energy diagram, which transition having longest wavelength ?
Q: Q_/ The acid-dissociation constant for chlorous acid, HClO2, at 25C° is 1.0×10^-2 . Calculate the…
A: Given -> Concentration of acid = 0.100 M Acid dissociation constant = Ka = 1.0 × 10-2
Q: true or false osmium tetroxide is an oxidizing agent.
A: Osmium tetroxide is an oxidizing agent . This statement is true of false given below with…
Q: Given the reaction 2 Cl2 (g) + 2H₂O(g) → 4HCl(g) + O2(g) What is AHO of this reaction? AHO Cl₂ = 0…
A:
Q: How many protons (P), neutrons (n) and electrons (e) are present in 18173Ta-3 ? a. 76 p 105 n 73 e…
A:
Q: Identify the isoelectronic elements. O Li, Na, K, Rb*, Cst ONe, Ar, Kr, Xe, He ON3, S2, Br, Cst,…
A: Here, we have to find the isoelectronic elements from the given options.
Q: Give the ground state electron configuration for the ion of Cs forms. O [kr]5s²4d105p6 O…
A:
Q: You were tasked to prepare nitric acid with a concentration of 1.64 M. What will be the volume (in…
A:
Q: How many grams and moles of glucose, C6H12O6, should be dissolved in 250 mL of solution to make a…
A:
Q: How many protons (P), neutrons (n) and electrons (e) are present in 18173Ta-3 ? a. 76 p 105 n 73 e…
A:
Q: 4 b. The following system establishes an equilibrium at 545 K NH₂ (8) + H₂S (8) AND > 0 NH₂HS (s)->…
A: 4) Le Chatelier's principle According to the Le Chatelier's principle when a change in concentration…
Q: Which gas below would be traveling with the greatest velocity at room temperature? 1.O2 2.CH4 3.…
A:
Q: Which is the correct order of reactivity for the following alkyl bromides in an El reaction?
A: Given : structure of alkyl halide. Reactivity of alkyl halide is directly proportional to the…
Q: Fe3Br8 – an important source of potassium bromide is produced through the following two-stage…
A: Given following balanced Chemical equations - Stage 1: 3 Fe + 3 Br2 ----> 3 FeBr2 Stage 2: 3…
Q: Identify the RELATIONSHIP of the two structures: Me a. different compounds O b.identical C.…
A: -> For finding relationship between two compounds we find configuration at each chiral center .…
Q: . Write the mechanism for the reaction of 2-Methyl-1-phenyl-2-propanol with HBr.
A:
Q: Choose the ground state electron configuration for Cr³+. O [Ar]4s23d6 O [Ar]4s23d¹ O [Ar]3d3 O [Ar]…
A: Here first we will write electronic configuration of Cr . In this electronic configuration instead…
Q: Question 17 Which of the following solutions will have the lowest freezing point? O 0.010 m Li2SO4…
A: Depression in freezing point is a colligative property which depends on the relative number of…
Q: Number of atoms of Carbon in 15 moles of C4H10 are: 90.33x10^23 361.32x10^23 O 6.23x10^23
A:
Q: nts Ionisation Energy: It is the energy required to an electron from an atom in the given graph,…
A: Ionization energy is the amount of energy required to remove an electron from the outer most shell…
Q: Section V: Chemical Equations 1. Part A: The molecular reaction occurring between the components in…
A: In the net ionic reaction the spectator ions i.e ions which do not participate in reaction is not…
Q: In the fluorination reaction of butadiene, the butadiene is adsorbed onto a catalyst C. To test the…
A: We are given pressure(kPa) and volume(cm3) data for the adsorption of butadiene on the surface of…
Q: A sample of solid gold has a density of 19.3 g/cm3 and a volume of 2.0 mL. What is the mass of the…
A: Density of solid gold = 19.3 g/cm3 Volume of solid gold = 2.0 ml Mass of the sample gold = ?
Q: Chemistry Identify unknown using NMR spectra.
A: A question based on NMR spectroscopy that is to be accomplished.
Q: What mass in grams of glucose (MM = 180.2 g/mol) would need to be dissolved in 100.0 g of water to…
A:
Q: Discuss the fundamentals of fluorescence
A:
Q: The monosaccharides depicted below are: НО НОН H Но H `OH ОН ОН O Diastereomers O Enantiomers O…
A: Non-mirror image stereoisomers are diastereomers. They have the same configuration at one or more…
Q: SR)4] comp
A: The count in d electron can helps to find the oxidation states, reaction rates and magnetic…
Q: 4.57 g of C12H22O11 were dissolved in 100 g of H₂O. Density of solution is 1 g/ml. Calculate the…
A:
Q: You are given a mixture containing a pair of enatiomers: A and B. Careful measurements that the…
A:
Q: Calculate the mass percent.by volume of 76.6 g of glucose (C₂H₁₂O MM = 180.2 g/mol) in 325 mL of…
A:
Q: Identify the orbital. O dxy orbital O dyz orbital O px orbital O Pz orbital O py orbital x
A: Types of orbitals, 1) s orbital - spherical shape 2) p orbital - dumb bell shape • Px orbital…
Q: The Henry's law constant at 25 °C for CO₂ in water is 0.035 M/atm. What is the solubility of CO₂, in…
A:
Q: For the redox reaction given below complete the following: Cr2O7-2+ H+ +FeCl2→ Cr+3 + FeCl3 + H2O…
A: Redox reaction : The reaction in which oxidation and reduction takes place simultaneously is called…
Q: Identify the elements correctly shown by decreasing radii size. Cu²+ > Cut OK+ > K ON ³-> N ON>№ ³-…
A:
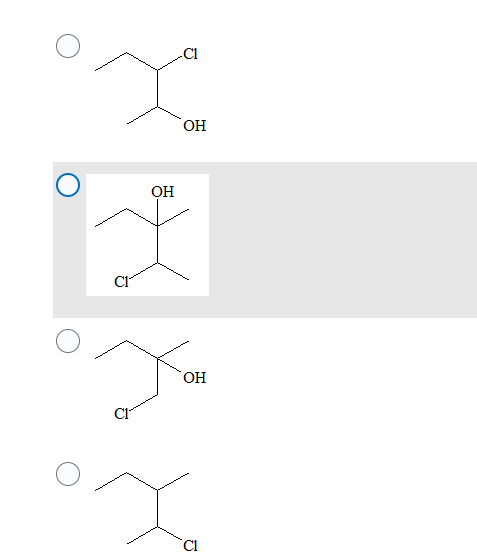
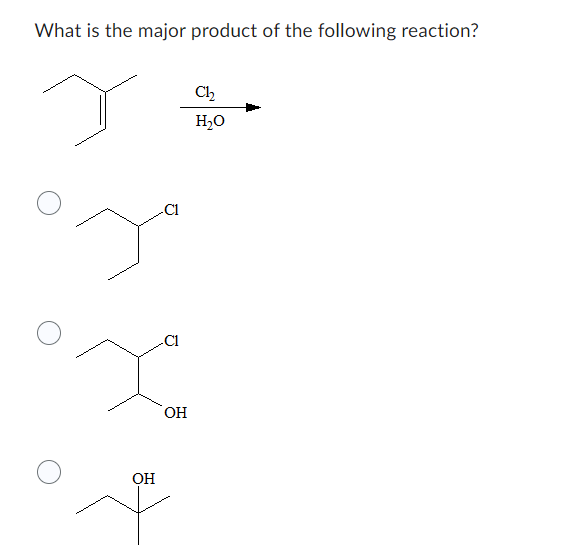
Step by step
Solved in 2 steps with 2 images

- (CH3)3CHOH is reacted with CH3-O-C--C6H5 in a transesterification reaction. What is the M+ of the product? || O 102 80 178 None of theseThe following product can be synthesized from the reaction of an alkene with Hg(OAc)2 followed by reduction with NaBH4. Draw all possible alkenes that will give the product shownIndicate the product of each reaction below
- 2. How many substitution product/s is/are formed when metabromo anisole is treated with ammonia?A. 0-no reactionB. 1C. 2D. 3Predict the major products formed when 2- methyl-1-butene reacts with: H2, Pt/25°C. Show the reaction mechanism the given alkene reactionsGive the following reaction A+3B --->2C.
- I recovered 3 mL of water, how many grams of water was produced in the reaction please show how? Cyclohexanol can attack the carbocation intermediate formed during the synthesis of cyclohexene. Draw this side product with the molecular formula C12H22O.Fill in the blank: The reaction of alkene with peroxyacid (RCO3H) is: A) a radical mechanism with carbon radical intermediate B) a concerted reaction C) a polar mechanism with carbocation intermediateIs the reagent for all elimination reactions conc. H2SO4? or just the reactions ones with OH?

