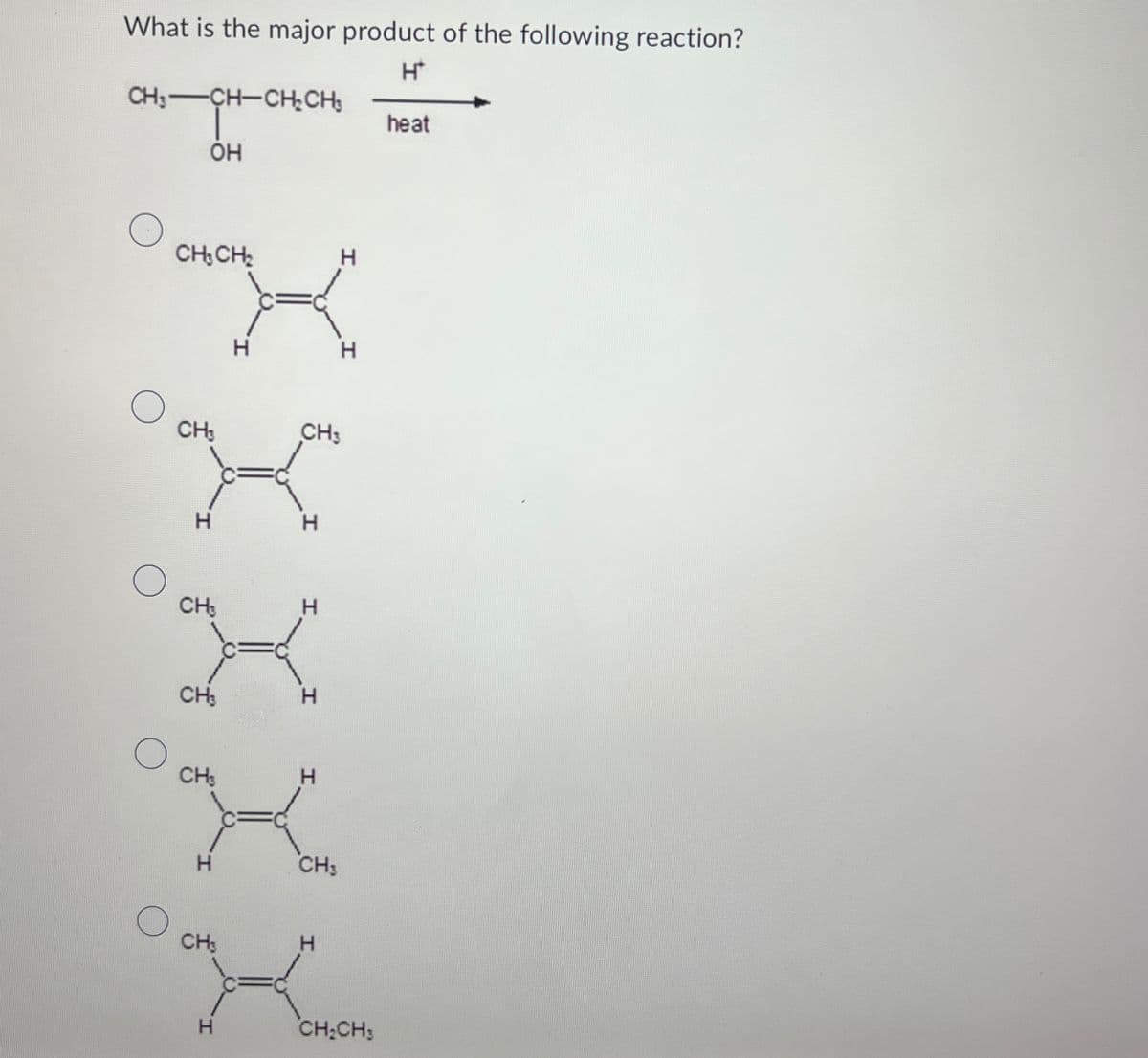
What is the major product of the following reaction? CH-CH-CH₂CH₂ -CH-CH-CH₂ H* heat OH CH3CH2 H H H О CH3 CH H H О CH3 H CH3 H CH H I. H CH3 CH H H CH2CH3
Q: Please help with a through c Part a) A student adds some granular zinc to a blue solution with…
A: Blue colour solution is of copper(II) sulfate. The Copper form will be the Reddish Brown deposit and…
Q: How many grams of CO2 gas occupy 64.3 L at STP
A:
Q: Question 9 Propose a chemical structure for the name below. Make sure to clearly define the…
A: Step 1: Step 2: Step 3: Step 4:
Q: calculate the pH of solution made by dissolving 3.5 grams of potassium oxide in 0.5L of pure water?
A: Given: massK2O=3.5g;MK2O=94.2g/mol;V=0.5LStep 1: Write the balanced chemical…
Q: The great work expert Hand written solution is not allowed please.
A: Answer to this question can be calculated by two methods .1st is direct assumption.As we know ideal…
Q: Draw the structure of the major organic product of the reaction. In those cases where there are two…
A: Step 1: Step 2: Step 3: Step 4:
Q: Calculating the pH of a 0.1 L aqueous solution of HA 2.50 10 - 3M (Ka = 1.74 10 -6) requires…
A: To calculate the pH of the given aqueous solution of HA, we'll use the acid dissociation constant…
Q: None
A: Step 1: Using the formula M1V1 = M2V2, we can get M2 by: M2=M1V1/V2 Step 2: Substitute the values to…
Q: Please give the major product of any of the given letter a, b, c or d. Explain the mechanism
A: 1) a)In the given Image, the overall reaction represents the Williamson ether synthesis, where an…
Q: please answer in text form and in proper format answer with must explanation , calculation for each…
A: Given: 298KA. The cell is under standard conditions.B.[] = 3.0 M[] = 0.010 M[] = 0.10 MC.[] = 0.010…
Q: A chemistry graduate student is given 250. mL of a 0.50M dimethylamine ((CH3)2NH) solution.…
A: Given:…
Q: Please don't provide handwritten solution ....
A: Approach to solving the question: - Detailed explanation: Given above Examples: - Key references: -
Q: Question 21 The reaction below proceeds with the formation of ONLY ONE (S)-isomer. Choose the…
A:
Q: Please don't provide handwritten solution. Show how the following synthesis can be performed.
A: Step 1: Step 2: Step 3: Step 4:
Q: In the following FTIR Spectrum, what functional group can you recognize?
A: In the IR spectrum there are two regions.The region which has wavenumber < 1500 cm-1 is called…
Q: Organic Chemistry problem. Please help solve. Suggest reaction that will allow the transformation…
A:
Q: 1. Predict the product of the following reaction and show the mechanism. a) 1.1 equiv LDA -78 C OH…
A: 1. Starting Material: 2-Keto-3-methylbutane - Draw the structure of 2-keto-3-methylbutane (also…
Q: None
A: Given: massC3H8=1.43lbs;V=5.3gal;T=16.58°C1gal=3.76L;1lbs=453.6g;MC=12.01g/mol;MH=1.01g/molStep…
Q: The following reaction has K= 3.03 when at equillibrium: CO(g) + 2H2(g) --- CH3OH(g) The reagent…
A: To calculate the final (equilibrium) concentrations of CO, H2, and CH3OH, use ICE table to relate…
Q: Use a mechanistic approach to provide the major product(s) for each of the following reactions. Be…
A:
Q: 4. (5 pts) Extra credit Explain why the "benzyne" reaction (Hint: look at one of the major products)…
A: References; Cooper, M. M., Stowe, R. L., Crandell, O. M., & Klymkowsky, M. W. (2019). Organic…
Q: 7 Draw the mirror image for each molecule. OH HC / COOH H OH CHO OH H-C-OH H CH3 CH₂OH 8 Determine…
A:
Q: 4NH3+502 → 4NO + 6H20 The standard enthalpies of formation for NH3, NO and H2O are 294.1 kj/mol,…
A:
Q: 16.36 For the reaction 2A+B+C+D+ 2E, data for a run with [A], 800 mmol/L and [B] = 2.00 mmol/L are…
A: Step 1:Step 2: Step 3: Step 4:
Q: Draw a structural formula for the alkene you would use to prepare the alcohol shown by…
A: Step 1: Step 2: Mechanism Step 3:Step 1: HydroborationThis step involves the reaction of the alkene…
Q: None
A: Step 1: Step 2: Step 3: Step 4:
Q: Draw the product of this reaction. Ignore inorganic byproducts. 1. NaNH2 H Q Problem 9 of 11 Atoms,…
A: Step 1: Step 2:The lone pair of electrons on the nitrogen atom of NaNH2 acts as a base and attacks…
Q: Help me solve this.
A: Given: pH=3.444;[HA]=0.222M;T=25°C=298.15KStep 1: Write the dissociation of…
Q: Question 27 Predict the FINAL (?) product (or a mixture of products) for the following synthetic…
A:
Q: Draw the product of the following epoxide reaction, including the stereochemistry at any stereogenic…
A: Step 1: Step 2: Step 3: Step 4:
Q: Question 28 Predict the FINAL (?) product (or a mixture of products) for the following synthetic…
A:
Q: ABCD Question 6 Please predict the product for each of the following reactions. Make sure to clearly…
A: Two major steps occur in the ring opening of epoxide in acidic conditions.First, epoxide gets…
Q: CIO (aq) + Cr(OH)3(s) CrO42(aq) + Cl¯(aq) basic What is the standard cell potential, E° cell, for…
A: Step 1:The standard electrode potential, often referred to as standard potential, indicates the…
Q: Question 22 Draw the enantiomer for the compound below: Me OMe CI A MeO Me B Me. OMe с D MeO Me Me.…
A: Step 1:Answer: AExplanation:We know that enantiomers are actually a mirror image of a chiral…
Q: To preview image click here Give a curved arrow pushing mechanism for the following reaction,…
A: Step 1:Carboxylic Acids.1) Draw the general structure of a carboxylic acid Step 2:2) T/F -…
Q: Identify the Haworth structure showing the ẞ-isomer of the following Fischer projection. H (Hint:…
A: Beta form -OH group is above the plane, here there are two form in which OH is above the palne . A…
Q: Please write out the mechanism for the acid (WHICH IS 3-BROMOBENZOIC ACID) reacting with anilide.…
A: Step 1: Step 2: Step 3: Step 4:
Q: 13 Draw the complete arrow pushing mechanism for the following reaction. Indicate the Lewis acid and…
A: References Smith, M. B. (2020). March's advanced organic chemistry: reactions, mechanisms, and…
Q: None
A: Here are the steps involved in the reaction:Step 1: Aldol condensation of 1 molecule of acetone and…
Q: None
A: Answer: O-C≡OClF5Hybridisationspsp3d2Electronic geometryLinearOctahedralMolecular…
Q: Predict the products of the following reactions. Show stereochemistry when applicable.
A: When bromine water (aqueous solution of bromine, Br2) reacts with an alkene, a bromine addition…
Q: Please answer the following chem question correctly
A:
Q: Question 3 Which set of Newman projections represents correct the most stable and the least stable…
A: Step 1: Step 2: Step 3: Step 4:
Q: Propose a chemical structure for the name below. Make sure to clearly define the stereochemistry…
A:
Q: Supply the missing isotope in the nuclear equation below. When entering isotopes, use the text…
A:
Q: Draw the major organic product(s) of the following reactions including stereochemistry when it is…
A: Step 1:In the presence of H₂SO₄/HgSO₄, the alkene (CH₃CH₂C=CCH₃) undergoes protonation. The acidic…
Q: The acid dissociation constant K of hypobromous acid (HBrO) is 2.3 × 109. Calculate the pH of a 2.8M…
A: Step 1: Data givenThe concentration of hypobromous acid (HBrO) solution = 2.8 MThe acid…
Q: Question 4 Which set of Newman projections represents correct the most stable and the least stable…
A: Step 1: Step 2: Step 3: Step 4:
Q: A solution contains 0.0150 M Pb2+(aq) and 0.0150 M Sr²+(aq). If you add SO2 -(aq), what will be the…
A:
Q: The corrected cell potential for the following galvanic cell was determined to be (1.62x10^0) V.…
A:
Please don't provide handwritin solutions
Step by step
Solved in 2 steps

- (CH3)3CCH=CHCl (1)NaNH2 -----> (2)H2O Which of the following is the product for the given reaction? a) (CH3)3CCH≡CH b) (CH3)3CCH=CH2 c) (CH3)3CCH=CHOH d) (CH3)3CCH=CHNH2illustrate how each reactant appeared prior to the reaction and how the product appeared after the reaction. 2Na+2H₂O --> 2NaOH+H₂What reagent is necessary to complete the reaction? CH3-CH2-CH-C-OH ? CHỊCH,CH C-0- Na* CH3 CH3 NaO O NaCl O Na O NaOH + H₂O
- Bicyclo[2.2.1]heptan-7-one + PCC (in CH₂Cl₂) => A.) Bicyclo[2.2.1]heptan-7-ol B.) Bicyclo[2.2.1]heptanoic acid C.) Cyclohexanecarbaldehyde D.) All the given choices are possible products E.) No reactionRank the compounds in each set in order of increasing acid strength.(a) CH3CH2COOH CH3CHBrCOOH CH3CBr2COOHWhich of the following will not undergo oxidation? a 1-methylcyclohexan-1-ol b Cyclohexanol c Hexan-2-ol d Hexan-1-ol
- Choose the correct product to complete the following reaction: HNO3 + H2O → ________ + NO3- OH- H+ K+ H3O+Draw all products of the reaction of (1S,2R)-1-bromo-1,2-dimethylcyclohexane in 80%H2O/20%CH3CH2OHat room temperature..When (s)-3-bromopent-1-ene is heated in water, which of the following compounds is not produced? A )(S)-pent-1en-3-ol B) ( R)-pent-1-en-3-ol C )pent-4-en-1-ol D)(E)-pent-2-en-1-ol E)(Z)-pent-2-en-1-ol
- What ketone could be used to prepare 3-methylpentan-3-ol via a Grignard reaction with CH3MgBr? a.) Pentan-2-oneb.) Pentan-3-onec.) Pentan-4-one d.) acetoneWhat product is formed when 3-methyl-2-cyclohexenone reacts with each of the following reagents? a. CH3MgBr followed by H3O+ b. (CH3CH2)2CuLi followed by H3O+ c. HBr d. CH3CH2SHDraw the products formed when CH3CH2C ≡ C−Na+reacts with each compound. a. CH3CH2CH2Brb. (CH3)2CHCH2CH2Clc. (CH3CH2)3CCld. BrCH2CH2CH2CH2OHe. ethylene oxide followed by H2Of. propene oxide followed by H2O



