Α ABCD Br Question 7 Please predict the product for each of the following reactions. Make sure to clearly indicate the correct stereochemistry for each compound: 1. NaOH 2. H₂O 1. NaSMe 2. H₂O HBr HCI 10 H₂O* H₂O H₂O CH3CH2OH 5 1. NaCN 2. H₂O 1. PhMgBr 2. H₂O 1. LiAlH4 2. H₂O 1. NaOEt 2. H₂O A B с D Br HO OH OH OH Br Br
Α ABCD Br Question 7 Please predict the product for each of the following reactions. Make sure to clearly indicate the correct stereochemistry for each compound: 1. NaOH 2. H₂O 1. NaSMe 2. H₂O HBr HCI 10 H₂O* H₂O H₂O CH3CH2OH 5 1. NaCN 2. H₂O 1. PhMgBr 2. H₂O 1. LiAlH4 2. H₂O 1. NaOEt 2. H₂O A B с D Br HO OH OH OH Br Br
Chapter25: Biomolecules: Carbohydrates
Section25.SE: Something Extra
Problem 49AP
Related questions
Question
Please answer the following chem question correctly
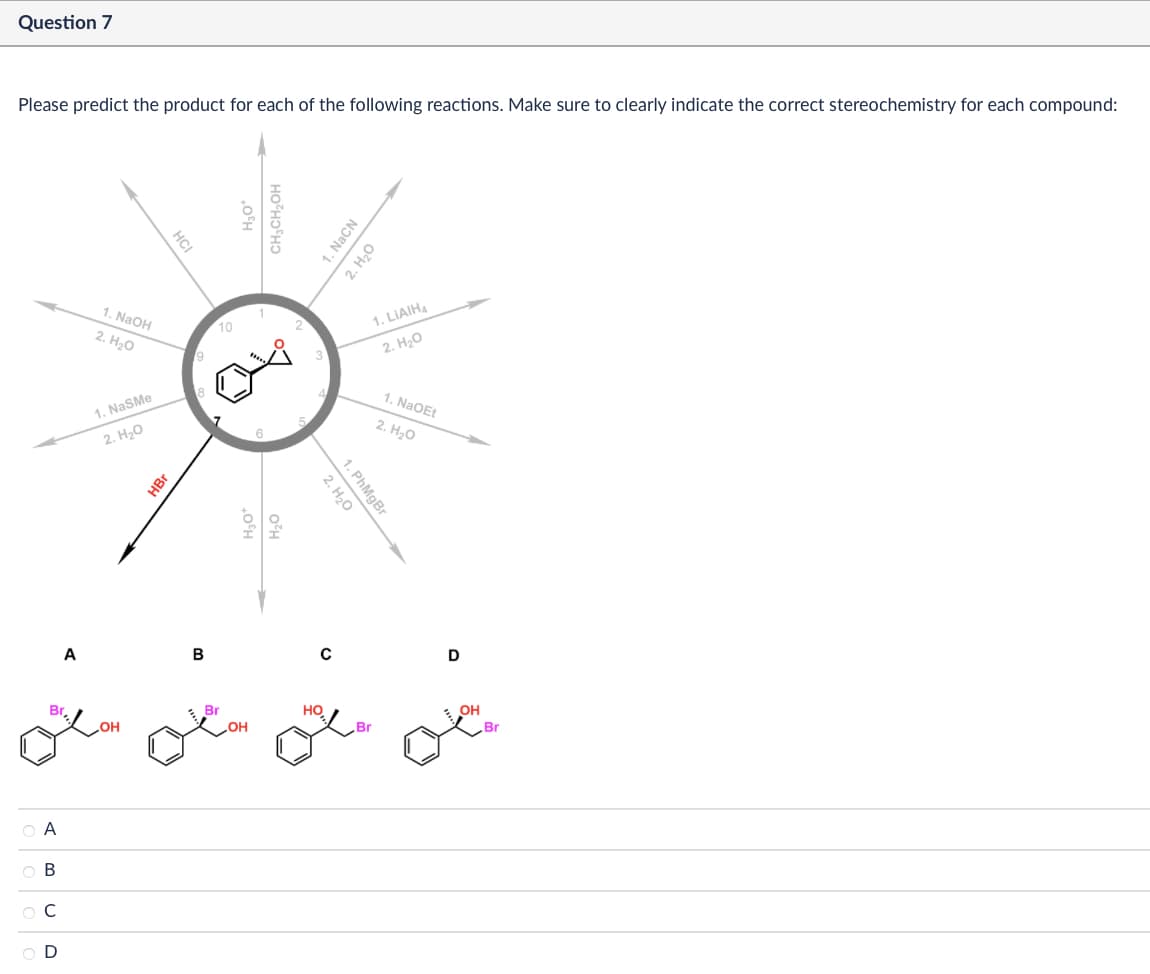
Transcribed Image Text:Α
ABCD
Br
Question 7
Please predict the product for each of the following reactions. Make sure to clearly indicate the correct stereochemistry for each compound:
1. NaOH
2. H₂O
1. NaSMe
2. H₂O
HBr
HCI
10
H₂O*
H₂O
H₂O
CH3CH2OH
5
1. NaCN
2. H₂O
1. PhMgBr
2. H₂O
1. LiAlH4
2. H₂O
1. NaOEt
2. H₂O
A
B
с
D
Br
HO
OH
OH
OH
Br
Br
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

