Q: 3. A solution is prepared by dissolving 35.0 g of hemoglobin in enough water to make up 1.00 L in…
A:
Q: A voltaic cell is 'constructed from a standard Mn2+ = -1.180V) and a standard I2 I half cell (E° Mn…
A: Reduction occurs at negative electrode (cathode) as here positive ions gain electrons. Oxidation…
Q: 11. Devise a synthesis of each compound from benzene, any organic alcohols having four or fewer…
A: Detail step by step synthesis reaction is given below by which we get the target molecule
Q: Will the following reaction be spontaneous under standard conditions? Zn(s) + 2H*(aq) → Zn+2(aq) +…
A:
Q: Effect of temperature on ethanol productivity for Saccharomyces cerevisiae is given below:…
A:
Q: It takes 238. kJ/mol to break a carbon-iodine single bond. Calculate the maximum wavelength of light…
A:
Q: Molecule SO2 Lewis Structure 3D representation Bond Angles Polarity (if yes, show dipole) 6+2(6) ö=s…
A:
Q: What is the pH of a 0.100 M solution of generic diprotic acid, which has a Kat of 2.1 x 10-5 and Ka2…
A: Given: The Ka1 of a generic diprotic acid = 2.1×10-5 The Ka2 of the acid = 3.1×10-9 We have to…
Q: The graph below shows the distribution of molecular velocities of four gases, NH3, O2, CO2 and Xe,…
A: In the above question, distribution curve of four gases are given. Four gases have different molar…
Q: A 50.0 mL HCI is pipetted out from a 1000-mL 0.0250 M stock solution. To this 50.0mL aliquot, a…
A: Given-> Volume of HCl pipette out = 50.0 ml Volume of stock solution = 1000 ml Molarity of stock…
Q: 3. Ammonia and phosphoric acid solutions are used to produce ammonium hydrogen phosphate fertilizer.…
A: Given-> Molarity of NH3(M1) = 14.8 M Molarity of H3PO4(M2) = 12.9 M Volume of H3PO4(V2) = 1000 L…
Q: solution is made initially with 0.510 M HIO₃ (Kc = 0.170). Once the equilibrium below is…
A: Equilibrium can be defined as a state of reaction in which rate of forward reaction become equal to…
Q: A freshly isolated sample of 0Y was found to have an activity of 7.7 x 105 disintegrations per…
A: All nuclear reactions follow first order kinetics. For first order reaction- ln (At/Ai) = -kt At:…
Q: 10. Alkyl diazonium salts decompose to form carbocations, which go on to form products of…
A:
Q: *9. Work out the spin arrangements and splitting patterns for the following spin system: HA HB…
A: As per bartleyby guidelines i answered only first question so please don't mind and post other…
Q: g) (aq) Enter electrons as e". Al half cell (E° red = A voltaic cell is constructed from a standard…
A: Given: A voltaic cell is constructed from a standard Al3+|Al half-cell and a standard F2|F-…
Q: The rate constant for this first-order reaction is 0.0330 s at 400 C. A products After how many…
A: Given reaction: A → products The rate constant of this reaction = 0.0330 s-1 We have to calculate…
Q: Choose the correct order based on the given criterion per line for comparative 1.5 M aqueous…
A: ->Acids has pH less than 7 , stronger acid has less pH. -> Base has pH greater than 7 ,…
Q: H3CO OCH3 heptanodioato de dimetilo
A: Solution -
Q: Question 37 of 50 Although AGCN(s) is very insoluble (Ksp 2.20 x 10 1), it will dissolve in higher…
A:
Q: Electron configuration
A:
Q: Draw a voltaic cell of the following reaction below (please include anode, cathode, and flow of…
A: This question is related to electrochemical cells. Voltaic cell is an electrochemical cell in which…
Q: он `NH2
A:
Q: Caper Give the starting alkene and regents needed to make the epoxide shown.
A: Note : Since you have posted multiple questions, we are entitled to answer the first only. Please…
Q: Determine which of the following pairs of reactants will result in a spontaneous reaction at 25…
A: These type of questions are solved using electrochemical series. It is a series in which elements of…
Q: 4. If the protein reacts with the blue (580 nm) dye to form a purple color (550 nm) complex, draw…
A: Protein reacts with blue dye to form a purple complex. We have to draw diagram to show how…
Q: Calculate the solubility of Cu(OH)2 in an aqueous solution of KCN 0,10 mol/L.
A:
Q: A C5Hg alkyne is reacted with Lindlar's catalyst and hydrogen gas to yield product with the…
A:
Q: Select the single best answer. For this pair of elements (F and Se), predict which one has more…
A:
Q: In a complexometric titration experiment: Determination of Water Hardness, solve for the missing…
A: Each mole of Ca2+ reacts with 1 mol of EDTA. Also, Moles = Mass / Molar Mass
Q: QUESTION 9 Click the peak that corresponds to the hydrogen on the carbonyl group. H H H. 1.0 2|0 2.0…
A: NMR spectroscopy helps us to find the structure of the unknown organic compound on basis of the…
Q: For each of the species below, identify any cyclic conjugated system, then: A. Determine the number…
A: For aromatic anti-aromatic cyclic and planar structure is important condition....
Q: A sample of krypton gas at a pressure of 1.02 atm and a temperature of 167° C, occupies a volume of…
A:
Q: Irn/takeAssignment/take Lv2 | Online teaching and X Activity Series of Metals…
A: Here, we have to find whether a reaction will occur or not when Ag(s) and hydrobromic acid are…
Q: Briefly describe how a phase-contrast microscope work and the kind of image that it produces. Give a…
A: Phase-contrast microscopy generates an image by using the different refractive indexes and densities…
Q: 6. Starting with benzene and any other reactants you need, show how you would synthesise the…
A: There are various types of palldium catalyzed coupling reaction- 1- suzuki coupling 2- sonogashira…
Q: Q11. Provide a mechanism that leads to the major product: NO2 CH NH2 Br Q12. Propose a set of…
A: Q 11. Aromatic Sn2. Q 12. ( see below).
Q: Calculate the nuclear binding energy (in J) and the binding energy per nucleon for the following…
A: Nuclear chemistry is the branch of chemistry in which we deal with nuclear fusion and fission…
Q: A. Explain, how can heating is used as remedy for condensation? B. SKetch a cross-section view of…
A:
Q: For carbon monoxide, the characteristic vibrational temperature is 3084K and the characteristic…
A:
Q: How does water activity affect enzymatic reaction?
A: The action of enzymes to carry out a reaction is known as enzymatic action. It depends on the…
Q: Name the following metal complexes or complex ions: formula name [FeCi, (co).]
A: Complex ion consist of metal and several ligand within the coordination sphere Here we are required…
Q: On the basis of the 18-electron rule, identify the first-row transition metal for each of the…
A:
Q: 1.) Compare the relative atomic radius of iron and carbon. Fe: С:
A: Since you have asked multiple questions, we will solve the first one for you. For remaining…
Q: In each pair of isotopes shown, indicate which one you would expect to be radioactive. 20 17 40 45…
A: Radioactivity can be defined as the process in which there is spontaneous emission of radiation in…
Q: Use the data to find AHn for the reaction. 2 NO2(g) =2 NO(g) + O2(g) Express your answer in…
A:
Q: Which of the following structures would be most readily reduced by a hydride delivery agent, such as…
A: ->NaBH4 and LiAlH4 are reducing agent . These are reducing agent because produce H- nucleophile .…
Q: One of the most promising possibilities for commercial fusion is combining deuterium (amu 2.014102…
A: Given that, the commercial fusion is combining deuterium with tritium to form Helium-4 and a…
Q: QUESTION 5 If 0.91 liters of 1.6174 M strontium nitrate is diluted to a final volume of 926.7858 mL,…
A: Initial volume of solution = 0.91 L = 0.91 * 103 mL initial molarity of solution = 1.6174 M Final…
Q: Which ion will be attracted to a magnetic field? A) C2* B) 02- C) F* D) Be E) All of the above
A: The ion which has unpaired electron in the orbital then it is called as paramagnetic substance. The…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

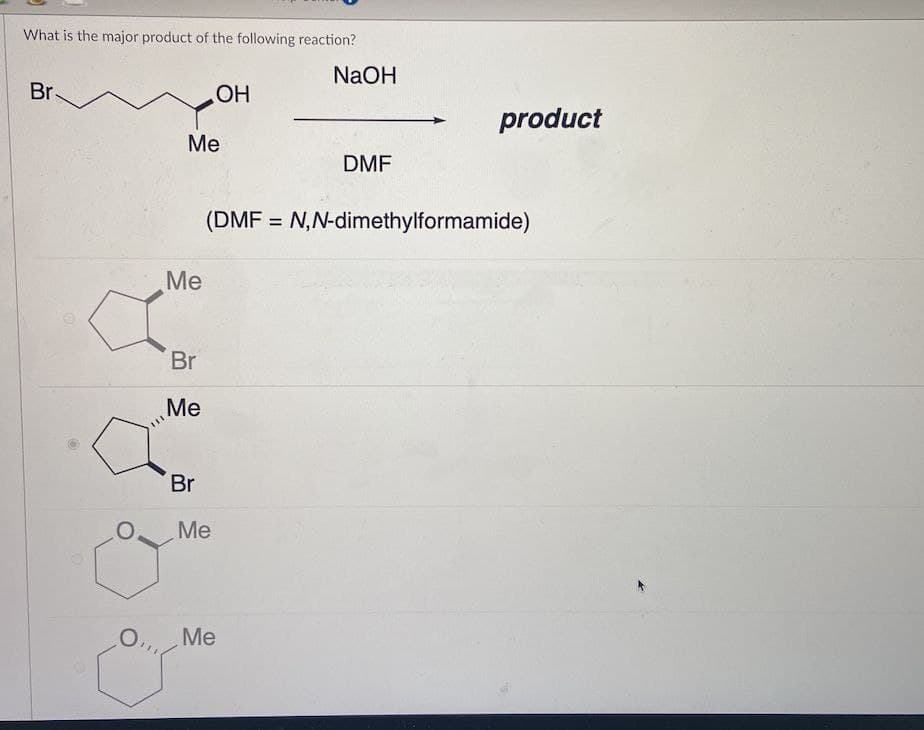
- Predict the major products of the following reactions. (a) the tosylate of cyclohexylmethanol + excess NH3(b) n-butyl tosylate + sodium acetylide, H¬C‚C:- +NaWhich product would likely be the major product for the following reaction?a. Bb. Cc. Ad. DChoose the answer that best represents the products of the following reaction. Potassium carbonate is reacted with phosphoric acid. A. 2 K3PO4 (aq) + 3 H2O (l) + 3 CO2 (g) B. K2CO3 + H3PO4 C. 3 K2CO3 + 2 H3PO4 D. 2 K3PO4 (aq) + 3 H2CO3 (aq)
- What are the products for the following ozonolysis reaction? A) I and II B) II and III C) I and III D) I and IVWhat are the products of the reactions below? d and e pleaseee. thx!Write the expected product for each of the following reactions: Diphenyl sulfide + 1 eq. NaOCl -------------> ? 1,2-Epoxy-3,3-dimethylbutane + 1 eq. CH3CH2SNa then CH3Br -------------> ? 1,2-Epoxy-3-phenylpropane + HCl ------------> ?
- write the major products for the following reactions b) →2.CH3CH2CH2OT1. c) CH3CO3H CH3ONa H-1,H2O b) CH3CO3H CH3ONa H-1,H2ORank the species below in order of increasing nucleophilicity in protic solvent. I. H2O II. CH3S— III. CH3COO— IV. t-BuO— I, II, IV, III I, III, II, IV I, III, IV, II I, II, III, IVWhat is the product of the reaction P4O6 + H2O? A) H3PO4 B) H2PO4 C) H3PO3 D) H2PO3 E) H2P4O7
- What would be the majority product of the following reaction?: [Pt(NH3)4]2+ + 2 Cl- → ? Select one:a. No reaction because chloride is not a good inbound binder b. trans-[PtCl2(NH3)2] c. cis-[PtCl2(NH3)2] d. [PtCl(NH3)3]+(2R, 3S) -3-phenylbutan-2-ol tosylate undergoes E2 removal when treated with sodium ethoxide.Draw the structures of the products obtained (one majority and one minority). Use Newman's projections for this reactive in this deduction.Show how you would convert propan-1-ol to the following compounds using tosylate intermediates. You may use whatever additional reagents are needed.(a) 1-bromopropane (b) propan-1-amine, CH3CH2CH2NH2

