Use the data to find AHn for the reaction. 2 NO2(g) =2 NO(g) + O2(g) Express your answer in kilojoules per mole. The data in the table show the equilibrium constant for this reaction measured at several different temperatures. > View Available Hint(s) Temperature Kp 170 K 3.8 x 10-3 ? 180 K 0.34 190 K 18.4 ΔΗ kJ 200 K 681 Submit • Part B Use the data to find ASm for the reaction. Express your answer in joules per mole.
Use the data to find AHn for the reaction. 2 NO2(g) =2 NO(g) + O2(g) Express your answer in kilojoules per mole. The data in the table show the equilibrium constant for this reaction measured at several different temperatures. > View Available Hint(s) Temperature Kp 170 K 3.8 x 10-3 ? 180 K 0.34 190 K 18.4 ΔΗ kJ 200 K 681 Submit • Part B Use the data to find ASm for the reaction. Express your answer in joules per mole.
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter13: Spontaneous Processes And Thermodynamic Equilibrium
Section: Chapter Questions
Problem 19P
Related questions
Question
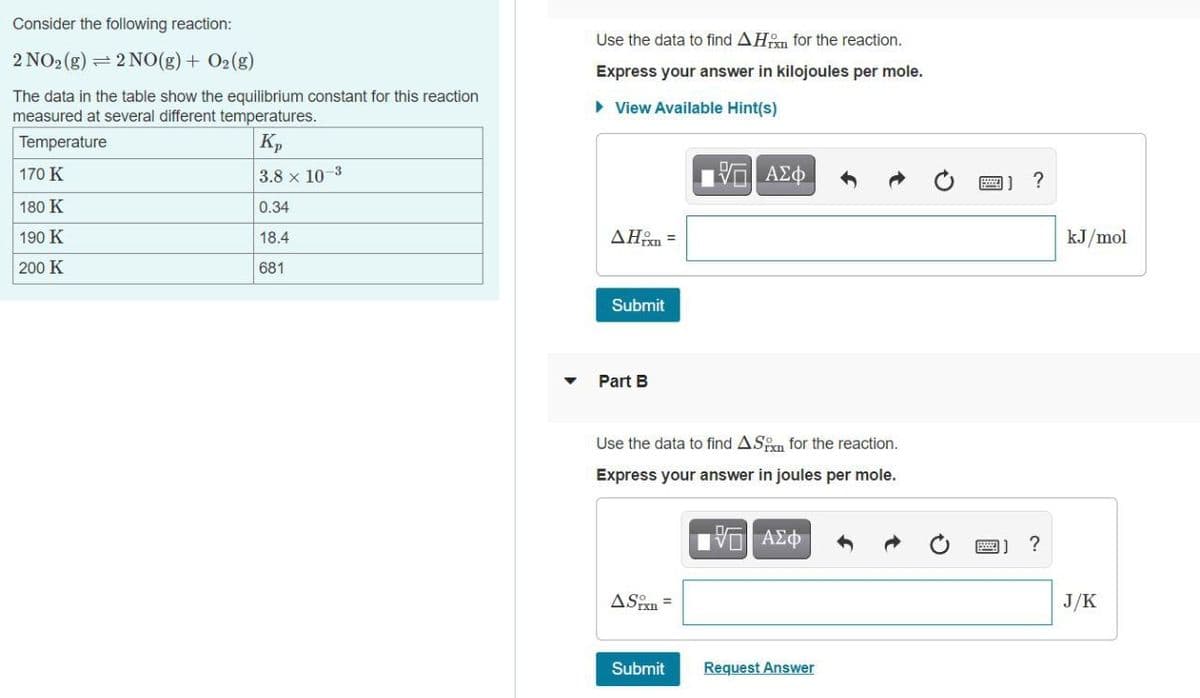
Transcribed Image Text:Consider the following reaction:
Use the data to find AHn for the reaction.
2 NO2 (g) = 2 NO(g) + O2(g)
Express your answer in kilojoules per mole.
The data in the table show the equilibrium constant for this reaction
measured at several different temperatures.
• View Available Hint(s)
Temperature
Kp
170 K
3.8 x 10-3
?
180 K
0.34
190 K
18.4
AHon =
kJ/mol
200 K
681
Submit
Part B
Use the data to find ASn for the reaction.
Express your answer in joules per mole.
?
ASin =
J/K
Submit
Request Answer
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning