Q: A raindrop has a mass of 50. mg and the Pacific Ocean has a mass of 7.08 × 10° kg. Use this informat...
A: 1 mole = avogadro number of particles = 6.022 x 1023 particles Given that 1 rain drop = 50 mg ...
Q: What would be net equation be for sodium acetate with boric acid added to produce a solution? (buffe...
A:
Q: . Calculate hHg (Density water = 1.00 g/mL Density of Hg = 13.6 g/mL) %3D %3D
A:
Q: Study the chemical equations that show how each substance behaves when dissolved in water. Then clas...
A: According to Bronsted- Lowry concept of acid and base , acid is the substance that loose H+ where ba...
Q: Indicate how the concentration of each species in the chemical equation will change to reestablish e...
A: According to the Le Chatelier's Principle : for a given chemical reaction at equilibrium, a change i...
Q: 1 O I and Il only O all of them O III only I, II, and II only none of them QUESTION 10 Which of the ...
A: With the addition of water or hydration bof alkynes can be hydrated to form enols that spontaneousl...
Q: Arrange the 0.10m aqueous soultions of the following solutes in decreasing order. According to free...
A:
Q: 49. Initial velocity is used to study enzyme kinetics because A. The substrate concentration increas...
A: Initial velocity is it used to study enzyme Kinetics because as the reaction proceed the substrate o...
Q: Structural Diagram with Approximate Bond Angles Formula Lewis Structure NO2* NO2- NH4+ NO3- 4 – 4
A: The detailed explanation of the given question is given below. The given concept has been taken fro...
Q: meo
A:
Q: 85.9 grams of lithium react with hydrochloric acid. How many liters of hydrogen gas are produced? Th...
A: Given Reaction :- 2Li + 2HCl -------> 2LiCl + H2 Mass of Li = 85.9 g Atomic mass of Li = 7 g/m...
Q: Question 4 0.21 g sample of [Co(acac)3] was treated with concentrated HCI, then the Co ion was preci...
A: To calculate the percentage of cobalt in the sample, we have to find the amount of cobalt that was p...
Q: A mixture of helium and oxygen gas is compressed from a volume of 53.0L to a volume of 20.0 L, while...
A:
Q: ) On any clean sheet of paper, draw both the condensed and line-bond structure of the following comp...
A: To draw both the condensed and line-bond structure of the following compounds.,
Q: Balance these net ionic equations. (a) Ag+(aq) + Br¯(aq) → AgBr(s) (b) Cd²*(aq) + S²-(aq) – (c) Sc3+...
A:
Q: Decide whether each molecule or polyatomic ion is polar or nonpolar. If the molecule or polyatomic i...
A:
Q: Write the K, expressions for the following two reactions. 1st attempt Part 1 Do not include states o...
A:
Q: Why does the rate of a chemical reaction increase with a rise in the temperature of the reactants? ...
A: Given statement is : Why does the rate of a chemical reaction increase with a rise in the temperatur...
Q: CH
A:
Q: What is the value of pKa if H30+ and CN- equal 6.30E-6
A: For the reaction., HCN(aq.) + H2O(l) <===> H3O+(aq.) + CN-(aq.) Given that :- Conc. Of HCN = 0...
Q: MnO, + Fe+2 + H+ → Mn++ 4 + Fe3+ + H20
A: Answer: There are two methods to balance a redox reaction: 1. Ion-electron method 2. oxidation numbe...
Q: Between the chlorination and bromination (with light) of 2-methylpropane (HC(CH3)ɔ), explain which r...
A: A question based on alkanes that is to be accomplished.
Q: (a) For a sample of gas at constant temperature, its pressure multiplied by its volume is a constant...
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for y...
Q: A mixture of xenon and argon gas is expanded from a volume of 19.0L to a volume of 39.0L, while the ...
A:
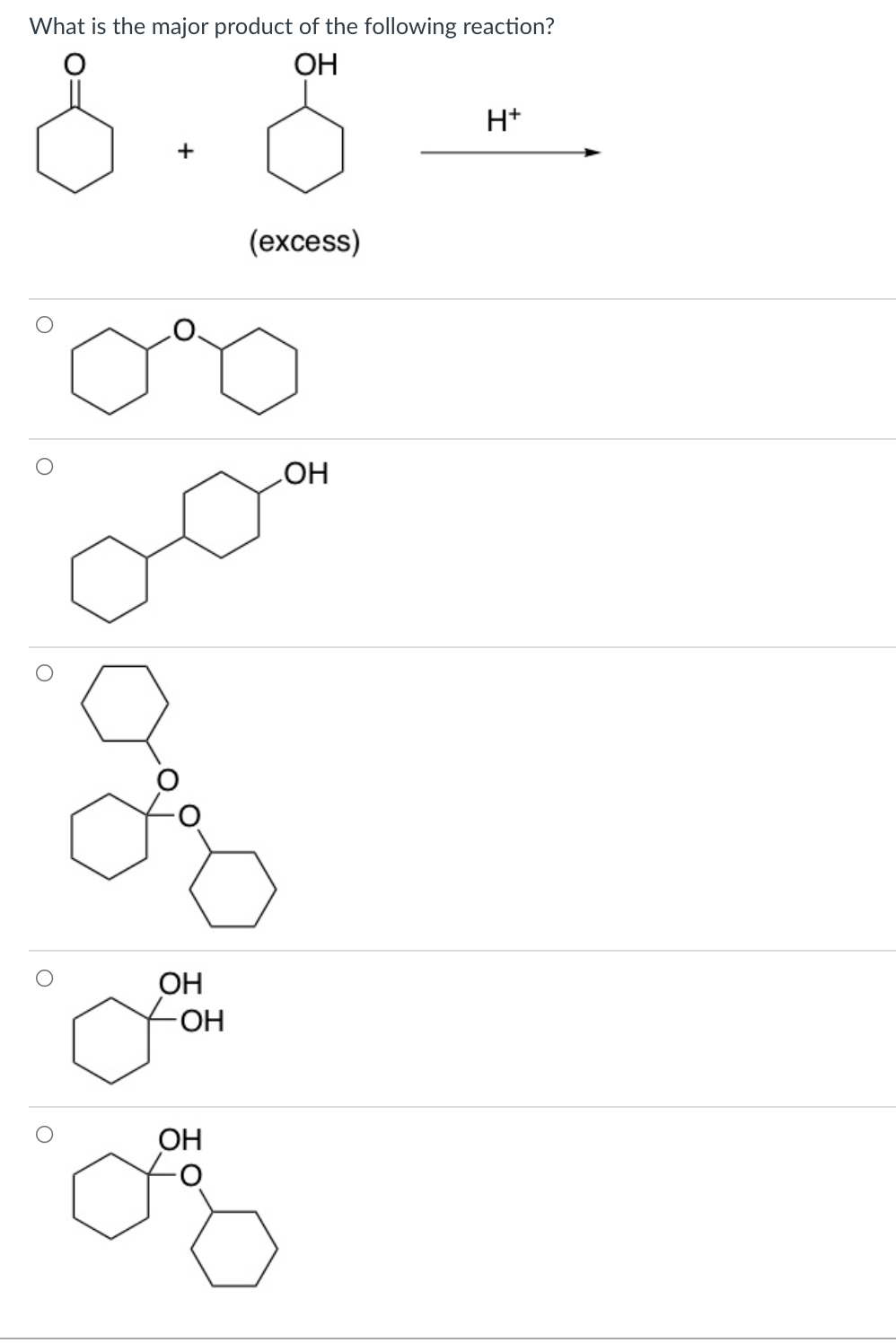
Q: Identify the major product(s) for the following reaction. Select all that apply. If the reaction doe...
A:
Q: Consider the reaction, N2O4(g) ⇌ 2 NO2(g) where the value for Kc is 2.0 x 10-6. The initial concentr...
A:
Q: What are the four different kinds of crystals/solids? Which unit particles make up each sort of crys...
A: Solids are classified into four categories According to the different type of particles and intermol...
Q: Choose all the correct names. There may be more than one. 2,5-dimethyl-3-hexyne 4,4-dimet...
A:
Q: Use the properties of the exact differential and the delining equations for the derived thermodynami...
A: we know certain relation and exact differential which can be used to proof the given relation
Q: In lab, you observed and classified multiple reactions. You mix a clear blue solution and a clear ye...
A: To select the option that satisfy that a chemical reaction is not take place.,
Q: rusting
A: In this question we have to explain that Why is rusting occurs rapidly in moist conditions as compar...
Q: State the relationships between the pressure of the gas and the number of gaseous particles.
A: If the number of particles increases the chance of collision with the walls of the container increas...
Q: The following precipitation reaction is performed using 2.000 g of each of the reactants. Fill in th...
A: The mass of the reactants (CaCl2 and Na3PO4) = 2.000 g
Q: A mixture of helium and oxygen gas is compressed from a volume of 96.0 L to a volume of 23.0 L, whil...
A:
Q: Calculate the molar concentration of OH− ions in a 0.077 M solution of ethylamine (C2H5NH2; Kb=6.4x1...
A: Given, Concentration of the ethylamine solution (C) = 0.077 M Kb for ethylamine (C2H5NH2) = 6.4x10-4...
Q: Determine whether each of the following salts will form a solution that is acidic, basic, or pH-neut...
A: The salts given are FeCl3 C6H5NH3NO2 NH4Br KClO2 NaBr
Q: Which of the following is much more acidic?
A: Option B is correct answer.
Q: As mentioned previously, molecules attract each other. The forces of attraction between molecules ar...
A:
Q: Drow the most electron-rich carbon atom in ho Compound below:
A: We have to determine the most electron-rich carbon atom in the given atom.
Q: Consider these compounds: A. Baco3 B. Baso4 E. Co(OH)2 D. MGF2 Complete the following statements by ...
A: Like dissolves like. Generally, solubility depends on the polarity of the molecule. Polar molecules ...
Q: explain this diagram
A: The diagram here depicts Ocean Acidification and carbonate ion formation essential for building bloc...
Q: Determine the molar concentration of Cl^{-} ions in 6.16E-1 M CaCl_{2}(aq).
A:
Q: How does the boiling point change as the formula mass of compound increases?
A: To solve this problem we have to know about boiling point of organic compounds.
Q: CO(g) + Cl2(g) COCI2(g) The above reaction obeys the mechanism: Cl2 2CI Fast equilibrium CI + CO COC...
A: The above mechanism is formation of COCl2. Where slowest step is rate determining step. Option C is ...
Q: H3O* CIF3 PF5
A:
Q: b) The equilibrium constants of K2 and K3 are given in the following equations. Determine the equili...
A:
Q: Question 5
A: The functional group is a key constituent that furnishes the characteristic chemical reaction to the...
Q: The rate of chemical reaction is affected or influenced by several factors. SATA * choose 5 Temp...
A:
Q: Gypsum is the hydrate of calcium sulfate, CaSO4 · 2 H2O. It is the primary material in sheet rock, ...
A: Given, Gypsum is the hydrate of calcium sulfate, CaSO4 · 2 H2O Molar mass of Ca = 40.078 g/mol Mo...
Q: ESTION 2 the addition of hydrogen bromide to alkynes in the absence of peroxides, which of the follo...
A: The reaction of the Alkyne with the hydrogen bromide in the absence of the hydrogen peroxide is call...
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Which product would likely be the major product for the following reaction?a. Cb. Dc. Ad. B6 H+ + 6 I- + ClO3- → 3 I2 + 3 H2O + Cl- What is the oxidizing reagent in this reaction?Consider the series of the trans effect: CO, CN-, C2H4 > PR3, H-, CH3- > C6H5- > NO2-, SCN-, I- > Br- >Cl- > py > NH3 > H20 What would be the major product of the following reaction? Select one:
- What is the final product of the following reactions? A) I B) II C) III D) IVWhat would be the majority product of the following reaction?: [Pt(NH3)4]2+ + 2 Cl- → ? Select one:a. No reaction because chloride is not a good inbound binder b. trans-[PtCl2(NH3)2] c. cis-[PtCl2(NH3)2] d. [PtCl(NH3)3]+illustrate how each reactant appeared prior to the reaction and how the product appeared after the reaction. 2Na+2H₂O --> 2NaOH+H₂
- Which reagent(s) would accomplish the following reaction? 1) LiAlH4 and 2) H2O 1) CH3MgBr and 2) H2O H2O, H+ NaH NaOH, H2OChoose the best reagents to complete the following reaction. Option E is (CH2OH)2, TSOH (Please show reaekson and don't use hend raiting please)Which product would likely be the major product for the following reaction?a. Bb. Cc. Ad. D
- Choose the best reagents to complete the following reaction. There's another answer choice not shown on screen: E) 1. CH3Li 2. H3O+ Hint: The correct answer is not option C.What is the major product of the following reaction? 1 2 3 4 NdaChoose the correct product to complete the following reaction: HNO3 + H2O → ________ + NO3- OH- H+ K+ H3O+

