Q: What mass of NaCl is present in 125.0 mL of a 0.100 M NaCl solution?
A: Volume of solution in L = 125/1000 = 0.125 L Since molarity = moles of solute / volume of solution…
Q: How many moles of ammonia are in 375. mL of a 2.25 M aqueous ammonia solution?
A: Given values are, Molarity= 2.25 M = 2.25 mol/L Volume of solution = 375 mL=0.375 L
Q: What volume in L of a 0.724 M Nal solution contains 0.405 mol of Nal ?
A:
Q: What is the molarity of a solution that contains 28 g of NaNO3 in 3.2 L of solution?
A: Given:Mass of NaNO3 = 28 g.Volume of NaNO3 solution = 3.2 L
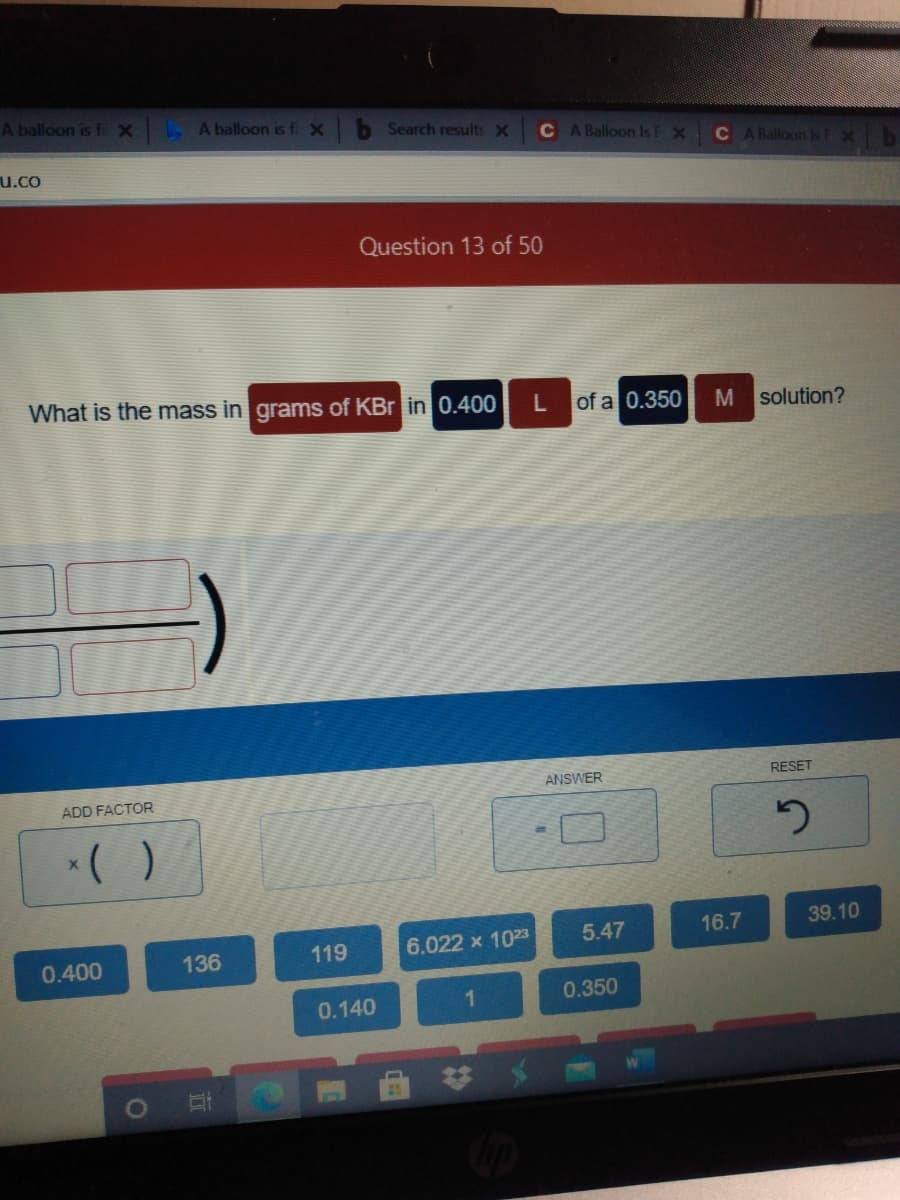
Q: How many grams of potassium chloride are needed to prepare 3.964 liters of a 0.2679 M solution?
A:
Q: What volume of 0.48 M NaCl solution contains 175.6 grams of NaCl?
A: First, calculate no of moles of NaCl No of moles of NaCl = Mass/Molar mass = 175.6g/(58.44gmol-1) =…
Q: What is the volume of a 2.4 M solution that contains 72.3 grams of TiO?
A:
Q: What is the molarity of a solution that contains 5.41 grams of magnesium oxide in a solution with a…
A:
Q: How many moles of HCl are in52.3 mL of a 1.17 M HCl solution?
A:
Q: How many grams of NaCl are present in 1263 ml of a 1.09 M solution?
A: Given , Volume of NaCl solution =1263 ml Molarity of solution = 1.09 M
Q: How many grams of NaCl are in 250 mL of a 3.5 M of NaCl solution?
A:
Q: How many moles of KI are present in 850 mL of a 1.55 M KI solution?
A: Molarity (M) can be defined as a moles of solute in per litre of solution. The S.I. unit for…
Q: How many moles of Calcium chloride are in 40.1 mL of a 3.0 M of CaCl2 solution?
A:
Q: How many grams of calcium chloride are needed to make 382 mL of a 2.54 M solution?
A: We have Molarity as 2.54 M, volume of solution as 382 ml, we have to calculate the mass of CaCl2…
Q: How many grams of calcium nitrate are in 134.7 mL of a 0.3212 M solution?
A: Molarity(M) is the moles of solute per unit volume of the solution in liters. It's unit is mol/L…
Q: How many grams of KOH are there in 0.520 L of a 0.033 M KOH solution?
A: Molarity=Number of molesVolume in liter of the solution
Q: How many moles of NH4SO4 are needed to make 3.45 L of a .250 M solution?
A: Moles of NH4SO4 can be calculated as follows
Q: moles of NaOH in 0.500 L of a 0.600 M NaOH solution
A: Given, Molarity = 0.600 M Volume = 0.500 L Formula, Molarity = (Moles / Volume) To calculate the…
Q: How many mL of a 9.4085 M CUSO4 solution contain 0.435 g CuSO4?
A: Concentration of CuSO4= 9.4085 M Mass of CuSO4 =0.435 grams Molar mass of CuSO4 =159.609 g/mol
Q: a chemist wants to make 5.5l of a 0.300 M CaCl2 solution. What mass of CaCl2 (in g) should the…
A: Given: Volume of solution = 5.5 L And concentration of CaCl2 = 0.300 M
Q: How many grams of sodium chloride (NaCl, 58.44 g/mol) are needed to make up 250. mL of a solution…
A: The molarity of a given solution is defined as the number of moles of the solute present in per…
Q: How many moles of NaF are in 35.8 grams of a 48.5% by mass NaF solution?
A: The mass of solute in gram present in 100 g solution is known as the mass percent concentration of a…
Q: How many grams of KOH are present in 29.5 mL of a 4.90 M KOH solution?
A: The number of moles of a solute (N) present in V mL of C (M) solution is calculated by using the…
Q: How many grams of strontium nitrate, Sr(NO3)2, are needed to prepare 3.40 x 10^3 mL, of a 0.925 M…
A: The number of moles of solute to the volume of solution in (L) is known as molarity.
Q: How many moles of NaCl are required to make 250 mL of a 3.00 M solution?
A: The given information are recorded as shown below,
Q: What volume in milliliters of a 0.30 M solution of glucose contains 0.025 mol of glucose?
A: From question data Molarity of glucose = 0.30 M Moles of glucose = 0.025 moles Formula used…
Q: How many grams of solute are in 0.428 L of 0.450 M K2CO3 solution?
A: The number of grams of solute is in 0.428 L of 0.450 M K2CO3 solution is calculated as,
Q: How many grams of potassium chloride are needed to prepare 3.607 liters of a 0.4464 M solution?
A:
Q: What volume of a 0.150 M NaCl solution contains 0.0500 moles of NaCl?
A:
Q: How many grams of solid potassium chloride KCl are needed to prepare 250 ml of a 1.50 M solution?
A: Applying molarity equation M =m/VL
Q: How many grams of calcium chloride (CaCl2) are in a 1 M calcium chloride solution?
A:
Q: What is the mass in grams of NaCN in 120.0 mL of a 2.40 x 105 M solution?
A: Definition: Molarity is a concentration term for a solution. The molarity of a given solution is…
Q: How many grams of CaCl2 were used to prepare 0.250 L of a 0.143 M CaCl2 solution?
A: Given :- molarity of CaCl2 solution = 0.143 M or 0.143 mol/L Volume of solution = 0.250 L To be…
Q: How many moles of NaCl are in 500.00 mL of a 1.25 M solution of NaCl?
A: Mole is a standard unit for measuring large quantities of very small entities such as molecules,…
Q: What mass of NaCl in grams is needed to prepare 2,500 g of 1.5%(m/m) NaCl solution?
A: Given data: Mass of the solution=2,500 g. The percent by mass of NaCl solution=1.5%. To calculate:…
Q: How many grams of Calcium Chloride in 100.0 mL of 1.8 Moles solution?
A: The volume of the solution is = 100.0 mL The molarity of the solution is = 1.8 M The mass of calcium…
Q: How many grams of solid potassium chlorate (KClO3, 122.55 g/mol) are needed to make 150 mL of 0.50 M…
A: Given data, Molarity = 0.50 M Volume = 150 mL (0.15L) Molar mass of potassium chlorate = 122.55…
Q: How many moles of NaOH are present in 25.00 mL of 0.500 M NaOH solution?
A: Given, the volume of solution =25.00 mL= 0.025 Lconcentration of NaOH solution =0.500 Mwe are asked…
Q: How many grams of potassium nitrate are in 11 ml of a 1.25 M solution?
A:
Q: There are 342 g of sucrose in 1.00 mol of sucrose. What is the molar concentration (molarity) of a…
A: Given Mass of sucrose = 171 gram Volume of solution = 1.75 Liter Molar mass of Sucrose =…
Q: How many moles of potassium bicarbonate, KHCO3, are present in 75.0 mL of a 0.750 M potassium…
A: molarity is calculated by number of moles of solute dissolved in one liter of solution.…
Q: How many grams of NaOH are in 0.930 L of a 0.500 M NaOH solution?
A: The particular amount of substance (solute) that undergoes dissolution in a liter (L) of solution is…
Q: How many grams of NaCl are needed to prepare 4.36 liters of a 1.601 M solution?
A: Molecular wt of NaCl is 58.44 g /mol 1molar NaCl solution means 1 mole NaCl present in 1 liter of…
Q: How many moles of calcium carbonate are there in a 2.50 L of a 3.44 M solution?
A: Given: Volume= 2.50 L Concentration= 3.44 M
Q: How many grams of NaOH are in 77.1 mL of a 5.59 M NaOH solution?
A:
Q: What volume of 0.48 M NaCI solution contains 175.6 grams of NaCl?
A: Given: Mass of NaCl (solute),w = 175.6 g Molarity of NaCl solution,M=0.48 M = 0.48 mol/L We know,…
Q: How many grams of potassium nitrate are in 25.8 ml of a 1.25 M solution?
A: Molarity of a solution is given by: Molarity=moles of solutevolume in L Moles of a solute can be…
Q: Barium chloride (BaCl2) gives green colors to fireworks. How many moles of BaCl2 are contained in 20…
A:
Q: What is the molarity of a solution that contains 2.30 g of H2C2O4⋅2H2O in 200. mL of solution?
A: Given: mass of H2C2O4⋅2H2O = 2.30 g molar mass of H2C2O4⋅2H2O = 126.07 g/mol volume of solution =…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- A mass of KHP is measured at 0.3051g and contains ______ moles. Group of answer choicesThe percentage of an additive in gasoline was measured six times with the following results: 0.13; 0.12; 0.16; 0.17; 0.20 and 0.11%. What is the 99% confidence interval for the additive percentage?The density or specific gravity of the liquid can be used in computing the equivalent weight of the reactant given in milliliters. A. True B. False
- Given the following data forMass of test tube, beaker and cyclohexane = 100.17 gMass of test tube and beaker = 84.07 gFreezing point of cyclohexane = 6.59 oCMass of weighing paper + naphthalene =1.080 gMass of weighing paper = 0.928 gFreezing point solution = 5.11oCKf = 20.8oC/mDetermine the followinga. mass of cyclohexane in g (2 decimal places); _____b. mass of naphthalene in g (4 decimal places); _____c. freezing point depression (2 decimal places); _____d. molality of solution (3 significant figures); _____e. moles of naphthalene (3 significant figures); _____f. molar mass of naphthalene, experimentally (3 significant figures); _____g. % error if theoretical molar mass of naphthalene is 128.17 g/ mole, USE ABSOLUTE VALUE (3 significant figure); ____Kernels of popcorn were weighed and then heated to pop. Water escaped in the process of popping. DATA DESCRIPTION TRIAL 1 TRIAL 2 Mass of Empty Evaporating Dish 35.6450g 35.6452g Mass of Evaporating Dish + Kernels Before Heating 42.3512 g 44.9572 g Mass of Evaporating Dish + Kernels After Heating 41.2540 g 42.6571 g What is the average percentage of water in the kernels? 65.180% 28.719% 21.208% 3.8913%Medicare does not cover the cost of this prescription medicine, which currently averages $297.78 per kilogram. The theoretical yield for the pure barium sulfate you were transferring was 500.00kg. You spilled 27.45 kg. What is the % yield of the transfer that you need to report? And also determine the consumer price of the spilled barium sulfate.
- If the mass of the container in the sample exercise (Figure 1.26)were measured to three decimal places before and after pumpingout the gas, could the density of the gas then be calculated tofour significant figures?8. Conversion problema. 25.345 μg/ml is converted to kg/cm3 b. 203.77 miles/sec is converted to cm/nsecSubtract 3.58 * 10 ^ - 2 kg from 1.41 * 10 ^ 11 ng and report your answer in g
- Solve for the the Dimensional Analysis (Predicted Volume of NaOH) Using the Data below:Given : Balanced Chemical Equation:NaOH (aq) + KHC8H4O4 (aq) → KNaC8H4O4 (aq) + H2O (l) Note: KHC8H4O4 = KHP Standardization of Sodium Hydroxide- Primary Standard used: 204.22 g/mol- Formula Mass of Primary Standard: Potassium hydrogen phthalate (KHC8H4O4)- % Purity of Primary Standard: 99.5% TRIALS 1 2 3 Actual weight of Std. KHP (g) 0.8026 0.8026 0.8021 Corrected weight of KHP (g) based on % purity of primary std. 0.7986g 0.7992g 0.7981gA student obtained 2.111 g of a mixture containing four components. After performing separation by chemical and physical methods, the student obtained the following mass data: Component 1: 0.486 g Component 2: 0.317 g Component 3: 0.127 g Component 4: 0.929 g Calculate the total percent recovery of the mixture. (Please show work)How can I convert 728.75468cm/s2 to N/kg ????



