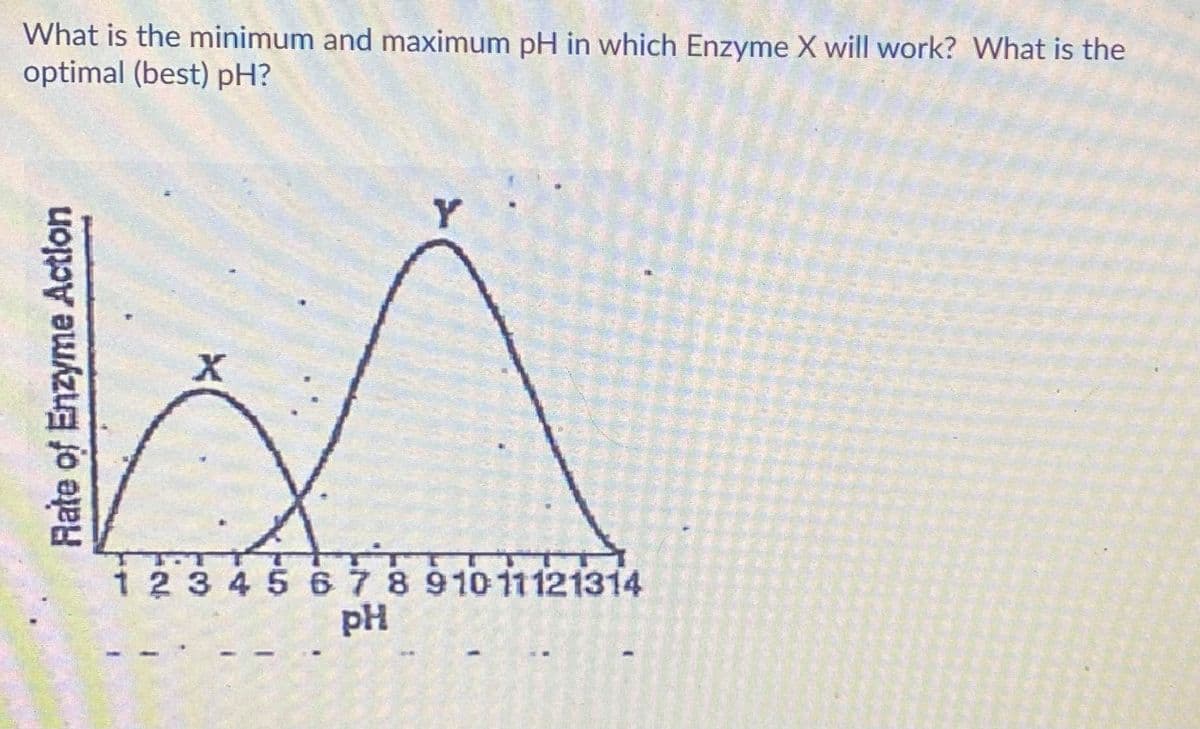
What is the minimum and maximum pH in which Enzyme X will work? What is the optimal (best) pH? Y X 1 2 3 45 67891011121314 pH Rate of Enzyme Action
Q: In metalloenzymes metals are Attached to enzyme through coordinate bonds. o Covalently attached to…
A: A coordinate bond is a covalent bond which formed by sharing a pair of electrons. It is also known…
Q: The line does not cross the X-axis when the Y-variable is equal to zero 1점 because Why does this…
A: The picture is showing the graph between the rate of a reaction and the enzyme concentration.
Q: Which of the following statements about competitive inhibitors of an enzyme catalyzed reaction is…
A: Enzymes are the molecules with speed up the rate of a biological reaction without being consumed in…
Q: (a) (c) 1/v + inhibitor 1/[S] (b) + inhibitor X 1/v 1/[S] + inhibitor + inhibitor (d) 1/v 1/V I i…
A: Non competitive inhibition is a type of inhibition where the inhibitor binds to a site other than…
Q: Enzyme C Substance X + Substance Y Substance W What data should be collected to support the…
A: Note: According to the guidelines, we are supposed to answer only one question. Please repost other…
Q: the enzyme and substrate should be mixed only when the reaction is initiated (when the absorbance…
A: Kinetics is the branch which deals with rate of changes. From above all given situations in the…
Q: B. X+Y Reaction Coordinate at does letter B represent? Letter B represents the activation energy of…
A: Enzymes are usually proteins which are never used up in the reaction. Their main function is to…
Q: 10 20 30 40 50 60 70 80 Temperature (°C)
A: Enzyme activity is influenced by many factors such as temperature, pH, concentration of the…
Q: which of the following is a primary function of the active site of an enzyme?
A: Given: Enzymes are the largest and most specialised class of protein molecules. They are composed of…
Q: 3. 4. 2 3 4. 5 6 7 8 pH of reaction According to Figure 1.7 which enzyme is most likely found in a…
A: The enzymes are biomolecules that catalyze biochemical reactions. These enzymes are very sensitive…
Q: What is the difference between the left and right? What type of inhibition is being shown? What are…
A: Note: Since you have posted multiple independent questions in the same request, we will solve the…
Q: True or False In the presence of enzymes, the value of free energy of activiation (delta G°‡) for…
A: Enzymes are biocatalysts that lower the activation energy and in turn, increase the reaction rate.…
Q: Define
A: Enzyme kinetic is a branch of Biochemistry. There are several factors that enzyme catalysis are…
Q: Which of the following is incorrect? a. Without an enzyme, reaction rate can be increased by…
A: Enzymes are the catalysts of biochemical reactions that increase the rate of reaction. Enzymes have…
Q: -Inhibitor +Inhibitor [S] (mM) Vη&νβσπ:(μmol/sec). V0&νβσπ&ν βσπ (μmol/sec) 0.0001 33 17 0.0005 71…
A: Km is the michaelis menton constant which is a substrate concentration at half Vmax. This can be…
Q: Given the active site and reaction mechanism below, what is the mechanism of irreversible inhibition…
A: The given example is uncompetitive inhibition. Uncompetitive inhibition, also known as…
Q: 8). Which statement best describes 1 point the enzyme represented in the graphs? * O 10 20 30 40 60…
A: Enzymes These are biological catalysts that speed up the rate of the biochemical reaction. Most…
Q: 0 10 20 30 40 50 60 70 temperature / °C a. What is the optimal temperature for this enzyme? b. What…
A: The enzyme is a substance found in living beings that works as a catalyst, controlling the speed at…
Q: Why does an optimal pH promote enzyme activity
A: Enzyme is the substance or molecule that act as a catalyst which regulates the rate at which…
Q: 8. Optimum (BEST) temperature for the action of this enzyme (graph below) is approximately 1. 50 1.…
A: Enzymes are very specific for their substrates and reaction conditions. These function at a specific…
Q: Carboxylesterase 0 1 2 3 4 5 6 7 pH Enzyme activity -
A: Carboxylesterases are a group of enzymes that hydrolyze compounds containing a functional group such…
Q: Which of the following can cause the rate of enzyme activity to increase? Increasing the temperature…
A: Catalysts are those substances that increase the rate of the reaction without undergoing any change…
Q: In which figure (s), the type of inhibition is reversible? Substrate Figure 1 O Figure 1 O Figure 2…
A: The active site of the enzyme is the site where the specific substrate can bind according to the…
Q: You measure the initial rate of an enzyme reaction as a function of substrate concentration in the…
A: Enzyme kinetics follow Michealis Menton equation which is Vo = Vmax[S]/(Km + [S]). It can be…
Q: ion of estion udy As you increase the amount of substrate in a reaction (while keeping the enzyme…
A: Enzymes are the biocatalyst that are responsible for reducing the activation energy. The product…
Q: The optimum temperature for enzyme activity in the body: O A. OC B. 10C O c. 40C O D. 60C 47
A: Introduction :- Proteins called enzymes aid in accelerating our bodies' chemical reactions, or…
Q: Enzyme X and enzyme Y catalyze the same reaction and exhibit the νo versus [S] curves shown below.…
A: Enzyme kinetics reaction comprises of the interaction between the enzyme and the substrate…
Q: Environmental Factors and Enzyme Activity LA 15- 10 5- 10 20 30 40 50 60 Temperature ("C) 9 10 11 12…
A: Enzymes are biological molecules that are a class of proteins except for ribozymes (a class of RNA)…
Q: Figure 5.7 LLLLA Substrate concentration The graph at the left in Figure 5.7 shows the reaction rate…
A: Enzyme molecules are proteinaceous biocatalyst that lowers the activation energy of the reaction and…
Q: Which of the two graphs, below, represents an enzyme catalyzed version of the reaction Y-->X?…
A: A catalytic reaction is a reaction where the reactants will form the products with the help of a…
Q: 100 increasing enzyme ativity 50 optimum pH 5 6 7 9 10 11 8 pH 20 40 Temperature 1. How do pH and…
A: Every enzyme exhibits maximum activity over the certain pH range in which a molecule exists in its…
Q: Which factor can impact enzyme activity? Select all that apply. Group of answer choices pH…
A: Enzymes are the biocatalysts that speed up the rate of any biochemical reaction in the body. They…
Q: An Eadie-Hofstee plot is shown below for the different types of enzyme inhibition. Match the line…
A: Eadie-Hofstee diagram also called as Woolf-Eadie-Augustinsson-Hofstee or Eadie-Augustinsson plot is…
Q: Most enzymes are very sensitive to even small changes in pH. Explain why a slight shift to more…
A: Enzymes are very crucial in all reactions in our body for metabolism and catabolism.
Q: Which of the following statements describes an enzyme ALLOSTERIC site? O It is where an inhibitor…
A: Allosteric site is the site that allows enzyme activity to be activated or inhibited by molecules.
Q: Which of the following factors does NOT always alter the activity of the enzyme? Reactant…
A: INTRODUCTION Enzymes Enzymes are biological catalyst. Enzymes are proteins that increase the…
Q: If a drug is built to bind to the active site of the enzyme, it will prevent substrate binding. This…
A: Enzyme are biocatalysts that enhance the biochemical reactions in our body. They are made up of 2…
Q: Identify the type of enzyme inhibition each of the following inhibitor characteristics is associated…
A: Enzymes are proteinaceous molecules that help in the catalysis of biochemical reactions. Most of the…
Q: Which of the following would be required for an irreversible enzyme inhibitor? O lonic interactions…
A: Introduction: Enzyme inhibitors are molecules or compounds that bind to enzymes and reduce the…
Q: Graph #4 Substrate concentration #4, the solid line represents the rate of an enzyme reaction with…
A: An inhibitor is any molecule which binds to the enzyme and decrease its catalytic efficiency.
Q: Which figure represents the non- competitively inhibited enzyme? Figure 1 Suhatrate Product Enzyme O…
A: Enzymes are essential components of all metabolic processes in the body. They are catalytic…
Q: Please choose one of these answers A. An allosteric inhibitor appeared B. There was a dramatic…
A: Enzymes are the molecules which speed up the rate of biological reactions without being consumed in…
Q: kcat is: a measure of the catalytic efficiency of the enzyme the rate constant for the…
A: All biochemical reactions are catalyzed by specialized proteins. They act as catalysts for these…
Q: The apo form of an enzyme: (select all that applies) Group of answer choices Is the cofactor-free…
A: Enzymes are biological catalysts which can only function in specified pH, pressure, and temperature…
Q: Which of the following is NOT TRUE about allostery? O The enzyme changes conformation upon binding…
A: When a chemical reaction occurs, a catalyst is a substance that accelerates the process without…
Q: Which of the following factors affect enzyme activity? O a. All of the mentioned answers are correct…
A: Enzymes are biological catalysts that increases the rate of chemical reaction without itself being…
Q: Below which is false about enzyme and catalysis? O Catalytic residues interact with substrates and…
A: Enzymes are known as biological catalysts in which help to catalyze the different biochemical…
Q: Identification of the active site of an enzyme: 1.6 The effect of changing pH
A: Enzymes are biological catalysts that carry out the essential biochemical reactions without…
Q: Which of the following statements is true about effect of substrate concentration on enzyme activity…
A: The formation of an enzyme-substrate complex is the first step in enzymatic catalysis. The enzyme…
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

- Which of the following environmental conditions may have aninfluence on enzyme activity?a. substrate concentrationb. temperaturec. pHd. All of these are correct.Identify the type of enzyme inhibition each of the following inhibitor characteristics is associated with: 1. An inhibitor that decreases enzyme activity by binding to a site on the enzyme other that the active site. 2. An inhibitor that inactivates enzymes by forming a strong covalent bond of the enzyme acitve site.an enzyme acts on a substrate X. The enzyme exists in four different forms, with different catalytic efficiencies. The table shows the kcatand KM values for each form of the enzyme. If the concentration of substrate X in a solution is 5 µM, which of the four forms of the enzyme is the most efficient? Form of Enzyme kcat (s-1) KM (µM) A 50 10 B 50 1 C 100 4 D 1000 100 a. Form A b. Form B c. Form D d. Form C
- What are the description of the ff?A. Enzyme influence on reactionvelocityB. Effect of temperature onenzymatic activityGive a complete and well descriptive definition of the following:1.1 Enzyme catalysis1.2 Co-enzyme1.3 Negative heterotropic co-cooperativitChoose only the letter, no explanation needed. Enzyme activity is affected by a variety of factors. What factor causes the enzyme to denature if it becomes extremely high? * Choices: A. Water's Effect B. pH C. Temperature D. Activator's Effect An inhibitor binds to the enzyme's active site, preventing the substrate from binding to it. What conclusions can you make from this situation? * A. No reaction occurred B. Non-competitive inhibition occurred C. Enzyme activity occurred D. Competitive inhibition occurred Each enzyme is very selective when it comes to its substrate. What can you conclude from this statement? * A. Any substrate can bind to the active site. B. Enzymes are used up in the reaction. C. Only a specific substrate can bind to the active site. D. Enzymes break down when not used. Lock : Key :: Active Site : _____________________________ * A. Substrate B. Active Site C. Coenzyme D. Cofactor Enzymes only speed up biological functions, so they are NOT used up in the…
- A researcher has measured the initial rate of an enzyme-catalyzed reaction as a function of substrate concentration in the presence and absence of 0.001μM inhibitor. She obtains the following data. What is the Vmax and Km for the "no inhibitor" and "+ inhibitor" experiments. Ans, What would be value of V0 for the "+inhibitor" at [S] = 0.4μM and the inhibitor is most likely a competitive, uncompetitive or mixed inhibitor.Does the enzyme described in each of the following statementsrequire a cofactor to be active?(a) Ni2+ is present in the active site.(b) Addition of FAD allows the reaction to occur.(c) The presence of K+ does not affect the reaction.What would be the turnover number and specific activity of an enzyme (molecular weight=32,000 D) in a reaction. (Vmax= 4 μmol of substrate reacted/min), with enzyme amount = 2 μg
- Which of the following statements is TRUE of enzyme catalysts? for heads up question 2 and 3 are false 1) To be effective, they must be present at the same concentration as their substrate. 2) They can increase the equilibrium constant for a given reaction by a thousand-fold or more. 3) They lower the free energy for conversion of substrate to product. 4) Their catalytic activity is dependent on pH. 5) They are generally equally active on D and L isomers of a given substrate.An Eadie-Hofstee plot is shown below for the different types of enzyme inhibition. Match the line withthe type of inhibition.(a) Line A Inhibition Mechanism. Ans: ________(b) Line B Inhibition Mechanism. Ans: ________(c) Line C Inhibition Mechanism. Ans: ________the amino acid glutamic acid is at the active site of an enzyme. Normally the enzyme is active at pH 7. at pH 4 (higher concentration of H+), the enzyme is inactive. Explain there observations

