Chapter11: Reactions Of Alkyl Halides: Nucleophilic Substitutions And Eliminations
Section11.SE: Something Extra
Problem 40AP: The following Walden cycle has been carried out. Explain the results, and indicate where Walden...
Related questions
Question
what is the name of these condensed structural formulas
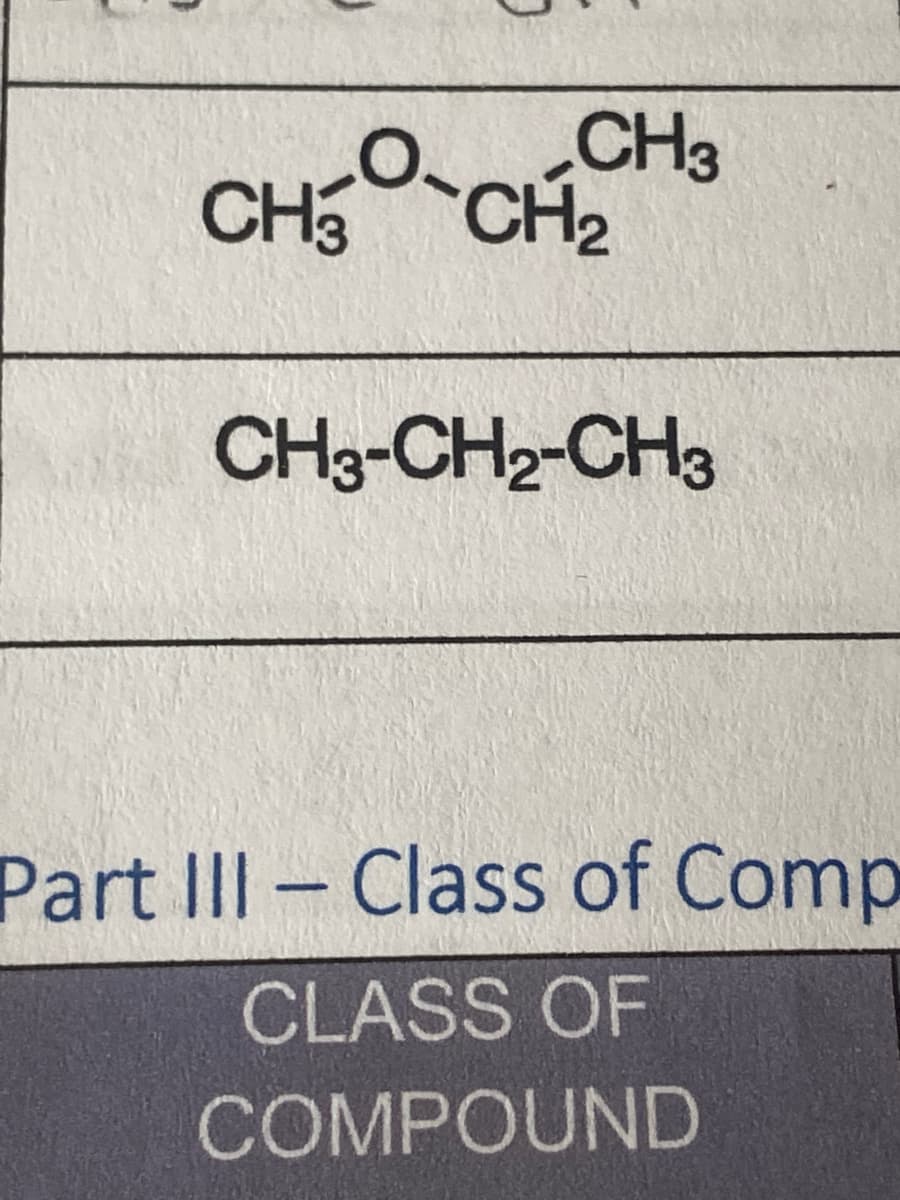
Transcribed Image Text:CHỖ CHỈ
CH₂
CH3-CH2-CH3
Part III Class of Comp
CLASS OF
COMPOUND
-
Expert Solution
Step 1
Since,
Rule of IUPAC-
1) Longest chain as parent chain.
2) Numbering start from those side where more prior group present.
3) First written name of substituent then after written parent name.
Thus,
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning