What is the numerical value of the activity coefficient of aqueous ammonia (NH,) at an ionic strength of 0.1? Use the linear interpolation of experimental data in Table 10-2 if necessary.
What is the numerical value of the activity coefficient of aqueous ammonia (NH,) at an ionic strength of 0.1? Use the linear interpolation of experimental data in Table 10-2 if necessary.
Chapter10: Effect Of Electrolytes On Chemical Equilibria
Section: Chapter Questions
Problem 10.18QAP
Related questions
Question
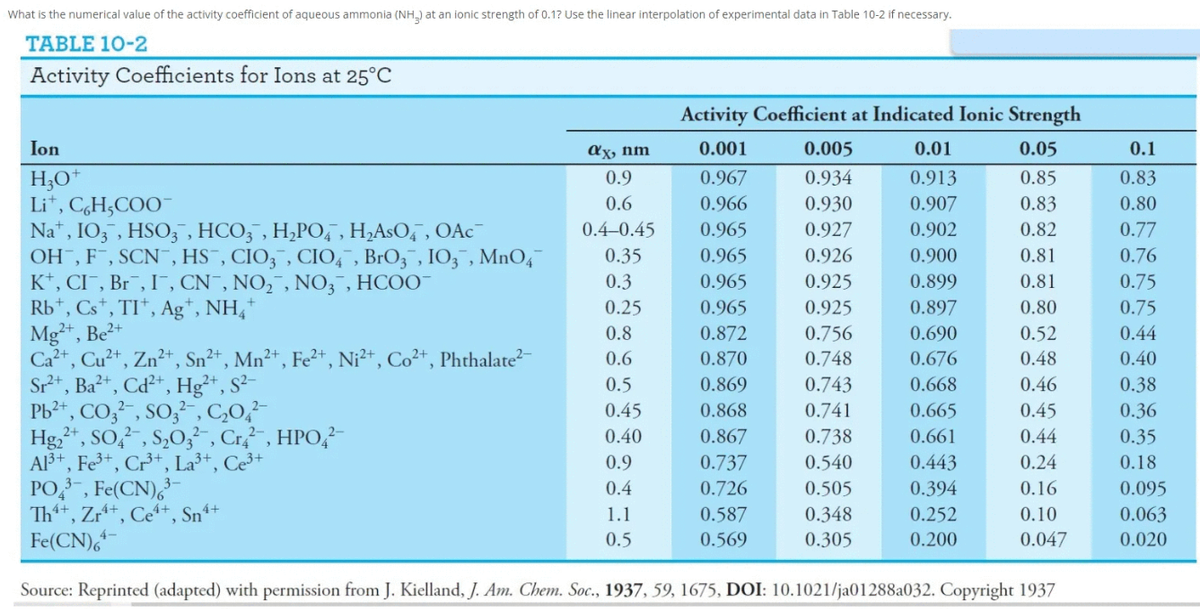
Transcribed Image Text:What is the numerical value of the activity coefficient of aqueous ammonia (NH_) at an ionic strength of 0.1? Use the linear interpolation of experimental data in Table 10-2 if necessary.
TABLE 10-2
Activity Coefficients for Ions at 25°C
Activity Coefficient at Indicated Ionic Strength
Ion
ax, nm
0.001
0.005
0.01
0.05
0.1
H;O*
Li*, C,H,COO¯
Na", IO, , HSO, , HCO; , H,PO, , H;AsO, , OAC¯
OH , F , SCN¯, HS¯, CIO3¯, CIO, , BrO3 , I03 , MnO,
K*, CI, Br, I, CN¯, NO,¯, NO; , HCOO-
Rb*, Cs*, TI*, Ag*, NH,*
Mg+, Be2+
Ca+, Cu+, Zn²+, Sn²+, Mn²+, Fe²*, Ni²*, Co²+, Phthalate²-
Sr*, Ba²+, Cd²+, Hg²*, S²-
Pb²*, CO,² , SO,²-, C,O,
Hg,*, SO,, S,0;, Cr, HPO,?-
Al3+, Fe³*, Cr+, La³+, Ce³+
PO, , Fe(CN),
Th+, Zr*+, Ce“+, Sn*+
Fe(CN),
0.9
0.967
0.934
0.913
0.85
0.83
0.6
0.966
0.930
0.907
0.83
0.80
0.4-0.45
0.965
0.927
0.902
0.82
0.77
0.35
0.965
0.926
0.900
0.81
0.76
0.3
0.965
0.925
0.899
0.81
0.75
0.25
0.965
0.925
0.897
0.80
0.75
0.8
0.872
0.756
0.690
0.52
0.44
0.6
0.870
0.748
0.676
0.48
0.40
0.5
0.869
0.743
0.668
0.46
0.38
0.45
0.868
0.741
0.665
0.45
0.36
0.40
0.867
0.738
0.661
0.44
0.35
0.9
0.737
0.540
0.443
0.24
0.18
0.4
0.726
0.505
0.394
0.16
0.095
1.1
0.587
0.348
0.252
0.10
0.063
0.5
0.569
0.305
0.200
0.047
0.020
Source: Reprinted (adapted) with permission from J. Kielland, J. Am. Chem. Soc., 1937, 59, 1675, DOI: 10.1021/ja01288a032. Copyright 1937
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

